Abstract
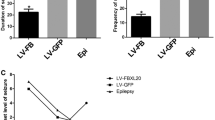
Alpha-soluble N-ethylmaleimide-sensitive factor (NSF) attachment protein (αSNAP) is a ubiquitous and indispensable component of membrane fusion machinery. There is accumulating evidence that mild alterations of αSNAP expression may be associated with specific pathological conditions in several neurological disorders. This study aimed to assess αSNAP expression in temporal lobe epilepsy (TLE) patients and pilocarpine-induced rat model and to determine whether altered αSNAP expression leads to increased susceptibility to seizures. The expression of αSNAP was assessed in the temporal lobe from patients with TLE and pilocarpine-induced epileptic rats. In addition, αSNAP expression was silenced by lentivirus pLKD-CMV-GFP-U6-NAPA (primer: GGAAGCATGCGAGATCTATGC) in animals. At day 7, the animals were kindled by pilocarpine and then the time of latency to seizure and the incidence of chronic idiopathic epilepsy seizures were assessed. The immunoreactivity to alpha-SNAP was utilized to measure expression of this protein in the animal. By immunohistochemistry, immunofluorescence, and western blotting, we found significantly lower αSNAP levels in patients with TLE. αSNAP expression showed no obvious change in pilocarpine-induced epileptic rats, from 6 h to 3 days after seizure, compared with the control group, in the acute stage; however, αSNAP levels were significantly lower in the chronic phase (day 7, months 1 and 2) in epileptic rats. Importantly, behavioral data revealed that αSNAP-small interfering RNA (siRNA) could decrease the time of latency to seizure and increase the incidence of chronic idiopathic epilepsy seizures compared with the control group. αSNAP is mainly expressed in the neuron brain tissue of patients with TLE and epileptic animals. Our findings suggest that decreasing αSNAP levels may increase epilepsy susceptibility, providing a new strategy for the treatment of this disease.




Similar content being viewed by others
References
An SJ, See MO et al (2003) Accumulation of microtubule-associated proteins in the hippocampal neurons of seizure-sensitive gerbils. Mol Cell 15(2):200–207
Andreeva AV, Kutuzov MA et al (2006) A ubiquitous membrane fusion protein alpha SNAP: a potential therapeutic target for cancer, diabetes and neurological disorders? Expert Opin Ther Targets 10(5):723–733
Burgoyne RD, Williams G (1997) NSF and SNAP are present on adrenal chromaffin granules. FEBS Lett 414(2):349–352
Cao Q, W Wang, et al (2014) Elevated expression of acid-sensing ion channel 3 inhibits epilepsy via activation of interneurons. Mol Neurobiol
Chae TH, Kim S et al (2004) The hyh mutation uncovers roles for alpha Snap in apical protein localization and control of neural cell fate. Nat Genet 36(3):264–270
Crevecoeur J, Kaminski RM et al (2014) Expression pattern of synaptic vesicle protein 2 (SV2) isoforms in patients with temporal lobe epilepsy and hippocampal sclerosis. Neuropathol Appl Neurobiol 40(2):191–204
Gassmann M, Grenacher B et al (2009) Quantifying Western blots: pitfalls of densitometry. Electrophoresis 30(11):1845–1855
Hanaya R, Hosoyama H et al (2012) Low distribution of synaptic vesicle protein 2A and synaptotagimin-1 in the cerebral cortex and hippocampus of spontaneously epileptic rats exhibiting both tonic convulsion and absence seizure. Neuroscience 221:12–20
Henderson KW, Gupta J et al (2014) Long-term seizure suppression and optogenetic analyses of synaptic connectivity in epileptic mice with hippocampal grafts of GABAergic interneurons. J Neurosci 34(40):13492–13504
Hirling H, Scheller RH (1996) Phosphorylation of synaptic vesicle proteins: modulation of the alpha SNAP interaction with the core complex. Proc Natl Acad Sci U S A 93(21):11945–11949
Hong HK, Chakravarti A et al (2004) The gene for soluble N-ethylmaleimide sensitive factor attachment protein alpha is mutated in hydrocephaly with hop gait (hyh) mice. Proc Natl Acad Sci U S A 101(6):1748–1753
Jahn R, Scheller RH (2006) SNAREs—engines for membrane fusion. Nat Rev Mol Cell Biol 7(9):631–643
Kanter-Schlifke I, Georgievska B et al (2007) Brain area, age and viral vector-specific glial cell-line-derived neurotrophic factor expression and transport in rat. Neuroreport 18(9):845–850
Kusmierczak M, Lajeunesse F et al (2014) Changes in long-range connectivity and neuronal reorganization in partial cortical deafferentation model of epileptogenesis. Neuroscience 284:153–64
Li J, Jiang G et al (2014) Altered expression of hypoxia-inducible factor-1α participates in the epileptogenesis in animal models. Synapse 68(9):402–409
Matveeva EA, Whiteheart SW et al (2001) Phosphorylation of the N-ethylmaleimide-sensitive factor is associated with depolarization-dependent neurotransmitter release from synaptosomes. J Biol Chem 276(15):12174–12181
Matveeva EA, Vanaman TC et al (2007) Asymmetric accumulation of hippocampal 7S SNARE complexes occurs regardless of kindling paradigm. Epilepsy Res 73(3):266–274
Matveeva EA, Vanaman TC et al (2008) Levetiracetam prevents kindling-induced asymmetric accumulation of hippocampal 7S SNARE complexes. Epilepsia 49(10):1749–1758
Matveeva EA, Davis VA et al (2012) Kindling-induced asymmetric accumulation of hippocampal 7S SNARE complexes correlates with enhanced glutamate release. Epilepsia 53(1):157–167
Moeller A, Zhao C et al (2012) Nucleotide-dependent conformational changes in the N-ethylmaleimide sensitive factor (NSF) and their potential role in SNARE complex disassembly. J Struct Biol 177(2):335–343
Morton AJ, Faull RL et al (2001) Abnormalities in the synaptic vesicle fusion machinery in Huntington’s disease. Brain Res Bull 56(2):111–117
Nishiki T, Nihonmatsu I et al (2001) Distribution of soluble N-ethylmaleimide fusion protein attachment proteins (SNAPs) in the rat nervous system. Neuroscience 107(3):363–371
Ohno Y, Ishihara S et al (2009) Preferential increase in the hippocampal synaptic vesicle protein 2A (SV2A) by pentylenetetrazole kindling. Biochem Biophys Res Commun 390(3):415–420
Ohno Y, Okumura T et al (2012) Kindling-associated SV2A expression in hilar GABAergic interneurons of the mouse dentate gyrus. Neurosci Lett 510(2):93–98
Puschel AW, O’Connor V et al (1994) The N-ethylmaleimide-sensitive fusion protein (NSF) is preferentially expressed in the nervous system. FEBS Lett 347(1):55–58
Racine RJ, Gartner JG et al (1972) Epileptiform activity and neural plasticity in limbic structures. Brain Res 47(1):262–268
Ramakrishnan NA, Drescher MJ et al (2012) The SNARE complex in neuronal and sensory cells. Mol Cell Neurosci 50(1):58–69
Sollner TH (2004) A mutation in the general membrane trafficking machinery and hydrocephaly. Proc Natl Acad Sci U S A 101(6):1431–1432
Steinacker P, Schwarz P et al (2005) Unchanged survival rates of 14-3-3gamma knockout mice after inoculation with pathological prion protein. Mol Cell Biol 25(4):1339–1346
Vivona S, Cipriano DJ et al (2013) Disassembly of all SNARE complexes by N-ethylmaleimide-sensitive factor (NSF) is initiated by a conserved 1:1 interaction between alpha-soluble NSF attachment protein (SNAP) and SNARE complex. J Biol Chem 288(34):24984–24991
Wang L, Shi J et al (2014) Hippocampal low-frequency stimulation increased SV2A expression and inhibited the seizure degree in pharmacoresistant amygdala-kindling epileptic rats. Epilepsy Res 108(9):1483–1491
Weber T, Zemelman BV et al (1998) SNAREpins: minimal machinery for membrane fusion. Cell 92(6):759–772
Weitzdoerfer R, M Dierssen, et al (2001) Fetal life in Down syndrome starts with normal neuronal density but impaired dendritic spines and synaptosomal structure. J Neural Transm Suppl (61): 59-70
Yin S, Guan Z et al (2005) Abnormal expression of epilepsy-related gene ERG1/NSF in the spontaneous recurrent seizure rats with spatial learning memory deficits induced by kainic acid. Brain Res 1053(1-2):195–202
Yu W, Kawasaki F et al (2011) Activity-dependent interactions of NSF and SNAP at living synapses. Mol Cell Neurosci 47(1):19–27
Acknowledgments
This work was supported by the National Natural Science Foundation of China (Nos. 81100966 and 81301110). The authors appreciate the patients and their families for their participation in this study.
Conflict of Interest
The authors declare that they have no competing interests.
Author information
Authors and Affiliations
Corresponding author
Additional information
Zhiqin Xi and Wanni Deng contributed equally to this work.
Rights and permissions
About this article
Cite this article
Xi, Z., Deng, W., Wang, L. et al. Association of Alpha-Soluble NSF Attachment Protein with Epileptic Seizure. J Mol Neurosci 57, 417–425 (2015). https://doi.org/10.1007/s12031-015-0596-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12031-015-0596-4