Abstract
Background
Hepatitis A virus (HAV) is the commonest cause of pediatric acute liver failure (PALF) in developing countries. Our objective was to develop and validate a HAV-etiology specific prognostic model in PALF.
Methods
All children with HAV induced PALF (IgM HAV reactive) were included. Outcome was defined at day 28. Only those with death or native liver survival were included. The model (Peds-HAV) was derived using the independent predictors of outcome and validated in a prospective independent cohort.
Results
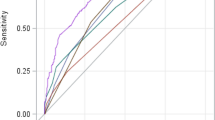
Hepatitis A accounted for 131 (45.9%) of total 285 PALF. After excluding 11 children who underwent liver transplant, 120 children (74 survivors and 46 death) were included. The first 75 patients formed the derivation cohort and the next 45 patients formed the prospective validation cohort. In the derivation cohort, INR: OR 2.208, (95% CI 1.321–3.690), p = 0.003, grade of hepatic encephalopathy (HE): OR 3.078, (95% CI 1.017–9.312), p = 0.047 and jaundice-to-HE interval: OR 1.171, (95% CI 1.044–1.314), p = 0.007 were independent predictors of death. The final model comprised three criteria: (1) presence of grade 3–4 HE, (2) INR greater than 3.1, and (3) jaundice to HE interval more than 10 days. Presence of 2 or more of these criteria predicted death with 90% sensitivity, 81.4% specificity and 84.9% accuracy. Peds-HAV model was superior to existing prognostic models. In the validation cohort, Peds-HAV model predicted death with 83.3% sensitivity and 92.6% specificity.
Conclusion
Peds-HAV model is a simple, bedside, dynamic, etiology (HAV) specific prognostic model based on 3 objective parameters with optimum sensitivity and specificity, hence should be used as liver transplant listing criteria in HAV induced PALF.
Graphic abstract




Similar content being viewed by others
References
Alam S, Khanna R, Sood V, et al. Profile and outcome of first 109 cases of paediatric acute liver failure at a specialized paediatric liver unit in India. Liver Int 2017;37(10):1508–1514
Poddar U, Thapa BR, Prasad A, et al. Natural history and risk factors in fulminant hepatic failure. Arch Dis Child 2002;87:54–56
Kaur S, Kumar P, Kumar V, et al. Etiology and prognostic factors of acute liver failure in children. Indian Pediatr 2013;50(7):677–679
Kayaalp C, Ersan V, Yılmaz S. Acute liver failure in Turkey: a systematic review. Turk J Gastroenterol 2014;25(1):35–40
Ciocca M, Ramonet M, Cuarterolo M, et al. Prognostic factors in paediatric acute liver failure. Arch Dis Child 2008;93(1):48–51
Squires RH Jr, Shneider BL, Bucuvalas J, et al. Acute liver failure in children: the first 348 patients in the pediatric acute liver failure study group. J Pediatr 2006;148:652–658
Rajanayagam J, Coman D, Cartwright D, et al. Pediatric acute liver failure: etiology, outcomes, and the role of serial pediatric end-stage liver disease scores. Pediatr Transplant 2013;17:362–368
O'Grady JG, Alexander GJ, Hayllar KM, et al. Early indicators of prognosis in fulminant hepatic failure. Gastroenterology 1989;97(2):439–445
Sanchez MC, D'Agostino DE. Pediatric end-stage liver disease score in acute liver failure to assess poor prognosis. J Pediatr Gastroenterol Nutr 2012;54(2):193–196
Liu E, MacKenzie T, Dobyns EL, et al. Characterization of acute liver failure and development of a continuous risk of death staging system in children. J Hepatol 2006;44(1):134–141
Lu BR, Zhang S, Narkewicz MR, et al. Evaluation of the liver injury unit scoring system to predict survival in a multinational study of pediatric acute liver failure. J Pediatr 2013;162(5):1010–1016
Pollack MM, Patel KM, Ruttimann UE. PRISM III: an updated pediatric risk of mortality score. Crit Care Med 1996;24:743–752
Matthews CE, Goonasekera C, Dhawan A, et al. Validity of pediatric index of mortality 2 (PIM2) score in pediatric acute liver failure. Crit Care 2014;18(6):665
Jain V, Dhawan A. Prognostic modeling in pediatric acute liver failure. Liver Transpl 2016;22(10):1418–1430
Sundaram V, Shneider BL, Dhawan A, et al. King's College Hospital Criteria for non-acetaminophen induced acute liver failure in an international cohort of children. J Pediatr 2013;162(2):319–323
Aggarwal R, Goel A. Hepatitis A: epidemiology in resource-poor countries. Curr Opin Infect Dis 2015;28(5):488–496
Dhawan A, Taylor RM, Cheeseman P, et al. Wilson's disease in children: 37-year experience and revised King's score for liver transplantation. Liver Transpl 2005;11(4):441–448
Atterbury CE, Maddrey WC, Conn HO. Neomycin-sorbitol and lactulose in the treatment of acute portal-systemic encephalopathy. A controlled, double-blind clinical trial. Am J Dig Dis 1978;23:398–406
Kumar R, Shalimar SH, et al. Prospective derivation and validation of early dynamic model for predicting outcome in patients with acute liver failure. Gut 2012;61(7):1068–1075
Jacobsen KH, Wiersma ST. Hepatitis A virus seroprevalence by age and world region, 1990 and 2005. Vaccine 2010;28:6653–6657
Franco E, Meleleo C, Serino L, et al. Hepatitis A: epidemiology and prevention in developing countries. World J Hepatol 2012;4(3):68–73
Mcphail MJ, Farne H, Senvar N, et al. Ability of King's College criteria and model for end-stage liver disease scores to predict mortality of patients with acute liver failure: a meta-analysis. Clin Gastroenterol Hepatol 2016;14(4):516–525
McDiarmid SV, Anand R, Lindblad AS, Principal Investigators, and Institutions of the Studies of Pediatric Liver Transplantation (SPLIT) Research Group. Development of a pediatric end-stage liver disease score to predict poor outcome in children awaiting liver transplantation. Transplantation 2002;74(2):173–181
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Bikrant Bihari Lal, Vikrant Sood, Snehavardhan Pandey, Rajeev Khanna, Samba Siva Rao Pasupuleti, Manish Siloliya, Guresh Kumar and Seema Alam declare no conflict of interest.
Ethical requirements
All procedures followed were in accordance with the ethical standards of the responsible committee on human experimentation (institutional and national) and with the Helsinki Declaration of 1975, as revised in 2008. Ethical approval was obtained from the institutional ethical committee (IEC/2019/66/ MA04).
Informed consent
Informed consent was obtained from all patients included in prospective part of study.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
12072_2020_10050_MOESM3_ESM.tif
Supplementary Figure 3: Hypothetical scenario showing listing and mortality based on Peds-HAV model at each day from day of admission (day 0) to day 7 and corresponding outcome of the patients at each day. The curved arrows indicate patients that would have been listed or delisted from days 0 to 7 (TIF 1052 kb)
Rights and permissions
About this article
Cite this article
Lal, B.B., Sood, V., Snehavardhan, P. et al. A novel, bedside, etiology specific prognostic model (Peds-HAV) in hepatitis A induced pediatric acute liver failure. Hepatol Int 14, 483–490 (2020). https://doi.org/10.1007/s12072-020-10050-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12072-020-10050-0