Abstract
Purpose
Cardiac insufficiency is a major morbidity in neonatal hyperthyroidism. It is important to assess the hemodynamics in neonates born to mothers with Graves’ disease (GD). This study prospectively evaluated the hemodynamic changes in neonates born to mothers with GD.
Methods
Overall, 80 newborns were enrolled. Thirty-six neonates were born to mothers with GD who were positive for thyroid-stimulating hormone (TSH) receptor antibody (TRAb), and 44 were born to mother negative for TRAb. The serum levels of TSH, free triiodothyronine (FT3), free thyroxine (FT4), and N-terminal-pro-B-type natriuretic peptide (NT-proBNP), the cardiac output, and cardiac index (CI) evaluated by echocardiography were compared between the two groups at several postnatal points (day of delivery and 5, 10, and 30 days of life).
Results
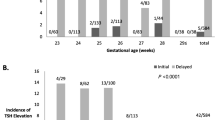
The TRAb-positive newborns had higher FT4 levels and CI on Day 5 (both p < 0.05) and higher FT3 (p < 0.05) and FT4 levels (p < 0.01) and CI (p < 0.01) but lower TSH levels (p < 0.05) on Day 10 than the TRAb-negative newborns. The TRAb-positive newborns had significantly higher NT-proBNP levels on Days 5 (median 752 vs. 563 pg/mL, p = 0.034) and 10 (median 789 vs. 552 pg/mL, p = 0.002) than the TRAb-negative newborns.
Conclusions
Hemodynamic changes in neonates born to TRAb-positive mothers with GD resulted in a higher CI and NT-proBNP levels than in those with TRAb-negative mothers from postnatal days 5 to 10.

Similar content being viewed by others
Availability of data and material
The datasets during and/or analyzed during the current study available from the corresponding author on reasonable request.
References
P. Sunshine, H. Kusumoto, J.P. Kriss, Survival time of circulating long-acting thyroid stimulator in neonatal thyrotoxicosis: implications for diagnosis and therapy of the disorder. Pediatrics 36(6), 869–876 (1965)
J.M. McKenzie, M. Zakarija, Fetal and neonatal hyperthyroidism and hypothyroidism due to maternal TSH receptor antibodies. Thyroid 2(2), 155–159 (1992). https://doi.org/10.1089/thy.1992.2.155
P. Laurberg, C. Bournaud, J. Karmisholt, J. Orgiazzi, Management of Graves’ hyperthyroidism in pregnancy: focus on both maternal and foetal thyroid function, and caution against surgical thyroidectomy in pregnancy. Eur. J. Endocrinol. 160(1), 1–8 (2009). https://doi.org/10.1530/eje-08-0663
D. Zimmerman, Fetal and neonatal hyperthyroidism. Thyroid 9(7), 727–733 (1999). https://doi.org/10.1089/thy.1999.9.727
M. Polak, Hyperthyroidism in early infancy: pathogenesis, clinical features and diagnosis with a focus on neonatal hyperthyroidism. Thyroid 8(12), 1171–1177 (1998). https://doi.org/10.1089/thy.1998.8.1171
G. Radetti, A. Zavallone, L. Gentili, P. Beck-Peccoz, G. Bona, Foetal and neonatal thyroid disorders. Minerva Pediatr. 54(5), 383–400 (2002)
E.S. Lightner, H.D. Allen, G. Loughlin, Neonatal hyperthyroidism and heart failure. A differential approach. Am. J. Dis. Child. 131(1), 68–70 (1977). https://doi.org/10.1001/archpedi.1977.02120140070012
Y.L. Bussmann, M.L. Tillman, A.S. Pagliara, Neonatal thyrotoxicosis associated with the hyperviscosity syndrome. J. Pediatr. 90(2), 266–268 (1977). https://doi.org/10.1016/s0022-3476(77)80648-3
D. O’Donovan, C. McMahon, C. Costigan, P. Oslizlok, D. Duff, Reversible pulmonary hypertension in neonatal Graves disease. Ir. Med. J. 90(4), 147–148 (1997)
A.A. Zuppa, P. Sindico, I. Savarese, V. D’Andrea, A. Fracchiolla, F. Cota, C. Romagnoli, Neonatal hyperthyroidism: neonatal clinical course of two brothers born to a mother with Graves-Basedow disease, before and after total thyroidectomy. J. Pediatr. Endocrinol. Metab. 20(4), 535–539 (2007). https://doi.org/10.1515/jpem.2007.20.4.535
L.Q. Gu, L. Zhao, W. Zhu, F.Y. Li, M.J. Zhang, Y. Liu, J.M. Liu, G. Ning, Y.J. Zhao, Relationships between serum levels of thyroid hormones and serum concentrations of asymmetric dimethylarginine (ADMA) and N-terminal-pro-B-type natriuretic peptide (NT-proBNP) in patients with Graves’ disease. Endocrine. 39(3), 266–271 (2011). https://doi.org/10.1007/s12020-011-9436-7
S. Arikan, A. Tuzcu, D. Gokalp, M. Bahceci, R. Danis, Hyperthyroidism may affect serum N-terminal pro-B-type natriuretic peptide levels independently of cardiac dysfunction. Clin. Endocrinol. 67(2), 202–207 (2007). https://doi.org/10.1111/j.1365-2265.2007.02861.x
B. Ozmen, D. Ozmen, Z. Parildar, I. Mutaf, O. Bayindir, Serum N-terminal-pro-B-type natriuretic peptide (NT-pro-BNP) levels in hyperthyroidism and hypothyroidism. Endocr. Res. 32(1-2), 1–8 (2007). https://doi.org/10.1080/07435800701670047
C. Massart, J. Orgiazzi, D. Maugendre, Clinical validity of a new commercial method for detection of TSH-receptor binding antibodies in sera from patients with Graves’ disease treated with antithyroid drugs. Clin. Chim. Acta. 304(1–2), 39–47 (2001). https://doi.org/10.1016/s0009-8981(00)00385-5
A.P. Weetman, Graves’ disease. N. Engl. J. Med. 343(17), 1236–1248 (2000). https://doi.org/10.1056/nejm200010263431707
E. Lechner, G. Wiesinger-Eidenberger, O. Wagner, M. Weissensteiner, E. Schreier-Lechner, D. Leibetseder, W. Arzt, G. Tulzer, Amino terminal pro B-type natriuretic peptide levels are elevated in the cord blood of neonates with congenital heart defect. Pediatr. Res. 66(4), 466–469 (2009). https://doi.org/10.1203/PDR.0b013e3181b3aee4
I. Farombi-Oghuvbu, T. Matthews, P.D. Mayne, H. Guerin, J.D. Corcoran, N-terminal pro-B-type natriuretic peptide: a measure of significant patent ductus arteriosus. Arch. Dis. Child. Fetal. Neonatal. Ed. 93(4), F257–F260 (2008). https://doi.org/10.1136/adc.2007.120691
E.W. Reynolds, J.G. Ellington, M. Vranicar, H.S. Bada, Brain-type natriuretic peptide in the diagnosis and management of persistent pulmonary hypertension of the newborn. Pediatrics 114(5), 1297–1304 (2004). https://doi.org/10.1542/peds.2004-0525
van der Kaay D. C., Wasserman J. D., Palmert M. R. Management of neonates born to mothers with Graves’ disease. Pediatrics 137(4) (2016). https://doi.org/10.1542/peds.2015-1878
T. Tajima, W. Jo, K. Fujikura, M. Fukushi, K. Fujieda, Elevated free thyroxine levels detected by a neonatal screening system. Pediatr. Res. 66(3), 312–316 (2009). https://doi.org/10.1203/PDR.0b013e3181b1bcbd
L. Schwachtgen, M. Herrmann, T. Georg, P. Schwarz, N. Marx, A. Lindinger, Reference values of NT-proBNP serum concentrations in the umbilical cord blood and in healthy neonates and children. Z. Kardiol. 94(6), 399–404 (2005). https://doi.org/10.1007/s00392-005-0246-x
N. Mitsuda, H. Tamaki, N. Amino, T. Hosono, K. Miyai, O. Tanizawa, Risk factors for developmental disorders in infants born to women with Graves disease. Obstet. Gynecol. 80(3 Pt 1), 359–364 (1992)
M.J. Kempers, D.A. van Tijn, A.S. van Trotsenburg, J.J. de Vijlder, B.M. Wiedijk, T. Vulsma, Central congenital hypothyroidism due to gestational hyperthyroidism: detection where prevention failed. J. Clin. Endocrinol. Metab. 88(12), 5851–5857 (2003). https://doi.org/10.1210/jc.2003-030665
L.E. Teichholz, T. Kreulen, M.V. Herman, R. Gorlin, Problems in echocardiographic volume determinations: echocardiographic-angiographic correlations in the presence of absence of asynergy. Am. J. Cardiol. 37(1), 7–11 (1976). https://doi.org/10.1016/0002-9149(76)90491-4
M. Zakarija, J.M. McKenzie, W.H. Hoffman, Prediction and therapy of intrauterine and late-onset neonatal hyperthyroidism. J. Clin. Endocrinol. Metab. 62(2), 368–371 (1986). https://doi.org/10.1210/jcem-62-2-368
M. Schultz, C. Kistorp, B. Langdahl, I. Raymond, P. Hildebrandt, J. Faber, N-terminal-pro-B-type natriuretic peptide in acute hyperthyroidism. Thyroid 17(3), 237–241 (2007). https://doi.org/10.1089/thy.2006.0258
J. Faber, N. Wiinberg, S. Schifter, J. Mehlsen, Haemodynamic changes following treatment of subclinical and overt hyperthyroidism. Eur. J. Endocrinol. 145(4), 391–396 (2001). https://doi.org/10.1530/eje.0.1450391
I. Klein, K. Ojamaa, Thyroid hormone and the cardiovascular system. N. Engl. J. Med. 344(7), 501–509 (2001). https://doi.org/10.1056/nejm200102153440707
J.A. Franklyn, K. Boelaert, Thyrotoxicosis. Lancet 379(9821), 1155–1166 (2012). https://doi.org/10.1016/s0140-6736(11)60782-4
F.D. Hobbs, R.C. Davis, A.K. Roalfe, R. Hare, M.K. Davies, J.E. Kenkre, Reliability of N-terminal pro-brain natriuretic peptide assay in diagnosis of heart failure: cohort study in representative and high risk community populations. BMJ 324(7352), 1498 (2002). https://doi.org/10.1136/bmj.324.7352.1498
R.S. Gardner, F. Ozalp, A.J. Murday, S.D. Robb, T.A. McDonagh, N-terminal pro-brain natriuretic peptide. A new gold standard in predicting mortality in patients with advanced heart failure. Eur. Heart J. 24(19), 1735–1743 (2003). https://doi.org/10.1016/j.ehj.2003.07.005
L.C. Costello-Boerrigter, G. Boerrigter, M.M. Redfield, R.J. Rodeheffer, L.H. Urban, D.W. Mahoney, S.J. Jacobsen, D.M. Heublein, J.C. Burnett Jr, Amino-terminal pro-B-type natriuretic peptide and B-type natriuretic peptide in the general community: determinants and detection of left ventricular dysfunction. J. Am. Coll. Cardiol. 47(2), 345–353 (2006). https://doi.org/10.1016/j.jacc.2005.09.025
M. Schultz, J. Faber, C. Kistorp, A. Jarlov, F. Pedersen, N. Wiinberg, P. Hildebrandt, N-terminal-pro-B-type natriuretic peptide (NT-pro-BNP) in different thyroid function states. Clin. Endocrinol. 60(1), 54–59 (2004). https://doi.org/10.1111/j.1365-2265.2004.01941.x
M. Kohno, T. Horio, K. Yasunari, K. Yokokawa, M. Ikeda, N. Kurihara, Y. Nishizawa, H. Morii, T. Takeda, Stimulation of brain natriuretic peptide release from the heart by thyroid hormone. Metabolism 42(8), 1059–1064 (1993). https://doi.org/10.1016/0026-0495(93)90023-h
T.S. Mir, R. Laux, H.H. Hellwege, B. Liedke, C. Heinze, H. von Buelow, S. Laer, J. Weil, Plasma concentrations of aminoterminal pro atrial natriuretic peptide and aminoterminal pro brain natriuretic peptide in healthy neonates: marked and rapid increase after birth. Pediatrics 112(4), 896–899 (2003). https://doi.org/10.1542/peds.112.4.896
B. Biondi, E.A. Palmieri, G. Lombardi, S. Fazio, Effects of subclinical thyroid dysfunction on the heart. Ann. Intern. Med. 137(11), 904–914 (2002). https://doi.org/10.7326/0003-4819-137-11-200212030-00011
M. Tadic, S. Ilic, C. Cuspidi, T. Marjanovic, V. Celic, Subclinical hyperthyroidism impacts left ventricular deformation: 2D and 3D echocardiographic study. Scand. Cardiovasc. J. 49(2), 74–81 (2015). https://doi.org/10.3109/14017431.2015.1015441
Acknowledgements
The authors thank Dr. Masaki Yamamoto, Department of Pediatrics, Seirei Hamamatsu General Hospital, Dr. Shinichi Nakashima, JA Shizuoka Kohseiren Enhu Hospital, and Dr. Jiro Kagawa, Fujieda Municipal General Hospital, for their valuable assistance.
Author contributions
TI designed and conducted the research, conducted the statistical analyses, interpreted the data, and drafted the initial manuscript. HU, SI, TB, and AO made substantial contributions to the acquisition of data. SI and HI contributed to the study design and provided comments on the manuscript. All authors revised and approved the final version.
Funding
This work was supported by JSPS KAKENHI Grant Number JP18K07787.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Consent to participate
Informed consent was obtained from the parents of each newborn.
Ethics approval
The study was conducted in accordance with the ethical principles described in the Declaration of Helsinki and was approved by the local ethics committees.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Ishikawa, T., Uchiyama, H., Iwashima, S. et al. Hemodynamic changes in neonates born to mothers with Graves’ disease. Endocrine 72, 171–178 (2021). https://doi.org/10.1007/s12020-020-02443-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12020-020-02443-w