Abstract
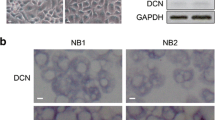
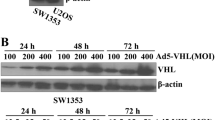
Oncolytic adenovirus is an emerging alternative to current therapeutics. The adenoviral E1A, the first protein expressed upon oncolytic adenoviral infection, has been identified as an antitumor agent, but the mechanisms of its tumor inhibition ability are unclear enough. Decorin is ubiquitous in the extracellular matrix (ECM), which regulates multiple functions through interaction with ECM. Here, we intended to explore the effects of adenoviral E1A on the tumor extracellular matrix during gene therapy. We demonstrated that reduced decorin expression was found in patients with lung cancer. The adenoviral E1A or a mutant adenoviral E1A with Rb-binding ability absent (E1A 30–60aa, 120–127aa deletion) could increase the expression of decorin and down-regulate VEGF, two members of tumor ECM, involved in both vasculogenesis and angiogenesis. E1A/mE1A-mediated suppressing the migration and invasion ability of tumor cells was depended on decorin. E1A interacted with decorin directly and induced the proteasomal degradation of VEGF. In addition, E1A or mE1A can inhibit tumor growth in a subcutaneous lung cancer xenograft model. It suggested that decorin might be a crucial mediator among ECM components for adenoviral E1A-mediated antitumor activities. These studies on adenovirus E1A provide a new mechanism for the emerging therapies of tumor gene therapy.






Similar content being viewed by others
References
Yano T, Kawashima O, Takeo S, Adachi H, Tagawa T, Fukuyama S, et al. A prospective observational study of pulmonary resection for non-small cell lung cancer in patients older than 75 years. Semin Thorac Cardiovasc Surg. 2017;29(4):540–7. https://doi.org/10.1053/j.semtcvs.2017.05.004.
Dayen C, Debieuvre D, Molinier O, Raffy O, Paganin F, Virally J, et al. New insights into stage and prognosis in small cell lung cancer: an analysis of 968 cases. J Thorac Dis. 2017;9(12):5101–11. https://doi.org/10.21037/jtd.2017.11.52.
Bommareddy PK, Patel A, Hossain S, Kaufman HL. Talimogene Laherparepvec (T-VEC) and other oncolytic viruses for the treatment of melanoma. Am J Clin Dermatol. 2017;18(1):1–15. https://doi.org/10.1007/s40257-016-0238-9.
Radko S, Koleva M, James KM, Jung R, Mymryk JS, Pelka P. Adenovirus E1A targets the DREF nuclear factor to regulate virus gene expression, DNA replication, and growth. J Virol. 2014;88(22):13469–81. https://doi.org/10.1128/JVI.02538-14.
Su JL, Chen PB, Chen YH, Chen SC, Chang YW, Jan YH, et al. Downregulation of microRNA miR-520 h by E1A contributes to anticancer activity. Cancer Res. 2010;70(12):5096–108. https://doi.org/10.1158/0008-5472.CAN-09-4148.
Chen HA, Chang YW, Tseng CF, Chiu CF, Hong CC, Wang W, et al. E1A-mediated inhibition of HSPA5 suppresses cell migration and invasion in triple-negative breast cancer. Ann Surg Oncol. 2015;22(3):889–98. https://doi.org/10.1245/s10434-014-4061-3.
Li Z, Day CP, Yang JY, Tsai WB, Lozano G, Shih HM, et al. Adenoviral E1A targets Mdm4 to stabilize tumor suppressor p53. Can Res. 2004;64(24):9080–5. https://doi.org/10.1158/0008-5472.CAN-04-2419.
Fang L, Cheng Q, Zhao J, Ge Y, Zhu Q, Zhao M, et al. A p53-independent apoptotic mechanism of adenoviral mutant E1A was involved in its selective antitumor activity for human cancer. Oncotarget. 2016;7(30):48309–20. https://doi.org/10.18632/oncotarget.10221.
Fang L, Cheng Q, Bai J, Qi YD, Liu JJ, Li LT, et al. An oncolytic adenovirus expressing interleukin-24 enhances antitumor activities in combination with paclitaxel in breast cancer cells. Mol Med Rep. 2013;8(5):1416–24. https://doi.org/10.3892/mmr.2013.1680.
Jiang G, Jiang AJ, Cheng Q, Tian H, Li LT, Zheng JN. A dual-regulated oncolytic adenovirus expressing interleukin-24 sensitizes melanoma cells to temozolomide via the induction of apoptosis. Tumour Biol. 2013;34(2):1263–71. https://doi.org/10.1007/s13277-013-0701-7.
Zhang W, Ge Y, Cheng Q, Zhang Q, Fang L, Zheng J. Decorin is a pivotal effector in the extracellular matrix and tumour microenvironment. Oncotarget. 2018;9(4):5480–91. https://doi.org/10.18632/oncotarget.23869.
Gubbiotti MA, Vallet SD, Ricard-Blum S, Iozzo RV. Decorin interacting network: a comprehensive analysis of decorin-binding partners and their versatile functions. Matrix Biol. 2016;55:7–21. https://doi.org/10.1016/j.matbio.2016.09.009.
Bierbaum S, Douglas T, Hanke T, Scharnweber D, Tippelt S, Monsees TK, et al. Collageneous matrix coatings on titanium implants modified with decorin and chondroitin sulfate: characterization and influence on osteoblastic cells. J Biomed Mater Res A. 2006;77(3):551–62. https://doi.org/10.1002/jbm.a.30572.
Bi XL, Yang W. Biological functions of decorin in cancer. Chin J Cancer. 2013;32(5):266–9. https://doi.org/10.5732/cjc.012.10301.
Parato KA, Senger D, Forsyth PA, Bell JC. Recent progress in the battle between oncolytic viruses and tumours. Nat Rev Cancer. 2005;5(12):965–76. https://doi.org/10.1038/nrc1750.
Choi IK, Lee YS, Yoo JY, Yoon AR, Kim H, Kim DS, et al. Effect of decorin on overcoming the extracellular matrix barrier for oncolytic virotherapy. Gene Ther. 2010;17(2):190–201. https://doi.org/10.1038/gt.2009.142.
Goetsch KP, Niesler CU. The extracellular matrix regulates the effect of decorin and transforming growth factor beta-2 (TGF-beta2) on myoblast migration. Biochem Biophys Res Commun. 2016;479(2):351–7. https://doi.org/10.1016/j.bbrc.2016.09.079.
Yao T, Zhang CG, Gong MT, Zhang M, Wang L, Ding W. Decorin-mediated inhibition of the migration of U87MG glioma cells involves activation of autophagy and suppression of TGF-beta signaling. FEBS Open Bio. 2016;6(7):707–19. https://doi.org/10.1002/2211-5463.12076.
Shi X, Liang W, Yang W, Xia R, Song Y. Decorin is responsible for progression of non-small-cell lung cancer by promoting cell proliferation and metastasis. Tumour Biol. 2015;36(5):3345–54. https://doi.org/10.1007/s13277-014-2968-8.
Liang S, Xu JF, Cao WJ, Li HP, Hu CP. Human decorin regulates proliferation and migration of human lung cancer A549 cells. Chin Med J (Engl). 2013;126(24):4736–41.
Wu Y, Xiao T, Li M, Zhang Y, Gao Y, Sun K. Decorin protein is down-regulated in non-small cell lung cancer tissue and significantly associated with histological type. Zhongguo Fei Ai Za Zhi. 2011;14(11):848–52. https://doi.org/10.3779/j.issn.1009-3419.2011.11.03.
Xu L, Wang F, Liu H, Xu XF, Mo WH, Xia YJ, et al. Increased expression of cellular repressor of E1A-stimulated gene (CREG) in gastric cancer patients: a mechanism of proliferation and metastasis in cancer. Dig Dis Sci. 2011;56(6):1645–55. https://doi.org/10.1007/s10620-010-1510-0.
Matsubara S, Wada Y, Gardner TA, Egawa M, Park MS, Hsieh CL, et al. A conditional replication-competent adenoviral vector, Ad-OC-E1a, to cotarget prostate cancer and bone stroma in an experimental model of androgen-independent prostate cancer bone metastasis. Can Res. 2001;61(16):6012–9.
Marshall KS, Cohen MJ, Fonseca GJ, Todorovic B, King CR, Yousef AF, et al. Identification and characterization of multiple conserved nuclear localization signals within adenovirus E1A. Virology. 2014;454–455:206–14. https://doi.org/10.1016/j.virol.2014.02.020.
Yu D, Hung MC. The erbB2 gene as a cancer therapeutic target and the tumor- and metastasis-suppressing function of E1A. Cancer Metast Rev. 1998;17(2):195–202.
Fang L, Huang Y, Hu X, Sun L, He X, Hu H, et al. A truncated minimal-E1a gene with potency to support adenoviral replication mediates antitumor activity by down-regulating Neu expression and preserving Rb function. Chem Biol Interact. 2009;181(1):1–7. https://doi.org/10.1016/j.cbi.2009.06.002.
Su CM, Chang TY, Hsu HP, Lai HH, Li JN, Lyu YJ, et al. A novel application of E1A in combination therapy with EGFR-TKI treatment in breast cancer. Oncotarget. 2016;7(39):63924–36. https://doi.org/10.18632/oncotarget.11737.
Flinterman MB, Mymryk JS, Klanrit P, Yousef AF, Lowe SW, Caldas C, et al. p400 function is required for the adenovirus E1A-mediated suppression of EGFR and tumour cell killing. Oncogene. 2007;26(48):6863–74. https://doi.org/10.1038/sj.onc.1210497.
Yamasaki Y, Tazawa H, Hashimoto Y, Kojima T, Kuroda S, Yano S, et al. A novel apoptotic mechanism of genetically engineered adenovirus-mediated tumour-specific p53 overexpression through E1A-dependent p21 and MDM2 suppression. Eur J Cancer. 2012;48(14):2282–91. https://doi.org/10.1016/j.ejca.2011.12.020.
Das S, El-Deiry WS, Somasundaram K. Regulation of the p53 homolog p73 by adenoviral oncogene E1A. J Biol Chem. 2003;278(20):18313–20. https://doi.org/10.1074/jbc.M211704200.
Su JL, Cheng X, Yamaguchi H, Chang YW, Hou CF, Lee DF, et al. FOXO3a-dependent mechanism of E1A-induced chemosensitization. Can Res. 2011;71(21):6878–87. https://doi.org/10.1158/0008-5472.CAN-11-0295.
Liao Y, Zou YY, Xia WY, Hung MC. Enhanced paclitaxel cytotoxicity and prolonged animal survival rate by a nonviral-mediated systemic delivery of E1A gene in orthotopic xenograft human breast cancer. Cancer Gene Ther. 2004;11(9):594–602. https://doi.org/10.1038/sj.cgt.7700743.
Ueno NT, Bartholomeusz C, Herrmann JL, Estrov Z, Shao R, Andreeff M, et al. E1A-mediated paclitaxel sensitization in HER-2/neu-overexpressing ovarian cancer SKOV3ip1 through apoptosis involving the caspase-3 pathway. Clin Cancer Res. 2000;6(1):250–9.
King CR, Zhang A, Tessier TM, Gameiro SF, Mymryk JS. Hacking the cell: network intrusion and exploitation by adenovirus E1A. mBio. 2018. https://doi.org/10.1128/mBio.00390-18.
Deissler H, Opalka B. Therapeutic transfer of DNA encoding adenoviral E1A. Recent Pat Anticancer Drug Discov. 2007;2(1):1–10.
Buraschi S, Neill T, Iozzo RV. Decorin is a devouring proteoglycan: remodeling of intracellular catabolism via autophagy and mitophagy. Matrix Biol. 2019;75–76:260–70. https://doi.org/10.1016/j.matbio.2017.10.005.
Acknowledgements
This project is supported by grants from the National Key Research and Development Plan (2018YFA0900900), the National Natural Science Foundation of China (Grant Nos. 81872488, 81372460), the Natural Science Foundation of Jiangsu Province (Grants Nos. BK20161156, BK20161157), the Education Department of Jiangsu Province (Grant Nos. 13KJB320028, 12KJA320001), Social Development Key Project of Jiangsu Province (Grant Nos. BE2016643), Jiangsu Provincial Medical Youth Talent (Grant Nos. QNRC2016774, QNRC2016773, CXTDA2017034, The Project of Invigorating Health Care through Science, Technology and Education) and Jiangsu Provincial Key Medical Discipline, The Project of Invigorating Health Care through Science, Technology and Education (No. ZDXKA2016014).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflicts of interest
The authors declare that they have no conflict of interest.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. Animal welfare and experimental procedures were carried out strictly in accordance with Guide for the Care and Use of Laboratory Animals (National Research Council, 1996) and all procedures performed in studies involving animals were in accordance with the ethical standards of the Animal Care Committee of the Xuzhou Medical University, Xuzhou, China.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Ge, Y., Zhang, W., Qin, J. et al. A novel role mediated by adenoviral E1A in suppressing cancer through modulating decorin. Med Oncol 36, 96 (2019). https://doi.org/10.1007/s12032-019-1325-6
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s12032-019-1325-6