Abstract
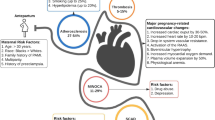
Cardiovascular disease is the leading cause of death among pregnant women and causes significant maternal and fetal morbidity. Pregnant women may have known cardiac disease, such as prosthetic heart valves, while others develop new complications, such as myocardial ischemia or cardiomyopathy. Tools for predicting the risk of adverse cardiac events during pregnancy exist. Data from international registries will improve our ability to accurately assess risks and manage and counsel patients. This review will also address the following: (1) Myocardial infarctions that occur during or after pregnancy are often related to coronary dissection and require special attention. (2) Peripartum cardiomyopathy causes heart failure, which can be severe and fatal. Women who recover need careful counseling about the risks of a subsequent pregnancy. (3) Anti-coagulation of mechanical heart valves during pregnancy involves challenging decisions about the safety of warfarin for the fetus, but low-molecular-weight heparin requires meticulous attention to anti-Xa levels.
Similar content being viewed by others
References
Papers of particular interest, published recently, have been highlighted as: • Of importance •• Of major importance
Cantwell R, Clutton-Brock T, Cooper G, Dawson A, Drife J, Garrod D, et al. Saving mothers' lives: Reviewing maternal deaths to make motherhood safer: 2006–2008. The eighth report of the confidential enquiries into maternal deaths in the united kingdom. BJOG :Int J Obstet Gynaecol. 2011;118 Suppl 1:1–203.
Creanga AA BC, Syverson C, Seed K, Bruce C, Callaghan WM. Pregnancy-related mortality in the United States, 2006–2010. Obstet Gynecol. 2015;125:5–12. This study reviews the major causes of maternal mortality in the United States.
Balci A, Sollie-Szarynska KM, van der Bijl AG, Ruys TP, Mulder BJ, Roos-Hesselink JW, et al. Prospective validation and assessment of cardiovascular and offspring risk models for pregnant women with congenital heart disease. Heart. 2014;100:1373–81.
Siu SC, Sermer M, Colman JM, Alvarez AN, Mercier LA, Morton BC, et al. Cardiac Disease in Pregnancy I. Prospective multicenter study of pregnancy outcomes in women with heart disease. Circulation. 2001;104:515–21.
Khairy P, Ouyang DW, Fernandes SM, Lee-Parritz A, Economy KE, Landzberg MJ. Pregnancy outcomes in women with congenital heart disease. Circulation. 2006;113:517–24.
Roos-Hesselink JW, Ruys TP, Stein JI, Thilen U, Webb GD, Niwa K, et al. Outcome of pregnancy in patients with structural or ischaemic heart disease: results of a registry of the european society of cardiology. Eur Heart J. 2013;34:657–65.
Siu SC, Colman JM, Sorensen S, Smallhorn JF, Farine D, Amankwah KS, et al. Adverse neonatal and cardiac outcomes are more common in pregnant women with cardiac disease. Circulation. 2002;105:2179–84.
Drenthen W, Boersma E, Balci A, Moons P, Roos-Hesselink JW, Mulder BJ, et al. Predictors of pregnancy complications in women with congenital heart disease. Eur Heart J. 2010;31:2124–32.
European Society of G, Association for European Paediatric C, German Society for Gender M, Regitz-Zagrosek V, Blomstrom Lundqvist C, Borghi C, et al. Esc guidelines on the management of cardiovascular diseases during pregnancy: the task force on the management of cardiovascular diseases during pregnancy of the european society of cardiology (esc). European heart journal. 2011;32:3147–97. The European guidelines on the management of cardiovascular disease during pregnancy. The level of risk associated with pregnancy is determined by the underlying heart disease and modified WHO classification.
Subbaiah M, Sharma V, Kumar S, Rajeshwari S, Kothari SS, Roy KK, et al. Heart disease in pregnancy: cardiac and obstetric outcomes. Arch Gynecol Obstet. 2013;288:23–7.
Ruys TP, Roos-Hesselink JW, Hall R, Subirana-Domenech MT, Grando-Ting J, Estensen M, et al. Heart failure in pregnant women with cardiac disease: data from the ROPAC. Heart. 2014;100:231–8. This data from ROPAC shows that women with heart failure during pregnancy have higher rates of maternal and fetal mortality than women without heart failure.
Pieper PG, Balci A, Aarnoudse JG, Kampman MA, Sollie KM, Groen H, et al. Uteroplacental blood flow, cardiac function, and pregnancy outcome in women with congenital heart disease. Circulation. 2013;128:2478–87.
Madazli R, Somunkiran A, Calay Z, Ilvan S, Aksu MF. Histomorphology of the placenta and the placental bed of growth restricted foetuses and correlation with the Doppler velocimetries of the uterine and umbilical arteries. Placenta. 2003;24:510–6.
Tanous D, Siu SC, Mason J, Greutmann M, Wald RM, Parker JD, et al. B-type natriuretic peptide in pregnant women with heart disease. J Am Coll Cardiol. 2010;56:1247–53.
Smith R, Silversides C, Downey K, Newton G, Macarthur A. Assessing the incidence of peripartum subclinical myocardial ischemia using the troponin t assay: An observational pilot study. International journal of obstetric anesthesia. 2014.
Elkayam U, Jalnapurkar S, Barakkat MN, Khatri N, Kealey AJ, Mehra A, et al. Pregnancy-associated acute myocardial infarction: A review of contemporary experience in 150 cases between 2006 and 2011. Circulation. 2014;129:1695–702. This study is a recent review of 150 cases of pregnancy-associated acute myocardial infarction, which showed a high proportion of coronary dissection.
Zimmerman FH, Cameron A, Fisher LD, Ng G. Myocardial infarction in young adults: angiographic characterization, risk factors and prognosis (coronary artery surgery study registry). J Am Coll Cardiol. 1995;26:654–61.
Tweet MS, Hayes SN, Pitta SR, Simari RD, Lerman A, Lennon RJ, et al. Clinical features, management, and prognosis of spontaneous coronary artery dissection. Circulation. 2012;126:579–88.
Wingrove CS, Garr E, Godsland IF, Stevenson JC. 17beta-oestradiol enhances release of matrix metalloproteinase-2 from human vascular smooth muscle cells. Biochim Biophys Acta. 1998;1406:169–74.
Manalo-Estrella P, Barker AE. Histopathologic findings in human aortic media associated with pregnancy. Arch Pathol. 1967;83:336–41.
Robinowitz M, Virmani R, McAllister HAJ. Spontaneous coronary artery dissection and eosinophilic inflammation: a cause and effect relationship? Am J Med. 1982;72:923–8.
Roth A, Elkayam U. Acute myocardial infarction associated with pregnancy. Ann Intern Med. 1996;125:751–62.
Roth A, Elkayam U. Acute myocardial infarction associated with pregnancy. J Am Coll Cardiol. 2008;52:171–80.
Goland S, Elkayam U. Anticoagulation in pregnancy. Cardiol Clin. 2012;30:395–405.
Choi JW, Pai SH. Tissue plasminogen activator levels change with plasma fibrinogen concentrations during pregnancy. Ann Hematol. 2002;81:611–5.
Ersboll AS, Hedegaard M, Sondergaard L, Ersboll M, Johansen M. Treatment with oral beta-blockers during pregnancy complicated by maternal heart disease increases the risk of fetal growth restriction. BJOG : Int J Obstet Gynaecol. 2014;121:618–26.
Alfonso F. Spontaneous coronary artery dissection: new insights from the tip of the iceberg? Circulation. 2012;126:667–70.
Alfonso F, Paulo M, Lennie V, Dutary J, Bernardo E, Jimenez-Quevedo P, et al. Spontaneous coronary artery dissection: long-term follow-up of a large series of patients prospectively managed with a “conservative” therapeutic strategy. J Am Coll Cardiol Intv. 2012;5:1062–70.
Colletti PM, Lee KH, Elkayam U. Cardiovascular imaging of the pregnant patient. AJR. Am J Roentgenol. 2013;200:515–21.
Arnoni RT, Arnoni AS, Bonini RC, de Almeida AF, Neto CA, Dinkhuysen JJ, et al. Risk factors associated with cardiac surgery during pregnancy. Ann Thorac Surg. 2003;76:1605–8.
Immer FF, Bansi AG, Immer-Bansi AS, McDougall J, Zehr KJ, Schaff HV, et al. Aortic dissection in pregnancy: analysis of risk factors and outcome. Ann Thorac Surg. 2003;76:309–14.
Nishimura RA, Otto CM, Bonow RO, Carabello BA, Erwin 3rd JP, Guyton RA, et al. 2014 aha/acc guideline for the management of patients with valvular heart disease: a report of the american college of cardiology/american heart association task force on practice guidelines. Circulation. 2014;129:e521–643. Recent ACC/AHA guidelines for the management of patients with valvular heart disease.
Parry AJ, Westaby S. Cardiopulmonary bypass during pregnancy. Ann Thorac Surg. 1996;61:1865–9.
Sliwa K, Hilfiker-Kleiner D, Petrie MC, Mebazaa A, Pieske B, Buchmann E, et al. Current state of knowledge on aetiology, diagnosis, management, and therapy of peripartum cardiomyopathy: a position statement from the heart failure association of the european society of cardiology working group on peripartum cardiomyopathy. Eur J Heart Fail. 2010;12:767–78. The European Society of Cardiolgoy Working Group on Peripartum Cardiomyopathy provides a review of the literature and expert opinion.
Hibbard JU, Lindheimer M, Lang RM. A modified definition for peripartum cardiomyopathy and prognosis based on echocardiography. Obstet Gynecol. 1999;94:311–6.
Elkayam U. Clinical characteristics of peripartum cardiomyopathy in the United States: diagnosis, prognosis, and management. J Am Coll Cardiol. 2011;58:659–70.
Kolte D, Khera S, Aronow WS, Palaniswamy C, Mujib M, Ahn C, et al. Temporal trends in incidence and outcomes of peripartum cardiomyopathy in the United States: a nationwide population-based study. J Am Heart Assoc. 2014;3, e001056.
Fett JD. Peripartum cardiomyopathy: a puzzle closer to solution. World J Cardiol. 2014;6:87–99.
Hilfiker-Kleiner D, Kaminski K, Podewski E, Bonda T, Schaefer A, Sliwa K, et al. A cathepsin D-cleaved 16 kDa form of prolactin mediates postpartum cardiomyopathy. Cell. 2007;128:589–600.
Patten IS, Rana S, Shahul S, Rowe GC, Jang C, Liu L, et al. Cardiac angiogenic imbalance leads to peripartum cardiomyopathy. Nature. 2012;485:333–8.
van Spaendonck-Zwarts KY, Posafalvi A, van den Berg MP, Hilfiker-Kleiner D, Bollen IA, Sliwa K, et al. Titin gene mutations are common in families with both peripartum cardiomyopathy and dilated cardiomyopathy. Eur Heart J. 2014;35:2165–73.
Writing Committee M, Yancy CW, Jessup M, Bozkurt B, Butler J, Casey Jr DE, et al. American College of Cardiology Foundation/American Heart Association Task Force on Practice G. 2013 accf/aha guideline for the management of heart failure: a report of the american college of cardiology foundation/american heart association task force on practice guidelines. Circulation. 2013;128:e240–327.
Sheppard R, Rajagopalan N, Safirstein J, Briller J. An update on treatments and outcomes in peripartum cardiomyopathy. Futur Cardiol. 2014;10:435–47.
Pillarisetti J, Kondur A, Alani A, Reddy M, Reddy M, Vacek J, et al. Peripartum cardiomyopathy: predictors of recovery and current state of implantable cardioverter-defibrillator use. J Am Coll Cardiol. 2014;63:2831–9.
Duncker D, Haghikia A, Konig T, Hohmann S, Gutleben KJ, Westenfeld R, et al. Risk for ventricular fibrillation in peripartum cardiomyopathy with severely reduced left ventricular function-value of the wearable cardioverter/defibrillator. Eur J Heart Fail. 2014;16:1331–6.
Elkayam U. Risk of subsequent pregnancy in women with a history of peripartum cardiomyopathy. J Am Coll Cardiol. 2014;64:1629–36. This paper summarizes the known literature regarding the risks associated with subsequent pregnancy for women who have or have not recovered myocardial function.
Elkayam U, Tummala PP, Rao K, Akhter MW, Karaalp IS, Wani OR, et al. Maternal and fetal outcomes of subsequent pregnancies in women with peripartum cardiomyopathy. N Engl J Med. 2001;344:1567–71.
Fett JD, Fristoe KL, Welsh SN. Risk of heart failure relapse in subsequent pregnancy among peripartum cardiomyopathy mothers. Int J Gynaecol Obstet: Off Organ Int Fed Gynaecol Obstet. 2010;109:34–6.
Sliwa K, Hilfiker-Kleiner D, Mebazaa A, Petrie MC, Maggioni AP, Regitz-Zagrosek V, et al. EURObservational research programme: a worldwide registry on peripartum cardiomyopathy (PPCM) in conjunction with the Heart Failure Association of the European Society of Cardiology Working Group on PPCM. Eur J Heart Fail. 2014;16:583–91.
Bremme KA. Haemostatic changes in pregnancy. Best practice & research. Clin Haematol. 2003;16:153–68.
McLintock C. Anticoagulant therapy in pregnant women with mechanical prosthetic heart valves: no easy option. Thromb Res. 2011;127 Suppl 3:S56–60.
Eikelboom JW, Connolly SJ, Brueckmann M, Granger CB, Kappetein AP, Mack MJ, et al. Dabigatran versus warfarin in patients with mechanical heart valves. N Engl J Med. 2013;369:1206–14.
Hall JG, Pauli RM, Wilson KM. Maternal and fetal sequelae of anticoagulation during pregnancy. Am J Med. 1980;68:122–40.
McLintock C. Thromboembolism in pregnancy: challenges and controversies in the prevention of pregnancy-associated venous thromboembolism and management of anticoagulation in women with mechanical prosthetic heart valves. Best practice & research. Clin Obstet Gynaecol. 2014;28:519–36.
Raghav S, Reutens D. Neurological sequelae of intrauterine warfarin exposure. J Clin Neurosci Off J Neurosurg Soc Australasia. 2007;14:99–103.
Vitale N, De Feo M, De Santo LS, Pollice A, Tedesco N, Cotrufo M. Dose-dependent fetal complications of warfarin in pregnant women with mechanical heart valves. J Am Coll Cardiol. 1999;33:1637–41.
Soma-Pillay P, Nene Z, Mathivha TM, Macdonald AP. The effect of warfarin dosage on maternal and fetal outcomes in pregnant women with prosthetic heart valves. Obstet Med. 2011;4:24–7.
De Santo LS, Romano G, Della Corte A, D’Oria V, Nappi G, Giordano S, et al. Mechanical aortic valve replacement in young women planning on pregnancy: maternal and fetal outcomes under low oral anticoagulation, a pilot observational study on a comprehensive pre-operative counseling protocol. J Am Coll Cardiol. 2012;59:1110–5.
Elkayam U, Goland S. The search for a safe and effective anticoagulation regimen in pregnant women with mechanical prosthetic heart valves. J Am Coll Cardiol. 2012;59:1116–8.
Sadler L, McCowan L, White H, Stewart A, Bracken M, North R. Pregnancy outcomes and cardiac complications in women with mechanical, bioprosthetic and homograft valves. BJOG : Int J Obstet Gynaecol. 2000;107:245–53.
Goland S, Schwartzenberg S, Fan J, Kozak N, Khatri N, Elkayam U. Monitoring of anti-xa in pregnant patients with mechanical prosthetic valves receiving low-molecular-weight heparin: peak or trough levels? J Cardiovasc Pharmacol Ther. 2014;19:451–6. This paper draws attention to the need to follow trough levels when managing patients with low-molecular-weight heparin.
Bates SM, Greer IA, Middeldorp S, Veenstra DL, Prabulos AM, Vandvik PO, et al. Vte, thrombophilia, antithrombotic therapy, and pregnancy: antithrombotic therapy and prevention of thrombosis, 9th ed: American college of chest physicians evidence-based clinical practice guidelines. Chest. 2012;141:e691S–736S.
Chowdary P, Adamidou D, Riddell A, Aghighi S, Griffioen A, Priest P, Moghadam L, Kelaher N, Huq FY, Kadir RA, Tuddenham EG, Gatt A. Thrombin generation assay identifies individual variability in responses to low molecular weight heparin in pregnancy: Implications for anticoagulant monitoring. British journal of haematology. 2014.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
Melinda Davis has no relevant disclosures.
Human and Animal Rights and Informed Consent
This article does not contain any studies with human or animal subjects performed by the author.
Additional information
This article is part of the Topical Collection on Women and Heart Disease
Rights and permissions
About this article
Cite this article
Davis, M.B. Pregnancy and Heart Disease Updates: Current Knowledge and Future Directions. Curr Cardiovasc Risk Rep 9, 50 (2015). https://doi.org/10.1007/s12170-015-0478-x
Published:
DOI: https://doi.org/10.1007/s12170-015-0478-x