Abstract
Purpose
Available data on hepatocellular carcinoma (HCC) recurrence after direct-acting antivirals (DAAs) treatment for hepatitis C virus (HCV) are conflicting. No randomized trials were done. This study aims to compare the 1-year HCC recurrence rates in patients who received DAAs after tumor ablation versus those who postponed HCV treatment for 1 year.
Methods
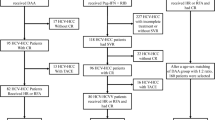
Included patients were randomized after complete HCC ablation into two groups: a postponed DAAs group for whom DAAs initiation was postponed for 12 months and a DAAs group who were given sofosbuvir/velpatasvir. Patients were followed for 1 year.
Results
Eighty-four HCV patients with a mean age of 56.35 ± 8.12 years were included; 78.57% of them were males. The number of lesions per patient ranged from 1 to 3 lesions, and the size of the largest lesion ranged from 1.5 to 5 cm. There were no statistically significant differences between both groups regarding baseline characteristics. In the DAAs group (43 patients), 11 patients had HCC recurrence, while 25 patients in the postponed DAAs group (41 patients) had HCC recurrence. Using Kaplan-Meier analysis, the 1-year recurrence-free survival (RFS) was significantly higher in the DAAs group (72.2% vs. 38%, P = 0.001). On multivariate analysis, both higher albumin levels (HR 0.147, 95% CI 0.066–0.329) and receiving DAAs (HR 0.358, 95% CI 0.176–0.730) 1 year after ablation were associated with significantly lower recurrence.
Conclusion
Direct-acting antiviral usage after complete hepatocellular carcinoma ablation significantly decreases the 1-year HCC recurrence rates, but the risk of recurrence is still not eliminated. The study registration number on clinicaltrials.gov: NCT04653818 (initial release on 28/11/2020).




Similar content being viewed by others
Data Availability
The data supporting this study’s findings are available from the corresponding author upon request.
References
Rumgay H, Arnold M, Ferlay J, et al. Global burden of primary liver cancer in 2020 and predictions to 2040. J Hepatol. 2022;77:1598–606. https://doi.org/10.1016/j.jhep.2022.08.021.
Villanueva A. Hepatocellular carcinoma. N Engl J Med. 2019;380:1450–62. https://doi.org/10.1056/NEJMra1713263.
Rashed WM, Kandeil MAM, Mahmoud MO, Ezzat S. Hepatocellular carcinoma (HCC) in Egypt: a comprehensive overview. J Egypt Natl Canc Inst. 2020;32:5. https://doi.org/10.1186/s43046-020-0016-x.
Reig M, Forner A, Rimola J, et al. BCLC strategy for prognosis prediction and treatment recommendation: the 2022 update. J Hepatol. 2022;76:681–93. https://doi.org/10.1016/j.jhep.2021.11.018.
Hsu CS, Chao YC, Lin HH, Chen DS, Kao JH. Systematic review: impact of interferon-based therapy on HCV-related hepatocellular carcinoma. Sci Rep. 2015;5:9954. https://doi.org/10.1038/srep09954.
D’Ambrosio R, Della Corte C, Colombo M. Hepatocellular carcinoma in patients with a sustained response to anti-hepatitis C therapy. Intern J Mol Sci. 2015;16:19698–712. https://doi.org/10.3390/ijms160819698.
Nishibatake Kinoshita M, Minami T, Tateishi R, et al. Impact of direct-acting antivirals on early recurrence of HCV-related HCC: comparison with interferon-based therapy. J Hepatol. 2019;70:78–86. https://doi.org/10.1016/j.jhep.2018.09.029.
Imai K, Takai K, Hanai T, Suetsugu A, Shiraki M, Shimizu M. Sustained virological response by direct-acting antivirals reduces the recurrence risk of hepatitis C-related hepatocellular carcinoma after curative treatment. Mol Clin Oncol. 2020;12:111–6. https://doi.org/10.3892/mco.2019.1956.
Reig M, Boix L, Bruix J. The impact of direct antiviral agents on the development and recurrence of hepatocellular carcinoma. Liver Intern. 2017;37:136–9. https://doi.org/10.1111/liv.13321.
Sapena V, Enea M, Torres F, et al. Hepatocellular carcinoma recurrence after direct-acting antiviral therapy: an individual patient data meta-analysis. Gut. 2022;71:593–604. https://doi.org/10.1136/gutjnl-2020-323663.
Ogawa E, Toyoda H, Iio E, et al. Hepatitis C virus cure rates are reduced in patients with active but not inactive hepatocellular carcinoma: a practice implication. Clin Infect Dis. 2020;71:2840–8. https://doi.org/10.1093/cid/ciz1160.
Mitchell DG, Bruix J, Sherman M, et al. LI-RADS (Liver Imaging Reporting and Data System): summary, discussion, and consensus of the LI-RADS management working group and future directions. Hepatology. 2015;61:1056–65. https://doi.org/10.1002/hep.27304.
Mariño Z, Darnell A, Lens S, et al. Time association between hepatitis C therapy and hepatocellular carcinoma emergence in cirrhosis: relevance of non-characterized nodules. J Hepatol. 2019;70:874–84. https://doi.org/10.1016/j.jhep.2019.01.005.
Kamal A, Elmoety AA, Rostom YA, Shater MS, Lashen SA. Percutaneous radiofrequency versus microwave ablation for management of hepatocellular carcinoma: a randomized controlled trial. J Gastrointest Oncol. 2019;10:562–71. https://doi.org/10.21037/jgo.2019.01.34.
Lencioni R, Llovet JM. Modified RECIST (mRECIST) assessment for hepatocellular carcinoma. Semin Liver Dis. 2010;30:52–60. https://doi.org/10.1055/s-0030-1247132.
Kamal A, Elsheaita A, Abdelnabi M. Association between direct-acting antiviral agents in hepatitis C virus treatment and hepatocellular carcinoma occurrence and recurrence: the endless debate. World J Clin Cases. 2022;10:1764–74. https://doi.org/10.12998/wjcc.v10.i6.1764.
Kamal A, Abd Elmoety A, Abdelmeguid Rostom Y, Said Shater M, Aldesoky Lashen S. Hepatocellular carcinoma recurrence after directly acting antivirals for chronic hepatitis C: a 2-year follow-up study. Clin Exp Hepatol. 2021;7:66–73. https://doi.org/10.5114/ceh.2021.104397.
ANRS collaborative study group on hepatocellular carcinoma. Lack of evidence of an effect of direct-acting antivirals on the recurrence of hepatocellular carcinoma: data from three ANRS cohorts. J Hepatol. 2016;65:734–40. https://doi.org/10.1016/j.jhep.2016.05.045.
Preda CM, Baicus C, Sandra I, et al. Recurrence rate of hepatocellular carcinoma in patients with treated hepatocellular carcinoma and hepatitis C virus associated cirrhosis after ombitasvir/paritaprevir/ ritonavir+dasabuvir+ribavirin therapy. United European Gastroenterol J. 2019;7:699–708. https://doi.org/10.1177/2050640619841254.
El Kassas M, Funk A, Salaheldin M, et al. Increased recurrence rates of hepatocellular carcinoma after DAA therapy in a hepatitis C-infected Egyptian cohort: a comparative analysis. J Viral Hepat. 2018;25:623–30. https://doi.org/10.1111/jvh.12854.
Chu PS, Nakamoto N, Taniki N, et al. On-treatment decrease of NKG2D correlates to early emergence of clinically evident hepatocellular carcinoma after interferon-free therapy for chronic hepatitis C. PLoS ONE. 2017;12: e0179096. https://doi.org/10.1371/journal.pone.0179096.
Debes JD, Van Tilborg M, Groothuismink ZMA, et al. Levels of cytokines in serum associate with development of hepatocellular carcinoma in patients with HCV infection treated with direct-acting antivirals. Gastroenterology. 2018;154:515–7. https://doi.org/10.1053/j.gastro.2017.10.035.
Villani R, Facciorusso A, Bellanti F, et al. DAAs rapidly reduce inflammation but increase serum VEGF level: a rationale for tumor risk during anti-HCV treatment. PLoS ONE. 2016;11: e0167934. https://doi.org/10.1371/journal.pone.0167934.
Faillaci F, Marzi L, Critelli R, et al. Liver angiopoietin-2 is a key predictor of de novo or recurrent hepatocellular cancer after hepatitis C virus direct-acting antivirals. Hepatology. 2018;68:1010–24. https://doi.org/10.1002/hep.29911.
Reig M, Cabibbo G. Antiviral therapy in the palliative setting of HCC (BCLC-B and-C). J Hepatol. 2021;4:1225–33. https://doi.org/10.1016/j.jhep.2021.01.046.
Rutledge SM, Zheng H, Li DK, Chung RT. No evidence for higher rates of hepatocellular carcinoma after direct-acting antiviral treatment: a meta-analysis. Hepatoma Res. 2019;5:31–42. https://doi.org/10.20517/2394-5079.2019.19.
Tampaki M, Savvanis S, Koskinas J. Impact of direct-acting antiviral agents on the development of Hepatocellular carcinoma: evidence and pathophysiological issues. Ann Gastroenterol. 2018;31:670–9. https://doi.org/10.20524/aog.2018.0306.
Nagaoki Y, Imamura M, Nishida Y, et al. The impact of interferon-free direct-acting antivirals on clinical outcome after curative treatment for hepatitis C virus-associated hepatocellular carcinoma: comparison with interferon-based therapy. J Med Virol. 2019;91:650–8. https://doi.org/10.1002/jmv.25352.
Cabibbo G, Celsa C, Calvaruso V, et al. Direct-acting antivirals after successful treatment of early hepatocellular carcinoma improve survival in HCV-cirrhotic patients. J Hepatol. 2019;71:265–73. https://doi.org/10.1016/j.jhep.2019.03.027.
El-Ghoneimy S, Hassan R, Abd Elbaser E, Dief E. Recurrence of hepatocellular carcinoma in hepatitis (C) virus related cirrhosis treated with direct acting antiviral therapy. Egypt J Hosp Med. 2022;88:3038–44. https://doi.org/10.21608/ejhm.2022.244534.
Wu EM, Wong LL, Hernandez BY, et al. Gender differences in hepatocellular cancer: disparities in nonalcoholic fatty liver disease/steatohepatitis and liver transplantation. Hepatoma Res. 2018;4:66–76. https://doi.org/10.20517/2394-5079.2018.87.
Reid AE, Koziel MJ, Aiza I, et al. Hepatitis C virus genotypes and viremia and hepatocellular carcinoma in the United States. Am J Gastroenterol. 1999;94:1619–26. https://doi.org/10.1111/j.1572-0241.1999.01153.x.
Akamatsu M, Yoshida H, Shiina S, et al. Neither hepatitis C virus genotype nor virus load affects survival of patients with hepatocellular carcinoma. Eur J Gastroenterol Hepatol. 2004;16:459–66. https://doi.org/10.1097/00042737-200405000-00004.
Calvaruso V, Cabibbo G, Cacciola I, et al. Incidence of hepatocellular carcinoma in patients with HCV-associated cirrhosis treated with direct-acting antiviral agents. Gastroenterology. 2018;155:411–21. https://doi.org/10.1053/j.gastro.2018.04.008.
Yamakado K, Takaki H, Nakatsuka A, et al. Radiofrequency ablation for hepatocellular carcinoma. Int J Gastrointest Interv. 2014;3:35–9. https://doi.org/10.1016/j.gii.2014.04.002.
Livraghi T, Goldberg SN, Lazzaroni S, et al. Hepatocellular carcinoma: radio-frequency ablation of medium and large lesions. Radiology. 2000;214:761–8. https://doi.org/10.1148/radiology.214.3.r00mr02761.
Funding
Self-funded.
Author information
Authors and Affiliations
Contributions
1- A.K., H.N., and M.M. designed the study with contribution of M.G., A.K., and H.O. 2- A.K., M.M., H.N., A.K, and M.G. enrolled participants while H.O. assigned participants to interventions. 3- A.K., M.M., and H.N. collected the data while A.K. and H.O. assembled it. 4- A.K., M.M. and H.O. analyzed data. 5- All authors interpreted data. 6- H.O. and A.K. wrote the article. 7- Other authors shared in the article writing. 8- All authors revised the article critically for intellectual content. 9- All authors approved the last version of the article.
Corresponding author
Ethics declarations
Ethics Approval
The study was conducted in accordance with the Declaration of Helsinki, revised in 2013, and Good Clinical Practice guidelines. It was approved by the Ethics Committee of Faculty of Medicine, Alexandria University (IRB No. 00012098).
Consent to Participate
All patients signed written informed consent prior to the study.
Consent for Publication
All authors gave their consent for publication.
Conflict of Interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Kamal, A., Metawea, M., Omar, H. et al. Hepatitis C Virus-Related One-Year Hepatocellular Carcinoma Recurrence After Directly Acting Antivirals: A Randomized Controlled Trial. J Gastrointest Canc (2024). https://doi.org/10.1007/s12029-024-01035-5
Accepted:
Published:
DOI: https://doi.org/10.1007/s12029-024-01035-5