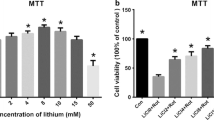
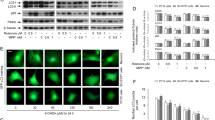
Abstract
Intracellular quality control regulated by autophagy process is important for maintenance of cellular homeostasis. Deregulation of autophagy and more specifically mitophagy leads to accumulation of the misfolded proteins and damaged mitochondria that in turn leads to the cell loss. Alteration of autophagy and mitophagy has shown to be involved in the number of disorders including neurodegenerative diseases. Autophagy and mitophagy could be activated by short-time acidification of the cytosol; however, most of the compounds which can induce it are toxic. Here, we tested several organic compounds which are involved in cellular metabolism on their ability to change intracellular pH and induce mitophagy/autophagy. We have found that lactate and pyruvate are able to reduce intracellular pH in non-toxic concentrations. Short-term (2 h) and long-term (24 h) incubation of the cells with lactate and pyruvateinduced mitophagy and autophagy. Incubation of the SH-SY5Y cells or primary neurons and astrocytes with lactate or pyruvate also activated mitophagy and autophagy after MPP + treatment that led to recovery of mitochondrial function and protection of these cells against apoptotic and necrotic death. Thus, pyruvate- or lactate-induced acidification of cytosol activates cell protective mitophagy and autophagy.






Similar content being viewed by others
Data Availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
Code Availability
Not applicable.
References
Bento CF, Renna M, Ghislat G, Puri C, Ashkenazi A, Vicinanza M, Menzies FM, Rubinsztein DC (2016) Mammalian autophagy: how does it work? Annu Rev Biochem 85:685–713. https://doi.org/10.1146/annurev-biochem-060815-014556
Farre JC, Subramani S (2004) Peroxisome turnover by micropexophagy: an autophagy-related process. Trends Cell Biol 14(9):515–523. https://doi.org/10.1016/j.tcb.2004.07.014
Kaushik S, Massey AC, Mizushima N, Cuervo AM (2008) Constitutive activation of chaperone-mediated autophagy in cells with impaired macroautophagy. Mol Biol Cell 19(5):2179–2192. https://doi.org/10.1091/mbc.E07-11-1155
Uttenweiler A, Schwarz H, Neumann H, Mayer A (2007) The vacuolar transporter chaperone (VTC) complex is required for microautophagy. Mol Biol Cell 18(1):166–175. https://doi.org/10.1091/mbc.e06-08-0664
Levine B, Klionsky DJ (2017) Autophagy wins the 2016 Nobel Prize in Physiology or Medicine: breakthroughs in baker’s yeast fuel advances in biomedical research. Proc Natl Acad Sci U S A 114(2):201–205. https://doi.org/10.1073/pnas.1619876114
Kim I, Rodriguez-Enriquez S, Lemasters JJ (2007) Selective degradation of mitochondria by mitophagy. Arch Biochem Biophys 462(2):245–253. https://doi.org/10.1016/j.abb.2007.03.034
Wang Y, Liu N, Lu B (2019) Mechanisms and roles of mitophagy in neurodegenerative diseases. CNS Neurosci Ther 25(7):859–875. https://doi.org/10.1111/cns.13140
Bratic A, Larsson NG (2013) The role of mitochondria in aging. J Clin Invest 123(3):951–957. https://doi.org/10.1172/JCI64125
Wallace DC (2012) Mitochondria and cancer. Nat Rev Cancer 12(10):685–698. https://doi.org/10.1038/nrc3365
Szendroedi J, Phielix E, Roden M (2011) The role of mitochondria in insulin resistance and type 2 diabetes mellitus. Nat Rev Endocrinol 8(2):92–103. https://doi.org/10.1038/nrendo.2011.138
Lezi E, Swerdlow RH (2012) Mitochondria in neurodegeneration. Adv Exp Med Biol 942:269–286. https://doi.org/10.1007/978-94-007-2869-1_12
Kitada T, Asakawa S, Hattori N, Matsumine H, Yamamura Y, Minoshima S, Yokochi M, Mizuno Y et al (1998) Mutations in the Parkin gene cause autosomal recessive juvenile parkinsonism. Nature 392(6676):605–608. https://doi.org/10.1038/33416
Arano T, Imai Y (2015) Mitophagy regulated by the PINK1-Parkin pathway. In: Ntuli TM (ed) Cell Death. IntechOpen. https://doi.org/10.5772/61284
Van Noorden R, Ledford H (2016) Medicine Nobel for research on how cells “eat themselves.” Nature 538(7623):18–19. https://doi.org/10.1038/nature.2016.20721
Abramov AY, Berezhnov AV, Fedotova EI, Zinchenko VP, Dolgacheva LP (2017) Interaction of misfolded proteins and mitochondria in neurodegenerative disorders. Biochem Soc Trans 45(4):1025–1033. https://doi.org/10.1042/BST20170024
Berezhnov AV, Soutar MP, Fedotova EI, Frolova MS, Plun-Favreau H, Zinchenko VP, Abramov AY (2016) Intracellular pH modulates autophagy and mitophagy. J Biol Chem 291(16):8701–8708. https://doi.org/10.1074/jbc.M115.691774
Behbahan I, McBrian M, Kurdistani S (2014) A protocol for measurement of intracellular pH. Bio-Protocol 4(2). https://doi.org/10.21769/BioProtoc.1027
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) Method. Methods 25(4):402–408. https://doi.org/10.1006/meth.2001.1262
Dauer W, Przedborski S (2003) Parkinson’s disease: mechanisms and models. Neuron 39(6):889–909. https://doi.org/10.1016/s0896-6273(03)00568-3
Riske L, Thomas RK, Baker GB, Dursun SM (2017) Lactate in the brain: an update on its relevance to brain energy, neurons, glia and panic disorder. Ther Adv Psychopharmacol 7(2):85–89. https://doi.org/10.1177/2045125316675579
Lezi E, Swerdlow RH (2016) Lactate’s effect on human neuroblastoma cell bioenergetic fluxes. Biochem Pharmacol 99:88–100. https://doi.org/10.1016/j.bcp.2015.11.002
Orij R, Postmus J, Ter Beek A, Brul S, Smits GJ (2009) In vivo measurement of cytosolic and mitochondrial pH using a pH-sensitive GFP derivative in Saccharomyces cerevisiae reveals a relation between intracellular pH and growth. Microbiology 155(Pt 1):268–278. https://doi.org/10.1099/mic.0.022038-0
Owens LM, Fralix TA, Murphy E, Cascio WE, Gettes LS (1996) Correlation of ischemia-induced extracellular and intracellular ion changes to cell-to-cell electrical uncoupling in isolated blood-perfused rabbit hearts. Experimental Working Group. Circulation 94(1):10–13. https://doi.org/10.1161/01.cir.94.1.10
Marino ML, Pellegrini P, Di Lernia G, Djavaheri-Mergny M, Brnjic S, Zhang X, Hagg M, Linder S et al (2012) Autophagy is a protective mechanism for human melanoma cells under acidic stress. J Biol Chem 287(36):30664–30676. https://doi.org/10.1074/jbc.M112.339127
Astrand PO, Hallback I, Hedman R, Saltin B (1963) Blood lactates after prolonged severe exercise. J Appl Physiol 18(3):619–622. https://doi.org/10.1152/jappl.1963.18.3.619
Messias LHD, Gobatto CA, Beck WR, Manchado-Gobatto FB (2017) The lactate minimum test: concept, methodological aspects and insights for future investigations in human and animal models. Front Physiol 8:389. https://doi.org/10.3389/fphys.2017.00389
Quistorff B, Secher NH, Van Lieshout JJ (2008) Lactate fuels the human brain during exercise. FASEB J 22(10):3443–3449. https://doi.org/10.1096/fj.08-106104
Gandhi S, Vaarmann A, Yao Z, Duchen MR, Wood NW, Abramov AY (2012) Dopamine induced neurodegeneration in a PINK1 model of Parkinson’s disease. PLoS One 7(5):e37564. https://doi.org/10.1371/journal.pone.0037564
Kovac S, Domijan AM, Walker MC, Abramov AY (2012) Prolonged seizure activity impairs mitochondrial bioenergetics and induces cell death. J Cell Sci 125(Pt 7):1796–1806. https://doi.org/10.1242/jcs.099176
Abramov AY, Angelova PR (2019) Cellular mechanisms of complex I-associated pathology. Biochem Soc Trans 47(6):1963–1969. https://doi.org/10.1042/BST20191042
Tauffenberger A, Fiumelli H, Almustafa S, Magistretti PJ (2019) Lactate and pyruvate promote oxidative stress resistance through hormetic ROS signaling. Cell Death Dis 10(9):653. https://doi.org/10.1038/s41419-019-1877-6
Georgakopoulos ND, Frison M, Alvarez MS, Bertrand H, Wells G, Campanella M (2017) Reversible Keap1 inhibitors are preferential pharmacological tools to modulate cellular mitophagy. Sci Rep 7(1):10303. https://doi.org/10.1038/s41598-017-07679-7
Funding
The study was funded by The Russian Foundation for Basic Research (RFBR), project number 20–34-70074. This work was also supported by the grant of the Russian Federation government no. 075–15-2019–1877.
Author information
Authors and Affiliations
Contributions
All authors contributed equally to this study.
Corresponding author
Ethics declarations
Ethics Approval
Animal studies were approved by the Animal Ethics Committee of the Institute of Cell Biophysics, Russian Academy of Sciences, and were performed in compliance with the Russian Federation legislation.
Consent to Participate
Not applicable.
Consent for Publication
Not applicable.
Conflict of Interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Fedotova, E.I., Dolgacheva, L.P., Abramov, A.Y. et al. Lactate and Pyruvate Activate Autophagy and Mitophagy that Protect Cells in Toxic Model of Parkinson’s Disease. Mol Neurobiol 59, 177–190 (2022). https://doi.org/10.1007/s12035-021-02583-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12035-021-02583-8