Abstract
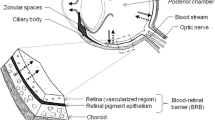
Vitreous humour (VH) is routinely collected for toxicological analyses. However, only a few studies have examined its possible use in genotyping. Because of its isolation from the rest of the body, later onset of postmortem changes, and availability, VH could be a potential source of DNA for postmortem identification and other molecular analyses in forensic genetics. During forensic autopsies, samples of different VH volumes (5 ml, 4 ml, 3 ml, 2 ml, 1 ml, and 0.5 ml) were taken from 66 cadavers 24–48 h after death. DNA was also isolated from the samples immediately after collection and after a specific time of storage (1, 2, and 6 months). DNA was isolated using phenol‒chloroform‒isoamyl alcohol (PCI), and the yield and purity of the obtained DNA were determined spectrophotometrically using a FastGene NanoView Photometer. The integrity of the isolated DNA molecule was determined by PCR amplification of the hTERT (113 bp) gene. The results showed that VH could be a reliable source of genetic material for forensic analysis, and the method used for DNA extraction was effective. The yield of the isolated DNA ranged from 6.20 to 609.5 ng/µl, and the purity of the samples was 1.24–2.34. The isolated DNA concentration and integrity depend on the sample volume, but the DNA purity does not. It is also shown that DNA can be extracted from VH samples that have been stored for up to 6 months at − 20 °C. Therefore, using VH can be a valuable material for DNA identification.




Similar content being viewed by others
References
Sołtyszewski I, Niemcunowicz-Janica A, Pepiński W, Spólnicka M, Zbiec R, Janica J. Vitreous humour as a potential DNA source for postmortem human identification. Folia Histochem Cytobiol. 2007;45:135–6.
Markowska J, Szopa M, Zawadzki M, Piekoszewski W. Ciało szkliste oka – rutynowy czy alternatywny materiał do badań w medycynie sądowej. Arch Med Sadowej Kryminol. 2017;67:201–13.
Thierauf A, Musshoff F, Madea B. Post-mortem biochemical investigations of vitreous humor. Forensic Sci Int. 2009;192:78–82.
Pigaiani N, Bertaso A, De Palo EF, Bortolotti F, Tagliaro F. Vitreous humor endogenous compounds analysis for post-mortem forensic investigation. Forensic Sci Int. 2020;310:110235. Available from: https://doi.org/10.1016/j.forsciint.2020.110235.
Calacal GC, Apaga DLT, Salvador JM, Jimenez JAD, Lagat LJ, Villacorta RPF, et al. Comparing different post-mortem human samples as DNA sources for downstream genotyping and identification. Forensic Sci Int Genet. 2015;19:212–20. Available from: https://doi.org/10.1016/j.fsigen.2015.07.017.
de Campos EG, da Costa BRB, dos Santos FS, Monedeiro F, Alves MNR, Santos Junior WJR, et al. Alternative matrices in forensic toxicology: a critical review. Forensic Toxicol. 2022;40:1–18. Available from: https://doi.org/10.1007/s11419-021-00596-5.
Metushi IG, Fitzgerald RL, McIntyre IM. Assessment and comparison of vitreous humor as an alternative matrix for forensic toxicology screening by GC-MS. J Anal Toxicol. 2016;40:243–7.
Lee HC, Ladd C. Preservation and collection of biological evidence. Croat Med J. 2001;42:225–8.
Ziętkiewicz E, Witt M, Daca P, Żebracka-Gala J, Goniewicz M, Jarząb B, et al. Current genetic methodologies in the identification of disaster victims and in forensic analysis. J Appl Genet. 2012;53:41–60.
Bulla A, De Witt B, Ammerlaan W, Betsou F, Lescuyer P. Blood DNA yield but not integrity or methylation is impacted after long-term storage. Biopreserv Biobank. 2016;14:29–38.
Bukyya JL, Tejasvi MLA, Avinash A, P CH, Talwade P, Afroz MM, et al. DNA profiling in forensic science: a review. Glob Med Genet. 2021;8:135–43.
Vitoševic K, Todorovic D, Slovic Z, Zivkovic-Zaric R, Todorovic M. Forensic genetics and genotyping. Serbian J Exp Clin Res. 2016;20:75–86.
Kirstahler P, Bjerrum SS, Friis-Møller A, La Cour M, Aarestrup FM, Westh H, et al. Genomics-based identification of microorganisms in human ocular body fluid. Sci Rep. 2018;8:1–14.
Mazoteras P, Bispo PJM, Höfling-Lima AL, Casaroli-Marano RP. DNA extraction methods for panbacterial and panfungal PCR detection in intraocular fluids. Curr Eye Res. 2015;40:697–706.
Jun K Il, Oh BL, Kim N, Shin JY, Moon J. Microbial diagnosis of endophthalmitis using nanopore amplicon sequencing. Int J Med Microbiol. 2021;311:151505. Available from: https://doi.org/10.1016/j.ijmm.2021.151505.
Varghese B, Rodrigues C, Deshmukh M, Natarajan S, Kamdar P, Mehta A. Broad-range bacterial and fungal DNA amplification on vitreous humor from suspected endophthalmitis patients. Mol Diagnosis Ther. 2006;10:319–26.
Bispo PJM, de Melo GB, Hofling-Lima AL, Pignatari ACC. Detection and gram discrimination of bacterial pathogens from aqueous and vitreous humor using real-time PCR assays. Investig Ophthalmol Vis Sci. 2011;52:873–81.
Salerno M, Cocimano G, Roccuzzo S, Russo I, Piombino-Mascali D, Márquez-Grant N, et al. New trends in immunohistochemical methods to estimate the time since death: a review. Diagnostics. 2022;12:1–18.
Diepenbroek M, Bayer B, Anslinger K. Pushing the boundaries: forensic dna phenotyping challenged by single-cell sequencing. Genes (Basel). 2021;12.
Perry J, Munshi T, Haizel T, Iyavoo S. Validation of reduced volume VeriFiler™ Express PCR Amplification Kit for buccal swab samples extracted using Prep-n-Go™ Buffer. J Forensic Sci. 2022;67:1971–8.
Butler JM. Short tandem repeat typing technologies used in human identity testing. Biotechniques. 2007;43.
Pontes ML, Fondevila M, Laréu MV, Medeiros R. SNP Markers as additional information to resolve complex kinship cases. Transfus Med Hemotherapy. 2015;42:385–8.
Hirschhorn JW, Snider JS, Lindsey KG, Schandl CA. Molecular profiling of vitreous fluid by massively parallel sequencing: a case series. J Am Soc Cytopathol. 2020;9:254–7.
Sosa C, Abecia E, Casalod Y, Baeta M, Núñez C, Luna A, et al. A preliminary study on the incidence of heteroplasmy in mitochondrial DNA from vitreous humour. Leg Med. 2009;11:S460–2. Available from: https://doi.org/10.1016/j.legalmed.2009.01.087.
Acknowledgements
The authors would like to thank the Faculty of Medical Sciences, University of Kragujevac and the Ministry of Science, Technological Development and Innovation of the Republic of Serbia (No 451-03-47/2023-01/200111).
Funding
This study was supported by grant (project JP:05/13) from the Faculty of Medical Sciences, University of Kragujevac, Serbia and by the Ministry of Science, Technological Development and Innovation of the Republic of Serbia (No 451-03-47/2023-01/200111).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethics approval
Institutional ethical approval was obtained in compliance with the Declaration of Helsinki. Authorization to use human biological samples for research was obtained. The Ethics Committee of the University of Kragujevac, Faculty of Medical Sciences, the Ethic Committee of University Clinical Centre of Kragujevac (No. 01–2798), Appeal Public Prosecutor’s Office from Kragujevac (A No. 79/13) and Higher Court in Kragujevac (SU-VIII-110/13) agreed to these investigations.
Competing interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Čanović, V., Slović, Ž., Todorović, D. et al. The use of vitreous humour as a potential source of DNA for postmortem identification in forensic science. Forensic Sci Med Pathol (2024). https://doi.org/10.1007/s12024-023-00760-w
Accepted:
Published:
DOI: https://doi.org/10.1007/s12024-023-00760-w