Abstract
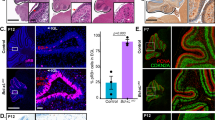
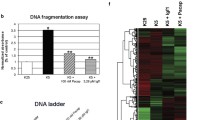
Secretin (SCT) is involved in a variety of physiological processes and has been implicated in preventing apoptosis during brain development. However, little is known about the molecular mechanism underlying its neuroprotective effects. The B cell lymphoma 2 (Bcl-2) family proteins, such as Bcl-2 and Bcl-xL, determine the commitment of neurons to apoptosis. In SCT knockout mice, we found reduced transcript levels of anti-apoptotic genes Bcl-2 and Bcl-xL, but not of pro-apoptotic gene Bax, in the developing cerebellum. SCT treatment on ex vivo cultured cerebellar slices triggered a time-dependent increase of Bcl-2 and Bcl-xL expression. This SCT-induced transcriptional regulation of Bcl-2 and Bcl-xL was dependent on the cyclic AMP (cAMP) response element-binding protein (CREB), which is a key survival factor at the convergence of multiple signaling cascades. We further demonstrated that activation of CREB by SCT was mediated by cAMP/protein kinase A (PKA) and mitogen-activated protein kinase (MAPK)/extracellular signal-regulated kinase 1/2 (ERK1/2) cascades. These findings, collectively, provide an uncharacterized signaling cascade for SCT-mediated neuronal survival, in which SCT promotes the key anti-apoptotic elements Bcl-2 and Bcl-xL in the intrinsic death pathway through PKA- and ERK-regulated CREB phosphorylation.





Similar content being viewed by others
References
Ballif BA, Blenis J (2001) Molecular mechanisms mediating mammalian mitogen-activated protein kinase (MAPK) kinase (MEK)-MAPK cell survival signals. Cell Growth Differ 12:397–408
Bhave S, Hoffman P (2004) Phosphatidylinositol 3′-OH kinase and protein kinase a pathways mediate the anti-apoptotic effect of pituitary adenylyl cyclase-activating polypeptide in cultured cerebellar granule neurons: modulation by ethanol. J Neurochem 88:359–369. https://doi.org/10.1046/j.1471-4159.2003.02167.x
Castorina A, Tiralongo A, Giunta S, Carnazza ML, Rasi G, D'Agata V (2008) PACAP and VIP prevent apoptosis in schwannoma cells. Brain Res 1241:29–35. https://doi.org/10.1016/j.brainres.2008.09.035
Cheng EHYA, Wei MC, Weiler S, Flavell RA, Mak TW, Lindsten T, Korsmeyer SJ (2001) BCL-2, BCL-XL sequester BH3 domain-only molecules preventing BAX- and BAK-mediated mitochondrial apoptosis. Mol Cell 8:705–711. https://doi.org/10.1016/S1097-2765(01)00320-3
Czabotar PE, Lessene G, Strasser A, Adams JM (2014) Control of apoptosis by the BCL-2 protein family: implications for physiology and therapy. Nat Rev Mol Cell Biol 15:49–63
Dohi K, Mizushima H, Nakajo S, Ohtaki H, Matsunaga S, Aruga T, Shioda S (2002) Pituitary adenylate cyclase-activating polypeptide (PACAP) prevents hippocampal neurons from apoptosis by inhibiting JNK/SAPK and p38 signal transduction pathways. Regul Pept 109:83–88. https://doi.org/10.1016/S0167-0115(02)00190-8
Du C, Fang M, Li Y, Li L, Wang X (2000) Smac, a mitochondrial protein that promotes cytochrome c–dependent caspase activation by eliminating IAP inhibition. Cell 102:33–42
Eskes R, Desagher S, Antonsson B, Martinou J-C (2000) Bid induces the oligomerization and insertion of Bax into the outer mitochondrial membrane. Mol Cell Biol 20:929–935
Falluel-Morel A, Aubert N, Vaudry D, Basille M, Fontaine M, Fournier A, Vaudry H, Gonzalez BJ (2004) Opposite regulation of the mitochondrial apoptotic pathway by C2-ceramide and PACAP through a MAP-kinase-dependent mechanism in cerebellar granule cells. J Neurochem 91:1231–1243
Fang X, Yu S, Eder A, Mao M, Bast RC Jr, Boyd D, Mills GB (1999) Regulation of BAD phosphorylation at serine 112 by the Ras-mitogen-activated protein kinase pathway. Oncogene 18:6635–6640
Fletcher JI, Meusburger S, Hawkins CJ, Riglar DT, Lee EF, Fairlie WD, Huang DCS, Adams JM (2008) Apoptosis is triggered when prosurvival Bcl-2 proteins cannot restrain Bax. Proc Natl Acad Sci U S A 105:18081–18087
Fuchs Y, Steller H (2011) Programmed cell death in animal development and disease. Cell 147:742–758. https://doi.org/10.1016/j.cell.2011.10.033
Ghoumari AM, Wehrlé R, Bernard O, Sotelo C, Dusart I (2000) Implication of Bcl-2 and Caspase-3 in age-related Purkinje cell death in murine organotypic culture: an in vitro model to study apoptosis. Eur J Neurosci 12:2935–2949
Gutiérrez-Cañas I, Rodríguez-Henche N, Bolaños O, Carmena MJ, Prieto JC, Juarranz MG (2003) VIP and PACAP are autocrine factors that protect the androgen-independent prostate cancer cell line PC-3 from apoptosis induced by serum withdrawal. Brit J Pharmacol 139:1050–1058. https://doi.org/10.1038/sj.bjp.0705317
Heaton MB, Moore DB, Paiva M, Gibbs T, Bernard O (1999) Bcl-2 overexpression protects the neonatal cerebellum from ethanol neurotoxicity. Brain Res 817:13–18. https://doi.org/10.1016/S0006-8993(98)01173-1
Hurtado de Mendoza T, Balana B, Slesinger PA, Verma IM (2011) Organotypic cerebellar cultures: apoptotic challenges and detection. J Vis Exp. https://doi.org/10.3791/2564
Hwang DW, Givens B, Nishijima I (2009) Ethanol-induced developmental neurodegeneration in secretin receptor-deficient mice. Neuroreport 20:698–701. https://doi.org/10.1097/WNR.0b013e32832a5c9e
Jukkola PI, Rogers JT, Kaspar BK, Weeber EJ, Nishijima I (2011) Secretin deficiency causes impairment in survival of neural progenitor cells in mice. Hum Mol Genet 20:1000–1007. https://doi.org/10.1093/hmg/ddq545
Kerr JF, Wyllie AH, Currie AR (1972) Apoptosis: a basic biological phenomenon with wideranging implications in tissue kinetics. Brit J Cancer 26:239–257
Kim HS, Yumkham S, Kim SH, Yea K, Shin YC, Ryu SH, Suh PG (2006) Secretin induces neurite outgrowth of PC12 through cAMP-mitogen-activated protein kinase pathway. Exp Mol Med 38:85–93. https://doi.org/10.1038/emm.2006.10
Kim S-J, Nian C, Widenmaier S, McIntosh CH (2008) Glucose-dependent insulinotropic polypeptide-mediated up-regulation of β-cell antiapoptotic Bcl-2 gene expression is coordinated by cyclic AMP (cAMP) response element binding protein (CREB) and cAMP-responsive CREB coactivator 2. Mol Cell Biol 28:1644–1656
Kluck RM, Bossy-Wetzel E, Green DR, Newmeyer DD (1997) The release of cytochrome c from mitochondria: a primary site for Bcl-2 regulation of apoptosis. Science 275:1132–1136
Kluck RM, Esposti MD, Perkins G, Renken C, Kuwana T, Bossy-Wetzel E, Goldberg M, Allen T, Barber MJ, Green DR, Newmeyer DD (1999) The pro-apoptotic proteins, bid and Bax, cause a limited permeabilization of the mitochondrial outer membrane that is enhanced by cytosol. J Cell Biol 147:809–822
Kuwana T, Bouchier-Hayes L, Chipuk JE, Bonzon C, Sullivan BA, Green DR, Newmeyer DD (2005) BH3 domains of BH3-only proteins differentially regulate Bax-mediated mitochondrial membrane permeabilization both directly and indirectly. Mol Cell 17:525–535
Lee VH, Lee LT, Chu JY, Lam IP, Siu FK, Vaudry H, Chow BK (2010) An indispensable role of secretin in mediating the osmoregulatory functions of angiotensin II. FASEB J 24:5024–5032
Li P, Nijhawan D, Budihardjo I, Srinivasula SM, Ahmad M, Alnemri ES, Wang X (1997) Cytochrome c and dATP-dependent formation of Apaf-1/Caspase-9 complex initiates an apoptotic protease Cascade. Cell 91:479–489. https://doi.org/10.1016/S0092-8674(00)80434-1
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2− ΔΔCT method. Methods 25:402–408
Luciano F, Jacquel A, Colosetti P, Herrant M, Cagnol S, Pages G, Auberger P (2003) Phosphorylation of Bim-EL by Erk1/2 on serine 69 promotes its degradation via the proteasome pathway and regulates its proapoptotic function. Oncogene 22:6785–6793
May V, Lutz E, MacKenzie C, Schutz KC, Dozark K, Braas KM (2010) Pituitary adenylate cyclase-activating polypeptide (PACAP)/PAC1HOP1 receptor activation coordinates multiple neurotrophic signaling pathways: Akt activation through phosphatidylinositol 3-kinase gamma and vesicle endocytosis for neuronal survival. J Biol Chem 285:9749–9761. https://doi.org/10.1074/jbc.M109.043117
Meier P, Finch A, Evan G (2000) Apoptosis in development. Nature 407:796–801
Merry DE, Veis DJ, Hickey WF, Korsmeyer SJ (1994) Bcl-2 protein expression is widespread in the developing nervous system and retained in the adult PNS. Development 120:301–311
Opferman JT, Kothari A (2017) Anti-apoptotic BCL-2 family members in development. Cell Death Differ 25:37–45. https://doi.org/10.1038/cdd.2017.170
Riccio A, Ahn S, Davenport CM, Blendy JA, Ginty DD (1999) Mediation by a CREB family transcription factor of NGF-dependent survival of sympathetic neurons. Science 286:2358–2361
Virdee K, Parone PA, Tolkovsky AM (2000) Phosphorylation of the pro-apoptotic protein BAD on serine 155, a novel site, contributes to cell survival. Curr Biol 10:1151–1154. https://doi.org/10.1016/S0960-9822(00)00702-8
Wang L, Zhang L, Chow BKC (2017) Secretin modulates the postnatal development of mouse cerebellar cortex via PKA- and ERK-dependent pathways. Front Cell Neurosci 11. https://doi.org/10.3389/fncel.2017.00382
Wei MC, Lindsten T, Mootha VK, Weiler S, Gross A, Ashiya M, Thompson CB, Korsmeyer SJ (2000) tBID, a membrane-targeted death ligand, oligomerizes BAK to release cytochrome c. Genes Dev 14:2060–2071
Weston CR, Balmanno K, Chalmers C, Hadfield K, Molton SA, Ley R, Wagner EF, Cook SJ (2003) Activation of ERK1/2 by ΔRaf-1: ER* represses Bim expression independently of the JNK or PI3K pathways. Oncogene 22:1281–1293
Willis SN, Fletcher JI, Kaufmann T, van Delft MF, Chen L, Czabotar PE, Ierino H, Lee EF, Fairlie WD, Bouillet P, Strasser A, Kluck RM, Adams JM, Huang DCS (2007) Apoptosis initiated when BH3 ligands engage multiple Bcl-2 homologs, not Bax or Bak. Science 315:856–859
Wilson BE, Mochon E, Boxer LM (1996) Induction of bcl-2 expression by phosphorylated CREB proteins during B-cell activation and rescue from apoptosis. Mol Cell Biol 16:5546–5556
Yamaguchi Y, Miura M (2015) Programmed cell death in neurodevelopment. Dev Cell 32:478–490. https://doi.org/10.1016/j.devcel.2015.01.019
Yang J, Liu X, Bhalla K, Kim CN, Ibrado AM, Cai J, Peng TI, Jones DP, Wang X (1997) Prevention of apoptosis by Bcl-2: release of cytochrome c from mitochondria blocked. Science 275:1129–1132
Yung W-H, Leung P-S, Ng SS, Zhang J, Chan SC, Chow BK (2001) Secretin facilitates GABA transmission in the cerebellum. J Neurosci 21:7063–7068
Zanjani H, Vogel M, Delhaye-Bouchaud N, Martinou J, Mariani J (1996) Increased cerebellar Purkinje cell numbers in mice overexpressing a human Bcl-2 transgene. J Comp Neurol 374:332–341
Funding
This work is supported by Hong Kong Research Grant Council grant GRF 765113M to Professor B.K.C. Chow, and National Natural Science Foundation of China (#31500842) to Dr. L. Zhang.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
All animal experiments were conducted according to the protocols approved by Committee on the Use of Living Animals in Teaching and Research at the University of Hong Kong.
Conflict of Interest Statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Wang, L., Zhang, L. & Chow, B.K.C. Secretin Prevents Apoptosis in the Developing Cerebellum Through Bcl-2 and Bcl-xL. J Mol Neurosci 68, 494–503 (2019). https://doi.org/10.1007/s12031-019-01287-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12031-019-01287-y