Abstract
Purpose/objective(s)
To improve the curative resection rates and prognoses, a variety of neoadjuvant (NA) strategies have been explored in PDAC. In our institution, non-metastatic PDACs have been treated with a NA intent with induction multiagent chemotherapy and SBRT. The primary endpoint was to increase R0 resection rate. The secondary endpoints were the analysis of the clinical tolerance, the pathological response, the local control (LC) and the OS.
Materials/methods
All consecutive patients with non-metastatic PDAC underwent SBRT as part of the NA strategy were included. A total dose of 40–62 Gy were delivered in 5–10 fractions. Surgery was performed after SBRT and restaging.
Results
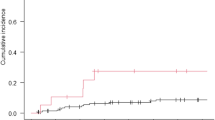
Since February 2014 to December 2018, 45 patients were enrolled. Thirty-two patients underwent surgery (71.1%), 10 out of 15 were initially unresectable disease patients (66.75%). R0 resection rate was 93% (30 patients) and pN0 status was achieved in 20 patients (60.6%). Tumour regression grade (TRG): 12 patients with complete response or marked response (TRG 0–1: 37.5%), 16 patients with moderate response (TRG 2: 50%) and four patients with poor response (TRG 3: 12.5%). The median follow-up was 16.2 m (range 6.6–59.6 m) since diagnosis. The LC rate achieved was very high (95.5%). Actuarial 12 and 24 m OS was 67.4% and 35.9% respectively. No grade 3 or higher toxicity related to SBRT was observed.
Conclusion
The results are encouraging, suggesting that SBRT has a significant role in the management of these patients and further studies will be necessary to prove these findings.


Similar content being viewed by others
References
International agency for research on Cancer. GLOBOCAN 2012. WHO Am Reg 2012. https://globocan.iarc.fr/old/bar_pop.asp?selection=206826&title=United+Kingdom&sex=0&statistic=0&window=1&grid=1&color1=5&color1e=&color2=4&color2e=&orientation=1&submit=Executr . Accessed 10 Mar 2017).
EUROCARE—Survival of cancer patients in Europe n.d. 2010. https://www.eurocare.it/. Accessed 24 July 2018).
Gillen S, Schuster T, MeyerzumBüschenfelde C, Friess H, Kleeff J. Preoperative/neoadjuvant therapy in pancreatic cancer: a systematic review and meta-analysis of response and resection percentages. PLoS Med. 2010;7:e1000267. https://doi.org/10.1371/journal.pmed.1000267.
Tinchon C, Hubmann E, Pichler A, Keil F, Pichler M, Rabl H, et al. Safety and efficacy of neoadjuvant FOLFIRINOX treatment in a series of patients with borderline resectable pancreatic ductal adenocarcinoma. Acta Oncol (Madr). 2013;52:1231–3. https://doi.org/10.3109/0284186X.2013.771821.
Peterson SL, Husnain M, Pollack T, Pimentel A, LoaizaBonilla A, Westendorf-Overley C, et al. Neoadjuvant Nab -paclitaxel and gemcitabine in borderline resectable or locally advanced unresectable pancreatic adenocarcinoma in patients who are ineligible for FOLFIRINOX. Anticancer Res. 2018;38:4035–9. https://doi.org/10.21873/anticanres.12692.
Pisters PWT, Abbruzzese JL, Janjan NA, Cleary KR, Charnsangavej C, Goswitz MS, et al. Rapid-fractionation preoperative chemoradiation, pancreaticoduodenectomy, and intraoperative radiation therapy for resectable pancreatic adenocarcinoma. J Clin Oncol. 1998;16:3843–50. https://doi.org/10.1200/JCO.1998.16.12.3843.
Evans DB, Varadhachary GR, Crane CH, Sun CC, Lee JE, Pisters PWT, et al. Preoperative gemcitabine-based chemoradiation for patients with resectable adenocarcinoma of the pancreatic head. J Clin Oncol. 2008;26:3496–502. https://doi.org/10.1200/JCO.2007.15.8634.
Esnaola NF, Chaudhary UB, O’Brien P, Garrett-Mayer E, Camp ER, Thomas MB, et al. Phase 2 trial of induction gemcitabine, oxaliplatin, and cetuximab followed by selective capecitabine-based chemoradiation in patients with borderline resectable or unresectable locally advanced pancreatic cancer. Int J Radiat Oncol Biol Phys. 2014;88:837–44. https://doi.org/10.1016/j.ijrobp.2013.12.030.
Katz MHG, Shi Q, Ahmad SA, Herman JM, Marsh RDW, Collisson E, et al. Preoperative modified FOLFIRINOX treatment followed by capecitabine-based chemoradiation for borderline resectable pancreatic cancer alliance for clinical trials in oncology trial A021101. JAMA Surg. 2016. https://doi.org/10.1001/jamasurg.2016.1137.
Murphy JE, Wo JY, Ryan DP, Jiang W, Yeap BY, Drapek LC, et al. Total neoadjuvant therapy with FOLFIRINOX followed by individualized chemoradiotherapy for borderline resectable pancreatic adenocarcinoma: a phase 2 clinical trial. JAMA Oncol. 2018;4:963–9. https://doi.org/10.1001/jamaoncol.2018.0329.
Huguet F, Hajj C, Winston CB, Shi W, Zhang Z, Wu AJ, et al. Chemotherapy and intensity-modulated radiation therapy for locally advanced pancreatic cancer achieves a high rate of R0 resection. Acta Oncol (Madr). 2017;56:384–90. https://doi.org/10.1080/0284186X.2016.1245862.
Koong AC, Le QT, Ho A, Fong B, Fisher G, Cho C, et al. Phase I study of stereotactic radiosurgery in patients with locally advanced pancreatic cancer. Int J Radiat Oncol Biol Phys. 2004;58:1017–21. https://doi.org/10.1016/j.ijrobp.2003.11.004.
Mellon EA, Hoffe SE, Springett GM, Frakes JM, Strom TJ, Hodul PJ, et al. Long-term outcomes of induction chemotherapy and neoadjuvant stereotactic body radiotherapy for borderline resectable and locally advanced pancreatic adenocarcinoma. Acta Oncol (Madr). 2015;54:979–85. https://doi.org/10.3109/0284186X.2015.1004367.
Mahadevan A, Miksad R, Goldstein M, Sullivan R, Bullock A, Buchbinder E, et al. Induction gemcitabine and stereotactic body radiotherapy for locally advanced nonmetastatic pancreas cancer. Int J Radiat Oncol Biol Phys. 2011;81:e615–e622622. https://doi.org/10.1016/j.ijrobp.2011.04.045.
Katz MHG, Ou F-S, Herman JM, Ahmad SA, Wolpin B, Marsh R, et al. Alliance for clinical trials in oncology (ALLIANCE) trial A021501: preoperative extended chemotherapy vs. chemotherapy plus hypofractionated radiation therapy for borderline resectable adenocarcinoma of the head of the pancreas. BMC Cancer. 2017;17:505. https://doi.org/10.1186/s12885-017-3441-z.
Petrelli F, Comito T, Ghidini A, Torri V, Scorsetti M, Barni S. Stereotactic body radiation therapy for locally advanced pancreatic cancer: a systematic review and pooled analysis of 19 trials. Int J Radiat Oncol Biol Phys. 2017;97:313–22. https://doi.org/10.1016/j.ijrobp.2016.10.030.
Stessin AM, Meyer JE, Sherr DL. Neoadjuvant radiation is associated with improved survival in patients with resectable pancreatic cancer: an analysis of data from the surveillance, epidemiology, and end results (SEER) registry. Int J Radiat Oncol Biol Phys. 2008;72:1128–33. https://doi.org/10.1016/j.ijrobp.2008.02.065.
Van Tienhoven G, Versteijne E, Suker M, Groothuis KBC, Busch OR, Bonsing BA, et al. Preoperative chemoradiotherapy versus immediate surgery for resectable and borderline resectable pancreatic cancer (PREOPANC-1): a randomized, controlled, multicenter phase III trial. J Clin Oncol. 2018;36:LBA4002. https://doi.org/10.1200/jco.2018.36.18_suppl.lba4002.
Mokdad AA, Minter RM, Zhu H, Augustine MM, Porembka MR, Wang SC, et al. Neoadjuvant therapy followed by resection versus upfront resection for resectable pancreatic cancer: a propensity score matched analysis. J Clin Oncol. 2017;35:515–22. https://doi.org/10.1200/JCO.2016.68.5081 (American Society of Clinical Oncology).
Tempero MA, Malafa MP, Behrman SW, Benson AB, Casper ES, Chiorean EG, et al. Pancreatic adenocarcinoma, version 2.2014: featured updates to the NCCN guidelines. J Natl Compr Canc Netw. 2014;12:1083–93.
Al-Hawary M, Bain A, Behrman SW, Benson III AB, Binder E, Cardin DB et al. Cassadie Moravek ¥ Pancreatic Cancer Action Network NCCN Guidelines Version 2.2018 Pancreatic Adenocarcinoma. 2018.
Tabernero S, Prados S, del Carmen Rubio M, de la Morena F, López M, Sánchez E. REED—Revista Española de Enfermedades Digestivas. Rev Esp Enferm Dig. 2019. https://doi.org/10.17235/reed.2019.5873/2018.
Ryan R, Gibbons D, Hyland JMP, Treanor D, White A, Mulcahy HE, et al. Pathological response following long-course neoadjuvant chemoradiotherapy for locally advanced rectal cancer. Histopathology. 2005;47:141–6. https://doi.org/10.1111/j.1365-2559.2005.02176.x.
Cancer Institute N. Common Terminology Criteria for Adverse Events (CTCAE) Version 4.0. 2009.
Chuong MD, Springett GM, Freilich JM, Park CK, Weber JM, Mellon EA, et al. Stereotactic body radiation therapy for locally advanced and borderline resectable pancreatic cancer is effective and well tolerated. Int J Radiat Oncol Biol Phys. 2013;86:516–22. https://doi.org/10.1016/j.ijrobp.2013.02.022.
Zhong J, Patel K, Switchenko J, Cassidy RJ, Hall WA, Gillespie T, et al. Outcomes for patients with locally advanced pancreatic adenocarcinoma treated with stereotactic body radiation therapy versus conventionally fractionated radiation. Cancer. 2017;123:3486–93. https://doi.org/10.1002/cncr.30706.
Chuong MD, Frakes JM, Figura N, Hoffe SE, Shridhar R, Mellon EA, et al. Histopathologic tumor response after induction chemotherapy and stereotactic body radiation therapy for borderline resectable pancreatic cancer. J Gastrointest Oncol. 2016;7:221–7. https://doi.org/10.3978/j.issn.2078-6891.2015.075.
Chatterjee D, Katz MH, Rashid A, Varadhachary GR, Wolff RA, Wang H, et al. Histologic grading of the extent of residual carcinoma following neoadjuvant chemoradiation in pancreatic ductal adenocarcinoma. Cancer. 2012;118:3182–90. https://doi.org/10.1002/cncr.26651.
Acknowledgements
We would like to recognize the support of our patients and colleagues.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that there is no conflict of interest regarding the publication of this article.
Ethical approval
Institutional Review Board approval was obtained. The number of the study is 18.08.1285-GHM.
Informed consent
Written informed consent was waived by the Institutional Review Board.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Chen-Zhao, X., Hernando, O., López, M. et al. A prospective observational study of the clinical and pathological impact of stereotactic body radiotherapy (SBRT) as a neoadjuvant strategy of chemoradiation in pancreatic cancer. Clin Transl Oncol 22, 1499–1505 (2020). https://doi.org/10.1007/s12094-020-02287-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12094-020-02287-w