Abstract
Purpose
The purpose of this study was to investigate whether obesity affects survival in metastatic colorectal cancer (mCRC) patients treated with bevacizumab combined with chemotherapy.
Methods
A total of 563 patients with mCRC who had received first-line chemotherapy in combination with bevacizumab were studied. Patients were grouped as obese (BMI levels > 30) or non-obese (BMI levels < 30). Progression-free survival (PFS) and overall survival (OS) were analyzed. Primary tumor location was also investigated in terms of PFS and OS.
Results
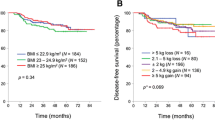
The median age of the patients was 59 years. The non-obese group had longer PFS than the obese group (P = 0.030). The 2-year survival rate of the non-obese group was also significantly higher (P = 0.036). The median PFS of non-obese patients was significantly longer in Kras wild-type patients (10.1 vs. 8.1 months, P = 0.010). Among patients with left-sided primary tumor location, median PFS and OS were significantly higher in the non-obese group (PFS non-obese, 11.5 months; obese, 8.8 months; P = 0.002) (OS non-obese, 29.4 months; obese, 21.4 months; P = 0.026).
Conclusions
Efficacy of bevacizumab may be lower in obese patients. Among patients with Kras wild-type left-sided tumors treated with bevacizumab-based regimens, the prognosis could be worse for obese patients than that for non-obese patients. There is a need for prospectively designed studies of obese patients to prove the efficacy and dosages of bevacizumab in treatment of mCRC.

Similar content being viewed by others
References
Chan AT, Giovannucci EL. Primary prevention of colorectal cancer. Gastroenterology [Internet]. 2010;138:2029–2043.e10. Available from : http://www.ncbi.nlm.nih.gov/pubmed/20420944.
Renehan AG, Tyson M, Egger M, Heller RF, Zwahlen M. Body-mass index and incidence of cancer: a systematic review and meta-analysis of prospective observational studies. Lancet [Internet]. 2008;371(9612):569–578. Available from: http://www.ncbi.nlm.nih.gov/pubmed/18280327. https://doi.org/10.1016/S0140-6736(08)60269-X.
Miyazawa-Hoshimoto S, Takahashi K, Bujo H, Hashimoto N, Saito Y. Elevated serum vascular endothelial growth factor is associated with visceral fat accumulation in human obese subjects. Diabetologia [Internet]. 2003;46(11):1483–1488. Available from. https://doi.org/10.1007/s00125-003-1221-6.
Silha JV, Krsek M, Sucharda P, Murphy LJ. Angiogenic factors are elevated in overweight and obese individuals. Int J Obes. (Lond). [Internet]. 2005. Available from http://www.ncbi.nlm.nih.gov/pubmed/15953938;29(11):1308–14. https://doi.org/10.1038/sj.ijo.0802987.
Parekh N, Chandran U, Bandera EV. Obesity in cancer survival. Annu. Rev. Nutr. [Internet]. 2012 Available from: http://www.ncbi.nlm.nih.gov/pubmed/22540252;32(1):311–42. https://doi.org/10.1146/annurev-nutr-071811-150713.
Hoffmeister M, Bläker H, Kloor M, Roth W, Toth C, Herpel E, et al. Body mass index and microsatellite instability in colorectal cancer: a population-based study. Cancer Epidemiol. Biomarkers Prev. [Internet]. 2013. Available from: http://www.ncbi.nlm.nih.gov/pubmed/24127414;22(12):2303–11. https://doi.org/10.1158/1055-9965.EPI-13-0239.
Krämer I, Lipp H-P. Bevacizumab, a humanized anti-angiogenic monoclonal antibody for the treatment of colorectal cancer. J. Clin. Pharm. Ther. [Internet]. 2007. Available from: http://www.ncbi.nlm.nih.gov/pubmed/17286784;32(1):1–14. https://doi.org/10.1111/j.1365-2710.2007.00800.x.
Ferrara N, Hillan KJ, Gerber H-P, Novotny W. Discovery and development of bevacizumab, an anti-VEGF antibody for treating cancer. Nat. Rev. Drug Discov. [Internet]. 2004;3:391–400. Available from: http://www.ncbi.nlm.nih.gov/pubmed/15136787.
Tokunaga K, Matsuzawa Y, Ishikawa K, Tarui S. A novel technique for the determination of body fat by computed tomography. Int. J. Obes. [Internet]. 1983;7:437–45. Available from: http://www.ncbi.nlm.nih.gov/pubmed/6642855.
Guiu B, Petit JM, Bonnetain F, Ladoire S, Guiu S, Cercueil J-P, et al. Visceral fat area is an independent predictive biomarker of outcome after first-line bevacizumab-based treatment in metastatic colorectal cancer. Gut [Internet]. 2010. Available from: http://www.ncbi.nlm.nih.gov/pubmed/19837679;59(3):341–7. https://doi.org/10.1136/gut.2009.188946.
Brändstedt J, Wangefjord S, Nodin B, Eberhard J, Sundström M, Manjer J, et al. Associations of anthropometric factors with KRAS and BRAF mutation status of primary colorectal cancer in men and women: a cohort study. PLoS One [Internet]. 2014;9(6):e98964. Available from: http://www.ncbi.nlm.nih.gov/pubmed/24918610. https://doi.org/10.1371/journal.pone.0098964.
Fedirko V, Romieu I, Aleksandrova K, Pischon T, Trichopoulos D, Peeters PH, et al. Pre-diagnostic anthropometry and survival after colorectal cancer diagnosis in Western European populations. Int. J. cancer [Internet]. 2014. Available from: http://www.ncbi.nlm.nih.gov/pubmed/24623514;135(8):1949–60. https://doi.org/10.1002/ijc.28841.
Venook AP, Niedzwiecki D, Innocenti F, Fruth B, Greene C, O’Neil BH, et al. Impact of primary (1o) tumor location on overall survival (OS) and progression-free survival (PFS) in patients (pts) with metastatic colorectal cancer (mCRC): analysis of CALGB/SWOG 80405 (Alliance). J. Clin. Oncol. 2016;34(15_suppl):3504–3504
Makey KL, Patterson SG, Robinson J, Loftin M, Waddell DE, Miele L, et al. Increased plasma levels of soluble vascular endothelial growth factor receptor 1 (sFlt-1) in women by moderate exercise and increased plasma levels of vascular endothelial growth factor in overweight/obese women. Eur J Cancer Prev [Internet]. 2013. Available from: http://www.ncbi.nlm.nih.gov/pubmed/22609636;22(1):83–9. https://doi.org/10.1097/CEJ.0b013e328353ed81.
Faruk Aykan N, Yildiz I, Sen F, Kilic L, Keskin S, Ciftci R, et al. Effect of increased body mass index (BMI) on time to tumour progression (TTP) in unresectable metastatic colorectal cancer (mCRC) patients treated with bevacizumab-based therapy. Med Oncol [Internet]. 2013. Available from: http://www.ncbi.nlm.nih.gov/pubmed/23921648;30(3):679. https://doi.org/10.1007/s12032-013-0679-4.
Koopman M, Antonini NF, Douma J, Wals J, Honkoop AH, Erdkamp FLG, et al. Sequential versus combination chemotherapy with capecitabine, irinotecan, and oxaliplatin in advanced colorectal cancer (CAIRO): a phase III randomised controlled trial. Lancet (London, England) [Internet]. 2007. Available from: http://www.ncbi.nlm.nih.gov/pubmed/17630036;370(9582):135–42. https://doi.org/10.1016/S0140-6736(07)61086-1.
Kaidar-Person O, Badarna H, Bar-Sela G. Bevacizumab for metastatic colon cancer: does patient BMI influence survival? Anticancer Drugs [Internet]. 2015. Available from: http://www.ncbi.nlm.nih.gov/pubmed/25563769;26(3):363–6. https://doi.org/10.1097/CAD.0000000000000201.
Patel GS, Ullah S, Beeke C, Hakendorf P, Padbury R, Price TJ, et al. Association of BMI with overall survival in patients with mCRC who received chemotherapy versus EGFR and VEGF-targeted therapies. Cancer Med [Internet]. 2015. Available from: http://www.ncbi.nlm.nih.gov/pubmed/26211512;4(10):1461–71. https://doi.org/10.1002/cam4.490.
Renfro LA, Loupakis F, Adams RA, Seymour MT, Heinemann V, Schmoll H-J, et al. Body mass index is prognostic in metastatic colorectal cancer: pooled analysis of patients from first-line clinical trials in the ARCAD database. J Clin Oncol [Internet]. 2016. Available from: http://www.ncbi.nlm.nih.gov/pubmed/26503203;34(2):144–50. https://doi.org/10.1200/JCO.2015.61.6441.
Cao Y. Angiogenesis modulates adipogenesis and obesity. J Clin Invest [Internet]. 2007;117:2362–8. Available from: http://www.pubmedcentral.nih.gov/articlerender.fcgi?artid=1963348&tool=pmcentrez&rendertype=abstract.
Berg A, Hoivik EA, Mjøs S, Holst F, Werner HMJ, Tangen IL, et al. Molecular profiling of endometrial carcinoma precursor, primary and metastatic lesions suggests different targets for treatment in obese compared to non-obese patients. Oncotarget [Internet]. 2015. Available from: http://www.ncbi.nlm.nih.gov/pubmed/25415225;6(2):1327–39. https://doi.org/10.18632/oncotarget.2675.
Bordonaro M, Lazarova D. Hypothesis: obesity is associated with a lower mutation threshold in colon cancer. J Cancer [Internet]. 2015. Available from: http://www.ncbi.nlm.nih.gov/pubmed/26284133;6(9):825–31. https://doi.org/10.7150/jca.12352.
Simkens LHJ, Koopman M, Mol L, Veldhuis GJ, Ten Bokkel HD, Muller EW, et al. Influence of body mass index on outcome in advanced colorectal cancer patients receiving chemotherapy with or without targeted therapy. Eur J Cancer [Internet]. 2011. Available from: http://www.ncbi.nlm.nih.gov/pubmed/21803570;47(17):2560–7. https://doi.org/10.1016/j.ejca.2011.06.038.
Tol J, Koopman M, Rodenburg CJ, Cats A, Creemers GJ, Schrama JG, et al. A randomised phase III study on capecitabine, oxaliplatin and bevacizumab with or without cetuximab in first-line advanced colorectal cancer, the CAIRO2 study of the Dutch Colorectal Cancer Group (DCCG). An interim analysis of toxicity. Ann Oncol [Internet]. 2008;19:734–8. Available from: http://annonc.oxfordjournals.org/content/early/2008/02/13/annonc.mdm607.full.
Ladoire S, Bonnetain F, Gauthier M, Zanetta S, Petit JM, Guiu S, et al. Visceral fat area as a new independent predictive factor of survival in patients with metastatic renal cell carcinoma treated with antiangiogenic agents. Oncologist [Internet]. 2011;16:71–81. Available from: http://www.pubmedcentral.nih.gov/articlerender.fcgi?artid=3228050&tool=pmcentrez&rendertype=abstract.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The study was approved by the ethics committee at Necmettin Erbakan University’s Meram Faculty of Medicine and carried out in accordance with Declaration of Helsinki principles and all applicable regulations.
Conflict of Interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Artaç, M., Korkmaz, L., Coşkun, H.Ş. et al. Bevacuzimab May Be Less Effective in Obese Metastatic Colorectal Cancer Patients. J Gastrointest Canc 50, 214–220 (2019). https://doi.org/10.1007/s12029-017-0047-2
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12029-017-0047-2