Abstract
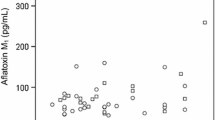
A study was conducted to investigate the frequency and levels of AFM1 and AFM2 in urine from children who attended the emergency service of a pediatric referral hospital in Bogota, Colombia. A survey on the consumption of foods likely to be a source of aflatoxins and on sociodemographic variables was conducted as well. The frequency of AFM1 in urine was found to be 41.7% with an average concentration in positive samples of 16 pg mL−1 ± 10.7 pg mL−1 (range > LOD–48.5 pg mL−1). The presence of AFM1 in the urine was related to the consumption of cereals likely to be contaminated with AFB1, especially corn and rice. No detectable levels of AFM2 were found in any sample. The results show that children’s exposure to aflatoxins in Colombia is indeed a problem and should be one of the priorities of the health authorities. Continuous monitoring of aflatoxins in foods should be carried out, in compliance with Colombian regulations, using analytical methods that allow determination and quantification of aflatoxins in different biological and non-biological matrices at trace levels.


Similar content being viewed by others
References
Ahn J, Kim D, Kim H, Jahng KY (2010) Quantitative determination of mycotoxins in urine by LC-MS/MS. Food Addit Contam Part A Chem Anal Control Expo Risk Assess 27:1674–1682. https://doi.org/10.1080/19440049.2010.505201
Ali N, Blaszkewicza M, Hossainb K, Dege GH (2017) Determination of aflatoxin M1 in urine samples indicates frequent dietary exposure to aflatoxin B1 in the Bangladeshi population. Int J Hyg Environ Health 220:271–281. https://doi.org/10.1016/j.ijheh.2016.11.002
Ayelign A, Woldegiorgis AZ, Adish A, De Boevre M, Heyndrickx E, De Saeger S (2017) Assessment of aflatoxin exposure among young children in Ethiopia using urinary biomarkers. Food Addit Contam Part A Chem Anal Control Expo Risk Assess 34:1606–1616. https://doi.org/10.1080/19440049.2017.1350290
Bianco G, Russo R, Marzocco S, Velotto S, Autore G, Severino L (2012) Modulation of macrophage activity by aflatoxins b1 and b2 and their metabolites aflatoxins M1 and M2. Toxicon 59:644–650. https://doi.org/10.1016/j.toxicon.2012.02.010
Céspedes AE, Diaz GJ (1997) Analysis of aflatoxins in poultry and pig feeds and feedstuffs used in Colombia. J AOAC Int 80:1215–1219
De Vries HR, Maxwell SM, Hendrickse RG (1990) Aflatoxin excretion in children with kwashiorkor or marasmic kwashiorkor—a clinical investigation. Mycopathologia 110:1–9
Diaz GJ, Perilla NS, Rojas Y (2001) Occurrence of aflatoxins in selected Colombian foods. Mycotoxin Res 17:15–20
Diaz GJ, Sánchez MP (2015) Determination of aflatoxin M1 in breast milk as a biomarker of maternal and infant exposure in Colombia. Food Addit Contam Part A Chem Anal Control Expo Risk Assess 32:1192–1198. https://doi.org/10.1080/19440049.2015.1049563
Diaz GJ, Krska R, Sulyok M (2015) Mycotoxins and cyanogenic glycosides in staple foods of three indigenous people of the Colombian Amazon. Food Addit Contam Part B Surveill 8:291–297. https://doi.org/10.1080/19393210.2015.1089948
Ediage EN, Di Mavungu JD, Song S, Sioen I, De Saeger S (2013) Multimycotoxin analysis in urines to assess infant exposure: a case study in Cameroon. Environ Int 57:50–59. https://doi.org/10.1016/j.envint.2013.04.002
EMEA – European Medicines Agency (2011) Guideline on bioanalytical method validation. EMEA/CHMP/EWP/192217/2009 Rev. 1 Corr. 2**. Available from: https://www.ema.europa.eu/en/bioanalytical-method-validation
EPA - US Environmental Protection Agency (1984) Guidelines establishing test procedures for the analysis of pollutants under the clean water act.40 cfr, appendix b to part 136: definition and procedure for the determination of the method detection limit. Washington (DC): USA. Available from: https://www.epa.gov/quality/guidelines-establishing-test-procedures-analysis-pollutants-code-federal-regulations-excerpt
Ezekiel CN, Warth B, Ogara IM, Abia WA, Ezekiel VC, Atehnkeng J, Sulyok M, Turner PC, Tayo GO, Krska R, Bandyopadhyay R (2014) Mycotoxin exposure in rural residents in northern Nigeria: a pilot study using multi-urinary biomarkers. Environ Int 66:138–145. https://doi.org/10.1016/j.envint.2014.02.003
Gerding J, Ali N, Schwartzbord J, Cramer B, Brown DL, Degen GH, Humpf HU (2015) A comparative study of the human urinary mycotoxin excretion patterns in Bangladesh, Germany, and Haiti using a rapid and sensitive LC-MS/MS approach. Mycotoxin Res 31:127–136. https://doi.org/10.1007/s12550-015-0223-9
Gross-Steinmeyer K, Eaton DL (2012) Dietary modulation of the biotransformation and genotoxicity of aflatoxin B1. Toxicology 299:69–79. https://doi.org/10.1016/j.tox.2012.05.016
IARC – International Agency for Research on Cancer (2012) Aflatoxins. In: Chemical agents and related occupations, Vol. 100F: a review of human carcinogens, pp. 225–248. Available from: https://monographs.iarc.fr/ENG/Monographs/vol100F/mono100F.pdf
Jager AV, Tonin FG, Souto PC, Privatti RT, Oliveira CA (2014) Determination of urinary biomarkers for assessment of short-term human exposure to aflatoxins in São Paulo, Brazil. Toxins 6:1996–2007. https://doi.org/10.3390/toxins6071996
Jolly P, Jiang Y, Ellis W, Awuah R, Nnedu O, Phillips T, Wang JS, Afriyie-Gyawu E, Tang L, Person S, Williams J, Jolly C (2006) Determinants of aflatoxin levels in Ghanaians: sociodemographic factors, knowledge of aflatoxin and food handling and consumption practices. Int J Hyg Environ Health 209:345–358. https://doi.org/10.1016/j.ijheh.2006.02.002
Kensler TW, Roebuck BD, Wogan GN, Groopman JD (2011) Aflatoxin: a 50-year odyssey of mechanistic and translational toxicology. Toxicol Sci 120:28–48. https://doi.org/10.1093/toxsci/kfq283
Kouadio JH, Lattanzio VM, Ouattara D, Kouakou B, Visconti A (2014) Assessment of mycotoxin exposure in Côte D′ivoire (Ivory Coast) through multi-biomarker analysis and possible correlation with food consumption patterns. Toxicol Int 21:248–257. https://doi.org/10.4103/0971-6580.155336
Makri A, Goveia M, Balbus J, Parkin R (2004) Children’s susceptibility to chemicals: a review by developmental stage. J Toxicol Environ Health B Crit Rev 7:417–435. https://doi.org/10.1080/10937400490512465
Piekkola S, Turner PC, Abdel-Hamid M, Ezzat S, El-daly M, El-kafrawy S, Savchenko E, Poussa T, Woo JC, Mykkänen H, El-Nezami H (2012) Characterisation of aflatoxin and deoxynivalenol exposure among pregnant Egyptian women. Food Addit Contam Part A Chem Anal Control Expo Risk Assess 29:962–971. https://doi.org/10.1080/19440049.2012.658442
Polychronaki N, Wild CP, Mykkänen H, Amra H, Abdel-Wahhab M, Sylla A, Diallo M, El-Nezami H, Turner PC (2008) Urinary biomarkers of aflatoxin exposure in young children from Egypt and Guinea. Food Chem Toxicol 46:519–526. https://doi.org/10.1016/j.fct.2007.08.034
Quattrocchi OA, de Adrizzi SA, Laba RF (1992) Introduccion a la HPLC: Aplicación y práctica. Buenos Aires, pp 301–328
Rojas E, Sarmiento F (2003) Pediatría diagnóstico y tratamiento, Colombia, p 445
Schwartzbord JR, Leroy JL, Severe L, Brown DL (2016) Urinary aflatoxin M1 in port-au-prince and a rural community in north-east Haiti. Food Addit Contam Part A Chem Anal Control Expo Risk Assess 33:1036–1042. https://doi.org/10.1080/19440049.2016.1185899
Sherif OS, Salama EE, Abdel-Wahhab MA (2009) Mycotoxins and child health: the need for health risk assessment. Intl J Hyg Environ Health 212:347–368. https://doi.org/10.1016/j.ijheh.2008.08.002
Warth B, Petchkongkaew A, Sulyok M, Krska R (2014) Utilising an LC-MS/MS-based multi-biomarker approach to assess mycotoxin exposure in the Bangkok metropolitan area and surrounding provinces. Food Addit Contam Part A Chem Anal Control Expo Risk Assess 31:2040–2046. https://doi.org/10.1080/19440049.2014.969329
WHO - World Health Organization (1995) Physical status: the use and interpretation of anthropometry. Report of a WHO Expert Committee. WHO Technical Report Series 854, pp. 9. Available https://apps.who.int/iris/bitstream/handle/10665/37003/WHO_TRS_854.pdf;jsessionid=974368CE703873214393AC8598E54C34?sequence=1
Williams JH, Phillips TD, Jolly PE, Stiles JK, Jolly CM, Aggarwal D (2004) Human aflatoxicosis in developing countries: a review of toxicology, exposure, potential health consequences, and interventions. Am J Clin Nutr 80:1106–1122. https://doi.org/10.1093/ajcn/80.5.1106
Acknowledgements
Thanks are due to all the patients who participated in this study and to the parents who approved the taking of the urine sample for the subsequent analysis.
Funding
The study was conducted with funds from the Toxicology Laboratory of the College of Veterinary Medicine, National University of Colombia.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
This study was approved by the Ethics Committee of the Faculty of Medicine, National University of Colombia (report no. 019-233-16) and the Ethics Committee of Fundación Hospital de la Misericordia (HOMI) (report no. 4522 CEI-52-16). An institutional consent was signed for the participation of patients, prior to the admission of this study.
Conflict of interest
No potential conflicts of interest were reported by the authors.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Sánchez, E.M., Diaz, G.J. Frequency and levels of aflatoxin M1 in urine of children in Bogota, Colombia. Mycotoxin Res 35, 271–278 (2019). https://doi.org/10.1007/s12550-019-00355-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12550-019-00355-x