Abstract
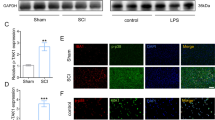
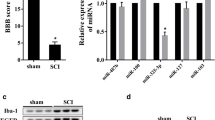
Microglial cell activation after spinal cord ischemia-reperfusion injury (SCIRI) commonly causes the secondary nerve motion function injury. This study aims to study the mechanism by which the drug dexmedetomidine (DEX) inhibits microglial cell activation and improves motion function of SCIRI mice. Mice SCIRI model was established, and microglia from spinal cord were isolated and cultured for subsequent molecule analysis of let-7a-1-3p, let-7a-2-3p, HMGB1, TNF-α, and IL-6. DEX was given by intraperitoneal injection. Mice motion function was evaluated by Basso mouse score. In vitro microglial cells were subjected to oxygen and glucose deprivation/reoxygenation (OGD/R) to imitate ischemia-reperfusion injury stimulation. DEX injection improves the mouse motion function in SCIRI model and upregulates let-7a-1/2-3p expression in the isolated activated microglia from SCIRI mice. In OGD/R-stimulated microglia, DEX treatment also caused the inactivation of cells, the upregulation of let-7a-1/2-3p expression, and the downregulation of HMGB1 expression. While the co-silencing of let-7a-1/2-3p in microglia in addition to DEX treatment restored the activation of microglia. HMGB1 is a targeted gene for let-7a-1/2-3p and negatively regulated by them. HMGB1 knockdown abrogates the pro-activation impact on microglial cell by let-7a-1/2-3p silencing. DEX inhibits the activation of microglial cell in the spinal cord of SCIRI mice, mediated by the let-7a-1/2-3p/HMGB1 pathway.






Similar content being viewed by others
References
Anwar MA, Al Shehabi TS, Eid AH (2016) Inflammogenesis of secondary spinal cord injury. Front Cell Neurosci 10:98. https://doi.org/10.3389/fncel.2016.00098
Aslan A, Cemek M, Eser O, Altunbaş K, Buyukokuroglu ME, Cosar M, Baş O, Ela Y, Fidan H (2009) Does dexmedetomidine reduce secondary damage after spinal cord injury? An experimental study. Eur Spine J 18:336–344. https://doi.org/10.1007/s00586-008-0872-x
Bao N, Fang B, Lv H, Jiang Y, Chen F, Wang Z, Ma H (2018) Upregulation of miR-199a-5p protects spinal cord against ischemia/reperfusion-induced injury via downregulation of ECE1 in rat. Cell Mol Neurobiol 38:1293–1303. https://doi.org/10.1007/s10571-018-0597-2
Bell MT, Agoston VA, Freeman KA, Puskas F, Herson PS, Mares J, Fullerton DA, Reece TB (2014) Interruption of spinal cord microglial signaling by alpha-2 agonist dexmedetomidine in a murine model of delayed paraplegia. J Vasc Surg 59:1090–1097. https://doi.org/10.1016/j.jvs.2013.04.050
Bell MT, Puskas F, Smith PD, Agoston VA, Fullerton DA, Meng X, Weyant MJ, Reece TB (2012a) Attenuation of spinal cord ischemia-reperfusion injury by specific α-2a receptor activation with dexmedetomidine. J Vasc Surg 56:1398–1402
Bell MT, Puskas F, Agoston VA, Cleveland JC, Freeman KA, Gamboni F, Herson PS, Meng X, Smith PD, Weyant MJ, Fullerton DA, Reece TB (2013) Toll-like receptor 4-dependent microglial activation mediates spinal cord ischemia-reperfusion injury. Circulation 128:S152–S156. https://doi.org/10.1161/CIRCULATIONAHA.112.000024
Bell MT, Puskas F, Smith PD, Agoston VA, Fullerton DA, Meng X, Weyant MJ, Reece TB (2012b) Attenuation of spinal cord ischemia-reperfusion injury by specific alpha-2a receptor activation with dexmedetomidine. J Vasc Surg 56:1398–1402. https://doi.org/10.1016/j.jvs.2012.04.012
Brites D, Fernandes A (2015) Neuroinflammation and depression: microglia activation, extracellular microvesicles and microRNA dysregulation. Front Cell Neurosci 9:476. https://doi.org/10.3389/fncel.2015.00476
Celik F, Göçmez C, Kamaşak K, Tufek A, Guzel A, Tokgoz O, Fırat U, Evliyaoğlu O (2013) The comparison of neuroprotective effects of intrathecal dexmedetomidine and metilprednisolone in spinal cord injury. Int J Surg 11:414–418. https://doi.org/10.1016/j.ijsu.2013.03.008
Cho KJ, Song J, Oh Y, Lee JE (2015) MicroRNA-let-7a regulates the function of microglia in inflammation. Mol Cell Neurosci 68:167–176. https://doi.org/10.1016/j.mcn.2015.07.004
Gurer B et al (2017) Comparative effects of vitamin D and methylprednisolone against ischemia/reperfusion injury of rabbit spinal cords. Eur J Pharmacol 813:50–60. https://doi.org/10.1016/j.ejphar.2017.07.028
Li XQ, Cao XZ, Wang J, Fang B, Tan WF, Ma H (2014) Sevoflurane preconditioning ameliorates neuronal deficits by inhibiting microglial MMP-9 expression after spinal cord ischemia/reperfusion in rats. Mol Brain 7:69. https://doi.org/10.1186/s13041-014-0069-7
Liu K, Yan L, Jiang X, Yu Y, Liu H, Gu T, Shi E (2017) Acquired inhibition of microRNA-124 protects against spinal cord ischemia-reperfusion injury partially through a mitophagy-dependent pathway. J Thorac Cardiovasc Surg 154:1498–1508. https://doi.org/10.1016/j.jtcvs.2017.05.046
Miranda V, Sousa J, Mansilha A (2018) Spinal cord injury in endovascular thoracoabdominal aortic aneurysm repair: prevalence, risk factors and preventive strategies. Int Angiol 37:112–126. https://doi.org/10.23736/S0392-9590.18.03960-3
Nieto-Diaz M, Esteban FJ, Reigada D, Muñoz-Galdeano T, Yunta M, Caballero-López M, Navarro-Ruiz R, Águila Á, Maza RM (2014) MicroRNA dysregulation in spinal cord injury: causes, consequences and therapeutics. Front Cell Neurosci 8:53. https://doi.org/10.3389/fncel.2014.00053
Paudel YN, Shaikh MF, Chakraborti A, Kumari Y, Aledo-Serrano Á, Aleksovska K, Alvim MKM, Othman I (2018) HMGB1: a common biomarker and potential target for TBI, neuroinflammation, epilepsy, and cognitive dysfunction. Front Neurosci 12:628. https://doi.org/10.3389/fnins.2018.00628
Qiao Y, Peng C, Li J, Wu D, Wang X (2018) LncRNA MALAT1 is neuroprotective in a rat model of spinal cord ischemia-reperfusion injury through miR-204 regulation. Curr Neurovasc Res 15:211–219. https://doi.org/10.2174/1567202615666180712153150
Rong H, Zhao Z, Feng J, Lei Y, Wu H, Sun R, Zhang Z, Hou B, Zhang W, Sun YE, Gu X, Ma Z, Liu Y (2017) The effects of dexmedetomidine pretreatment on the pro- and anti-inflammation systems after spinal cord injury in rats. Brain Behav Immun 64:195–207. https://doi.org/10.1016/j.bbi.2017.03.006
Shi Y, Guo X, Zhang J, Zhou H, Sun B, Feng J (2018) DNA binding protein HMGB1 secreted by activated microglia promotes the apoptosis of hippocampal neurons in diabetes complicated with OSA. Brain Behav Immun 73:482–492. https://doi.org/10.1016/j.bbi.2018.06.012
Smith PD, Bell MT, Puskas F, Meng X, Cleveland JC Jr, Weyant MJ, Fullerton DA, Reece TB (2013) Preservation of motor function after spinal cord ischemia and reperfusion injury through microglial inhibition. Ann Thorac Surg 95:1647–1653. https://doi.org/10.1016/j.athoracsur.2012.11.075
Sun Z, Zhao T, Lv S, Gao Y, Masters J, Weng H (2018) Dexmedetomidine attenuates spinal cord ischemia-reperfusion injury through both anti-inflammation and anti-apoptosis mechanisms in rabbits. J Transl Med 16:209. https://doi.org/10.1186/s12967-018-1583-7
Tan JA, Ho KM (2010) Use of dexmedetomidine as a sedative and analgesic agent in critically ill adult patients: a meta-analysis. Intensive Care Med 36:926–939. https://doi.org/10.1007/s00134-010-1877-6
Tian X, Liu C, Shu Z, Chen G (2017) Review: therapeutic targeting of HMGB1 in stroke. Curr Drug Deliv 14:785–790. https://doi.org/10.2174/1567201813666160808111933
Zheng B, Zhang S, Ying Y, Guo X, Li H, Xu L, Ruan X (2018) Administration of Dexmedetomidine inhibited NLRP3 inflammasome and microglial cell activities in hippocampus of traumatic brain injury rats. Biosci Rep 38:BSR20180892. https://doi.org/10.1042/BSR20180892
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
All protocols involved animals have been approved by the animal ethics committee of Renmin Hospital of Wuhan University.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
ESM 1
(PNG 149 kb)
Rights and permissions
About this article
Cite this article
Ha sen ta na, Nuo, M., Meng, Qt. et al. The Pathway of Let-7a-1/2-3p and HMGB1 Mediated Dexmedetomidine Inhibiting Microglia Activation in Spinal Cord Ischemia-Reperfusion Injury Mice. J Mol Neurosci 69, 106–114 (2019). https://doi.org/10.1007/s12031-019-01338-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12031-019-01338-4