Abstract
Background
Lenalidomide is used for the treatment of multiple myeloma in combination with dexamethasone. The purpose of this study was to compare the pharmacokinetics (PKs) and assess the bioequivalence of two formulations of lenalidomide 25 mg: Lenalid® 25 mg tablet (test formulation) and Revlimid® 25 mg capsule (reference formulation).
Methods
A randomized, single-dose, two-treatment, two-period, two-sequence crossover study was conducted in 42 healthy subjects. All subjects were randomly assigned to one of the two sequences, and they received a single dose of test or reference formulation in the first period and the alternative formulation during the next period under fasting conditions. Serial blood samples for PK evaluation were collected up to 24 h post-dose and the PK parameters were estimated by non-compartmental methods. Throughout the study, tolerability was assessed on the basis of adverse events, vital signs, and clinical laboratory tests.
Results
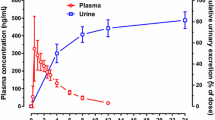
The test formulation showed similar PK profiles to those of the reference formulation. The geometric mean ratio and 90% confidence interval (CI) of the test formulation to the reference formulation for maximum plasma concentration (Cmax) was 0.9995 (0.9250–1.0799) and the corresponding value for the area under the concentration–time curve from time zero to time of last quantifiable concentration (AUCt) was 0.9648 (0.9451–0.9850). Both CIs were within the conventional bioequivalence range of 0.8–1.25. The tolerability profile was not significantly different between the two formulations.
Conclusion
This study found that the PKs of the two formulations of lenalidomide 25 mg were similar and the test formulation met the regulatory criteria for assuming bioequivalence with the reference formulation.
Funding
Samyang Biopharmaceutical Corp.


Similar content being viewed by others
References
Celgene Corporation. REVLIMID [lenalidomide] capsules, for oral use. Prescribing Information. 2017; https://media.celgene.com/content/uploads/revlimid-pi.pdf.
Zeldis JB, Knight R, Hussein M, Chopra R, Muller G. A review of the history, properties, and use of the immunomodulatory compound lenalidomide. Ann N Y Acad Sci. 2011;1222:76–82.
Zhu YX, Kortuem KM, Stewart AK. Molecular mechanism of action of immune-modulatory drugs thalidomide, lenalidomide and pomalidomide in multiple myeloma. Leuk Lymphoma. 2013;54:683–7.
Blumel S, Broadway-Duren J. Approaches to managing safety with lenalidomide in hematologic malignancies. J Adv Pract Oncol. 2014;5:269–79.
Chen N, Kasserra C, Reyes J, Liu L, Lau H. Single-dose pharmacokinetics of lenalidomide in healthy volunteers: dose proportionality, food effect, and racial sensitivity. Cancer Chemother Pharmacol. 2012;70:717–25.
Chen N, Wen L, Lau H, Surapaneni S, Kumar G. Pharmacokinetics, metabolism and excretion of [(14)C]-lenalidomide following oral administration in healthy male subjects. Cancer Chemother Pharmacol. 2012;69:789–97.
Chen N, Zhou S, Palmisano M. Clinical pharmacokinetics and pharmacodynamics of lenalidomide. Clin Pharmacokinet. 2017;56:139–52.
US Food and Drug Administration. Draft Guidance on Lenalidomide. 2013; https://www.fda.gov/downloads/drugs/guidancecomplianceregulatoryinformation/guidances/ucm326798.pdf.
European Medicines Agency. Lenalidomide hard gelatine capsules 2.5, 5, 7.5, 10, 15 and 25 mg product-specific bioequivalence guidance: Draft. 2015; http://www.ema.europa.eu/docs/en_GB/document_library/Scientific_guideline/2015/10/WC500195125.pdf.
Chen N, Ye Y, Liu L, et al. Lenalidomide at therapeutic and supratherapeutic doses does not prolong QTc intervals in the thorough QTc study conducted in healthy men. Basic Clin Pharmacol Toxicol. 2013;113:179–86.
Acknowledgments
Funding
This study and the article processing fee in this journal were funded by the sponsor Samyang Biopharmaceutical Corp., Seongnam-si, Gyeonggi-do, Korea. Also this study was supported by a grant of the Korea Health Technology R&D Project through the Korea Health Industry Development Institute (KHIDI), funded by the Ministry of Health &Welfare, Republic of Korea (grant number: HI14C1063) and a research fund of Chungnam National University and supported by the Basic Science Research Program through the National Research Foundation of Korea (NRF) funded by the Ministry of Education (NRF-2017R1D1A1B04033515). All authors had full access to all of the data in this study and take complete responsibility for the integrity of the data and accuracy of the data analysis.
Authorship
All named authors meet the International Committee of Medical Journal Editors (ICMJE) criteria for authorship for this article, take responsibility for the integrity of the work as a whole, and have given their approval for this version to be published.
Disclosures
Sang Yeob Park is an employee of Samyang Biopharmaceuticals Corp. Hye Jung Lim is an employee of Samyang Biopharmaceuticals Corp. Sa-Won Lee is an employee of Samyang Biopharmaceuticals Corp. Min-Hyo Seo is an employee of Samyang Biopharmaceuticals Corp. SeungHwan Lee, Jun Gi Hwang, JaeWoo Kim, and Jang Hee Hong have nothing to disclose.
Compliance with Ethics Guidelines
This study was conducted in accordance with the ethical principles of the 1964 Declaration of Helsinki and its later amendments, as well as the rules of Korean Good Clinical Practice. All subjects provided written informed consent after receiving a detailed explanation about the study prior to any study related procedures.
Data Availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
Author information
Authors and Affiliations
Corresponding author
Additional information
Enhanced content
To view enhanced content for this article go to http://www.medengine.com/Redeem/5E89D3601649762E.
Rights and permissions
About this article
Cite this article
Lee, S., Hwang, J.G., Park, S.Y. et al. Single-Dose Comparative Pharmacokinetics of Two Formulations of Lenalidomide 25 mg in Healthy Subjects: A Randomized Crossover Study. Adv Ther 35, 210–217 (2018). https://doi.org/10.1007/s12325-018-0660-x
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12325-018-0660-x