Abstract
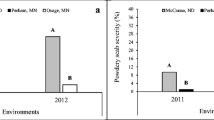
Potato mop-top virus (PMTV) is transmitted by the powdery scab pathogen (Spongospora subterranea f.sp. subterranea (Sss)) and no effective disease control methods are currently available for either pathogen. Eighty-one advanced breeding selections of potato (Solanum tuberosum L.) from different market classes and a broad genetic base were evaluated for sensitivity to PMTV-induced tuber necrosis in a field in North Dakota known to be infested with PMTV. Commercial cultivars, ranging from sensitive to tolerant in their reaction to PMTV-induced tuber necrosis incidence were included in each market class as internal controls. Results of tuber assessments revealed high variability in PMTV-induced tuber necrosis incidence and severity among selections. Based on PMTV-induced tuber necrosis incidence results over a two-year period, a total of 17 advanced selections were found to be tolerant, nine - moderately tolerant, eight - moderately sensitive, and six were found to be sensitive. The russet-skinned types had lower tuber necrosis incidence than the red-, white- and yellow-skinned types. Increases in the incidence of PMTV tuber necrosis during the storage period was influenced significantly by selection type and skin-color. Further studies are needed to investigate if tolerant selections are resistant to the virus to determine their suitability as parents in breeding programs to introduce PMTV resistance into commercial potato cultivars. In the short term, tolerant selections with other desirable agronomic characteristics could be released as commercial cultivars for growers to utilize as a means to limit the economic impact of PMTV-induced tuber necrosis.
Resumen
El virus de trapeador de la papa (potato mop top virus, PMTV) se transmite por el patógeno de la roña polvorienta (Spongospora subterránea f. sp. subterránea (Sss)) y no hay métodos efectivos disponibles actualmente de control de las enfermedades para cualquiera de estos patógenos. Se evaluaron 81 selecciones avanzadas de papa (Solanum tuberosum L.) de diferentes tipos de mercado y de una amplia base genética, para la sensibilidad de la necrosis de tubérculo inducida por PMTV, en un campo en Dakota del Norte que se sabe que esta infestado con PMTV. Las variedades comerciales, comprendidas desde sensibles a tolerantes en su reacción a la incidencia de la necrosis del tubérculo inducida por el PMTV, se incluyeron en cada clase de mercado como testigos internos. Los resultados en las evaluaciones de tubérculo revelaron alta variabilidad en la incidencia de la necrosis del tubérculo inducida por el PMTV y en la severidad entre selecciones. Con base a los resultados de la incidencia de la necrosis del tubérculo en un período de dos años, se encontraron un total de 17 selecciones avanzadas que fueron tolerantes, nueve moderadamente tolerantes, ocho moderadamente sensibles y seis susceptibles. Las de piel tipo russet tuvieron incidencia más baja de necrosis de tubérculo que las de tipo de piel roja, blanca y amarilla. El incremento en la incidencia de la necrosis de tubérculo por PMTV durante el período de almacenamiento fue influenciado significativamente por el tipo de selección y por el color de la piel. Se necesitan más estudios para determinar si las selecciones tolerantes son resistentes al virus, para decidir si pueden utilizarse como progenitores en programas de mejoramiento para introducir la resistencia a PMTV a variedades comerciales de papa. En el corto plazo, las selecciones tolerantes con otras características agronómicas deseables pudieran liberarse como variedades comerciales, para que las usen los productores como un medio para limitar el impacto económico de la necrosis del tubérculo inducida por PMTV.
Similar content being viewed by others
References
Arif, M., L. Torrance, and B. Reavy. 1995. Acquisition and transmission of Potato mop-top furovirus by a culture of Spongospora subterranea f. sp. subterranea derived from a single cystosorus. Annals of Applied Biology 126: 493–503.
Calvert, E.L. 1968. The reaction of potato varieties to Potato mop-top virus. Record of Agricultural Research (Ministry of Agriculture Northern Ireland) 17: 31–40.
Calvert, E.L., and B.D. Harrison. 1966. Potato mop-top, a soil-borne virus. Plant Pathology 15: 134–139.
Carnegie, S.F., T. Davey, and G.S. Saddler. 2010. Effect of temperature on the transmission of Potato mop-top virus from seed tuber and by its vector, Spongospora subterranea. Plant Pathology 59: 22–30.
Carnegie, S.F., T. Davey, and G.S. Saddler. 2012. Prevalence and distribution of Potato mop-top virus in Scotland. Plant Pathology 61: 623–631.
Carnegie, S.F., G.S. Saddler, and J.C. Peters. 2009. Cultivar susceptibility to Potato mop-top virus (PMTV) infection and symptom expression. Aspects of Applied Biology 94: 51–54.
Cooper, J.I., and B.D. Harrison. 1973. Distribution of Potato mop-top virus in Scotland in relation to soil and climate. Plant Pathology 22: 73–78.
Cooper, J.I., and A.T. Jones. 1983. Responses of plants to viruses: proposals for the use of terms. Phytopathology 73: 127–128.
Crosslin, J.M. 2011. First report of Potato mop-top virus on potatoes in Washington State. Plant Disease 95: 1483.
Davey, T. 2009. The importance of Potato mop-top virus (PMTV) in Scottish seed potatoes. PhD Doctoral Dissertation. Heriot Watt University, Scotland.
Davey, T., I. Browning, S.F. Carnegie, W.J. Mitchell, G.S. Saddler. 2008. Soil: the principal source of Potato mop top virus (PMTV) infection. Abstracts of the 13th European Association for Potato Research Virology Section Meeting, Aviemore, Scotland, UK, 17--22 June 2007.
Davey, T., S.F. Carnegie, G.S. Saddler, and W.J. Mitchell. 2014. The importance of the infected seed tuber and soil inoculum in transmitting Potato mop-top virus to potato plants. Plant Pathology 63: 88–97.
David, N., I. Mallik, J.M. Crosslin, and N.C. Gudmestad. 2010. First report of Potato mop-top virus on potatoes in North Dakota. Plant Disease 94: 1506.
Domfeh, O., F.G. Bittara, and N.C. Gudmestad. 2015. Sensitivity of potato cultivars to Potato mop-top virus-induced tuber necrosis. Plant Disease 99: 1–9.
Harrison, B.D. 1974. Potato mop-top virus. CMI/AAB Descriptions of Plant Viruses No. 138. Wellesbourne: Association of Applied Biologists.
Harrison, B.D., and R.A.C. Jones. 1970. Host range and properties of Potato mop-top virus. Annals of Applied Biology 65: 393–402.
Harrison, B.D., and R.A.C. Jones. 1971. Factors affecting the development of spraing in potato tubers infected with Potato mop-top virus. Annals of Applied Biology 68: 281–289.
Harrison, J.G., R.J. Searle, and N.A. Williams. 1997. Powdery scab disease of potato – a review. Plant Pathology 46: 1–25.
Hinostroza, A.M., and E.R. French. 1972. Potato mop-top virus in cork-diseased Peruvian potatoes. American Potato Journal 49: 234–239.
Jeffries, C.J. 1998. Potato. In FAO/IPGRI Technical guidelines for the safe movement of Germplasm, vol. 19, 77. Rome: Food and Agricultural Organization of the United Nations/International Plant Genetic Resources Institute.
Jones, R.A.C. 1988. Epidemiology and control of Potato mop-top virus. In Developments in Applied Biology II. Viruses with Fungal Vectors, ed. J.I. Cooper and M.J.C. Asher, 255–270. Wellesbourne: Association of applied Biologists.
Jones, R.A.C., and B.D. Harrison. 1969. The behavior of Potato mop-top virus in soil, and evidence for its transmission by Spongospora subterranea (Wallr.) Lagerh. Annals of Applied Biology 63: 1–17.
Jones, R.A.C., and B.D. Harrison. 1972. Ecological studies on Potato mop‐top virus in Scotland. Annals of Applied Biology 71: 47–57.
Kurppa, A. 1989. Reaction of potato cultivars to primary and secondary infection by Potato mop-top furovirus and strategies for virus detection. EPPO Bulletin 19: 593–598.
Lambert, D.H., L. Levy, V.A. Mavrodieva, S.B. Johnson, M.J. Babcock, and M.E. Vayda. 2003. First report of Potato mop-top virus on potato from the United States. Plant Disease 87: 872.
Latvala-Kilby, S., J.M. Aura, N. Pupola, A. Hannukkala, and J.P.T. Valkonen. 2009. Detection of Potato mop-top virus in potato tubers and sprouts: combinations of RNA2 and RNA3 variants and incidence of symptomless infections. Phytopathology 99: 519–531.
Mallik, I., and N.C. Gudmestad. 2015. First Report of Potato mop-top virus causing tuber necrosis in Colorado and New Mexico. Plant Disease 99: 164.
Merz, U. 2008. Powdery scab of potato - Occurrence, life cycle and epidemiology. American Journal of Potato Research 85: 241–246.
Miller, J. 2001. Powdery scab workshop - summary notes. Alamosa, CO January 11, 2001. http://www.uidaho.edu/ag/plantdisease/scabnote.htm. Accessed 9 June 2005.
Millikin, G.A., and D.E. Johnson. 1992. One-way treatment structure in a completely randomized design with heterogeneous errors. In Analysis of Messy Data, Vol. 1. Designed Experiments, 16–28. London: Chapman and Hall.
Molgaard, J.P., and S.L. Nielsen. 1996. Influence of post-harvest temperature treatments, storage period and harvest date on development of spraing caused by Tobacco rattle virus and Potato mop-top virus. Potato Research 39: 571–579.
Nakayama, T., T. Maoka, T. Hataya, M. Shimizu, H. Fuwa, S. Tsuda, and M. Mori. 2010. Diagnosis of Potato mop-top virus in soil using bait plant bioassay and PCR-microplate hybridization. American Journal of Potato Research 87: 218–225.
Nielsen, S.L., and B. Engsbro. 1992. Susceptibility of potato cultivars to spraing caused by primary infection of Tobacco rattle virus and Potato mop-top virus. Danish Journal of Plant and Soil Science 96: 507–516.
Nielsen, S.L., and J.P. Molgaard. 1997. Incidence, appearance and development of Potato mop-top furovirus-induced spraing in potato cultivars and the influence on yield, distribution in Denmark and detection of the virus in tubers by ELISA. Potato Research 40: 101–110.
Nitzan, N., T.F. Cummings, D.A. Johnson, J.S. Miller, D.L. Batchelor, C. Olsen, R.A. Quick, and C.R. Brown. 2008. Resistance to root galling caused by the powdery scab pathogen Spongospora subterranea in potato. Plant Disease 92: 1643–1649.
Perla, V., S.S. Jayanty, D.G. Holm, and R.D. Davidson. 2014. Relationship between tuber storage proteins and tuber powdery scab resistance in potato. American Journal of Potato Research 91: 233–245.
Robinson, D.J. 1992. Detection of Tobacco rattle virus by reverse transcription and polymerase chain reaction. Journal of Virological Methods 40: 57–66.
Rudorf, W. 1958. The significance of wild species for potato breeding. European Potato Journal 1: 10–20.
Ryden, K., L. Lovegren, and M. Sandgren. 1989. Investigation on Potato mop-top furovirus in Sweden. EPPO Bulletin 19: 579–583.
Salazar, L.F., and R.A.C. Jones. 1975. Some studies on the distribution of Potato mop-top virus in Peru. American Potato Journal 52: 143–150.
Sandgren, M. 1995. Potato mop-top virus (PMTV): Distribution in Sweden, development of symptoms during storage and cultivar trials in field and glasshouse. Potato Research 38: 387–397.
Sandgren, M., R.L. Plaisted, K.N. Watanabe, S. Olsson, and J.P.T. Valkonen. 2002. Evaluation of some North and South American potato breeding lines for resistance to Potato mop-top virus in Sweden. American Journal of Potato Research 79: 205–210.
Scott, K.P., S. Kashiwazaki, B. Reavy, and B.D. Harrison. 1994. The nucleotide sequence of Potato mop-top virus RNA 2: a novel type of genome organization for a furovirus. Journal of General Virology 75: 3561–3568.
Sokmen, M.A., H. Barker, and L. Torrance. 1998. Factors affecting the detection of Potato mop-top virus in potato tubers and improvement of test procedures for more reliable assays. Annals of Applied Biology 133: 55–63.
Tenorio, J., Y. Franco, C. Chuquillanqui, R.A. Owens, and L.F. Salazar. 2006. Reaction of potato varieties to Potato mop-top virus infection in the Andes. American Journal of Potato Research 83: 423–431.
Torrance, L., and M.A. Mayo. 1997. Proposed re-classification of furoviruses. Archives of Virology 142: 435–439.
Torrance, L., G.H. Cowan, M.A. Sokmen, and B. Reavy. 1999. A naturally occurring deleted form of RNA 2 of Potato mop-top virus. Journal of General Virology 80: 2211–2215.
Wale, S.J. 2000. Summary of the session on national potato production and the powdery scab situation. In Merz U and Lees AK eds, Proceedings of the First European Powdery Scab Workshop, Aberdeen, Scotland, 20–22 July, 3–9.
Wastie, R.L. 1991. Resistance to powdery scab of seedling progenies of Solanum tuberosum. Potato Research 34: 249–252.
Wastie, R.L. 1994. Inheritance of resistance to fungal diseases. In Potato genetics, ed. J.E. Bradshaw and G.R. Mackay, 421. Wallingford: CAB International.
Whitworth, J.L., and J.M. Crosslin. 2013. Detection of Potato mop-top virus (Furovirus) on potato in southeast Idaho. Plant Disease 97: 149.
Xu, H., T.-L. DeHaan, and S.H. De Boer. 2004. Detection and confirmation of Potato mop-top virus in potatoes produced in the United States and Canada. Plant Disease 88: 363–367.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Domfeh, O., Thompson, A.L. & Gudmestad, N.C. Sensitivity to Tuber Necrosis Caused by Potato Mop-Top Virus in Advanced Potato (Solanum tuberosum L.) Breeding Selections. Am. J. Potato Res. 92, 636–647 (2015). https://doi.org/10.1007/s12230-015-9477-1
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12230-015-9477-1