Abstract
Background
Pseudohypoparathyroidism (PHP) is a rare disorder characterized by hypocalcemia, hyperphosphatemia, and resistance to parathyroid hormone (PTH). According to different GNAS mutations, PHP is divided into several subtypes, among which autosomal-dominant PHP1B (AD-PHP1B) is caused by STX16 deletion and epigenetic alteration of GNAS. Although the deletion of STX16 exons 2−6 is commonly observed, other mutations involving STX16 can also result in AD-PHP1B.
Materials and methods
The clinical information of a 38-year-old male PHP patient was collected. The genomic DNA from peripheral blood cells was extracted for genetic analysis of GNAS and upstream STX16 by methylation-specific multiplex ligation-dependent probe amplification (MS-MLPA) and whole-exome sequencing (WES). Sanger sequencing was performed to verify the break point of the novel long-range deletion.
Results
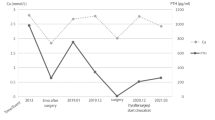
The patient’s medical history of tetany and seizure as well as laboratory examination showing hypocalcemia and elevated PTH levels indicated the diagnosis of PHP. The results of MS-MLPA showed loss of methylation of GNAS A/B:TSS-DMR and half-reduced copy number of STX16 exon 1−9, which revealed the subtype of AD-PHP1B. Furthermore, the WES study displayed a 87.5 kb missing upstream of GNAS. A 87.5 kb deletion spanning STX16 and NPEPL1 together with an insertion of 28 bp of unknown origin was verified by PCR along with Sanger sequencing.
Conclusions
A novel deletion of 87.5 kb spanning STX16 and NPEPL1 was discovered in an AD-PHP1B patient, which provides new information on molecular defects leading to AD-PHP1B.




Similar content being viewed by others
References
G. Mantovani, M. Bastepe, D. Monk, L. de Sanctis, S. Thiele, A. Usardi et al., Diagnosis and management of pseudohypoparathyroidism and related disorders: first international Consensus Statement. Nat. Rev. Endocrinol. 14(8), 476–500 (2018). https://doi.org/10.1038/s41574-018-0042-0
A. Nakamura, E. Hamaguchi, R. Horikawa, Y. Nishimura, K. Matsubara, S. Sano et al., Complex genomic rearrangement within the GNAS region associated with familial pseudohypoparathyroidism type 1b. J. Clin. Endocrinol. Metab. 101(7), 2623–2627 (2016). https://doi.org/10.1210/jc.2016-1725
L. Akin, S. Kurtoglu, A. Yildiz, M.A. Akin, M. Kendirici, Vitamin D deficiency rickets mimicking pseudohypoparathyroidism. J. Clin. Res. Pediatr. Endocrinol. 2(4), 173–175 (2010). https://doi.org/10.4274/jcrpe.v2i4.173
M.A. Levine, An update on the clinical and molecular characteristics of pseudohypoparathyroidism. Curr. Opin. Endocrinol. Diabetes Obes. 19(6), 443–451 (2012). https://doi.org/10.1097/MED.0b013e32835a255c
F.M. Elli, L. de Sanctis, E. Peverelli, P. Bordogna, B. Pivetta, G. Miolo et al., Autosomal dominant pseudohypoparathyroidism type Ib: a novel inherited deletion ablating STX16 causes loss of imprinting at the A/B DMR. J. Clin. Endocrinol. Metab. 99(4), E724–E728 (2014). https://doi.org/10.1210/jc.2013-3704
M. Bastepe, J.E. Pincus, T. Sugimoto, K. Tojo, M. Kanatani, Y. Azuma et al., Positional dissociation between the genetic mutation responsible for pseudohypoparathyroidism type Ib and the associated methylation defect at exon A/B: evidence for a long-range regulatory element within the imprinted GNAS1 locus. Hum. Mol. Genet. 10(12), 1231–1241 (2001). https://doi.org/10.1093/hmg/10.12.1231
N. Richard, G. Abeguile, N. Coudray, H. Mittre, N. Gruchy, J. Andrieux et al., A new deletion ablating NESP55 causes loss of maternal imprint of A/B GNAS and autosomal dominant pseudohypoparathyroidism type Ib. J. Clin. Endocrinol. Metab. 97(5), E863–E867 (2012). https://doi.org/10.1210/jc.2011-2804
A. Linglart, R.C. Gensure, R.C. Olney, H. Juppner, M. Bastepe, A novel STX16 deletion in autosomal dominant pseudohypoparathyroidism type Ib redefines the boundaries of a cis-acting imprinting control element of GNAS. Am. J. Hum. Genet. 76(5), 804–814 (2005). https://doi.org/10.1086/429932
M. Bastepe, L.F. Frohlich, A. Linglart, H.S. Abu-Zahra, K. Tojo, L.M. Ward et al., Deletion of the NESP55 differentially methylated region causes loss of maternal GNAS imprints and pseudohypoparathyroidism type Ib. Nat. Genet. 37(1), 25–27 (2005). https://doi.org/10.1038/ng1487
T. Eggermann, G. Perez de Nanclares, E.R. Maher, I.K. Temple, Z. Tumer, D. Monk et al., Imprinting disorders: a group of congenital disorders with overlapping patterns of molecular changes affecting imprinted loci. Clin. Epigenetics 7, 123 2015). https://doi.org/10.1186/s13148-015-0143-8
M. Bastepe, A.H. Lane, H. Juppner, Paternal uniparental isodisomy of chromosome 20q-and the resulting changes in GNAS1 methylation-as a plausible cause of pseudohypoparathyroidism. Am. J. Hum. Genet. 68(5), 1283–1289 (2001). https://doi.org/10.1086/320117
S. Turan, J. Ignatius, J.S. Moilanen, O. Kuismin, H. Stewart, N.P. Mann et al., De novo STX16 deletions: an infrequent cause of pseudohypoparathyroidism type Ib that should be excluded in sporadic cases. J. Clin. Endocrinol. Metab. 97(12), E2314–E2319 (2012). https://doi.org/10.1210/jc.2012-2920
P.J. Wittkopp, G. Kalay, Cis-regulatory elements: molecular mechanisms and evolutionary processes underlying divergence. Nat. Rev. Genet. 13(1), 59–69 (2011). https://doi.org/10.1038/nrg3095
H. Neto, A. Kaupisch, L.L. Collins, G.W. Gould, Syntaxin 16 is a master recruitment factor for cytokinesis. Mol. Biol. Cell 24(23), 3663–3674 (2013). https://doi.org/10.1091/mbc.E13-06-0302
G. Kang, H. Yun, C.H. Sun, I. Park, S. Lee, J. Kwon et al., Integrated genomic analyses identify frequent gene fusion events and VHL inactivation in gastrointestinal stromal tumors. Oncotarget 7(6), 6538–6551 (2016). https://doi.org/10.18632/oncotarget.3731
M. Bastepe, H. Juppner, GNAS locus and pseudohypoparathyroidism. Horm. Res. 63(2), 65–74 (2005). https://doi.org/10.1159/000083895
X. Chu, Y. Zhu, O. Wang, M. Nie, T. Quan, Y. Xue et al., Clinical and genetic characteristics of pseudohypoparathyroidism in the Chinese population. Clin. Endocrinol. 88(2), 285–294 (2018). https://doi.org/10.1111/cen.13516
Z. Coban-Akdemir, J.J. White, X. Song, S.N. Jhangiani, J.M. Fatih, T. Gambin et al., Identifying genes whose mutant transcripts cause dominant disease traits by potential gain-of-function alleles. Am. J. Hum. Genet. 103(2), 171–187 (2018). https://doi.org/10.1016/j.ajhg.2018.06.009
I.M. de Lange, A.A. Verrijn Stuart, R.B. van der Luijt, H.K. Ploos van Amstel, M.M. van Haelst, Macrosomia, obesity, and macrocephaly as first clinical presentation of PHP1b caused by STX16 deletion. Am. J. Med. Genet. A 170(9), 2431–2435 (2016). https://doi.org/10.1002/ajmg.a.37818
E. Fernandez-Rebollo, B. Lecumberri, I. Garin, J. Arroyo, A. Bernal-Chico, F. Goni et al., New mechanisms involved in paternal 20q disomy associated with pseudohypoparathyroidism. Eur. J. Endocrinol. 163(6), 953–962 (2010). https://doi.org/10.1530/eje-10-0435
R. Guerreiro, J. Bras, S. Batista, P. Pires, M.H. Ribeiro, M.R. Almeida et al., Pseudohypoparathyroidism type I-b with neurological involvement is associated with a homozygous PTH1R mutation. Genes Brain Behav. 15(7), 669–677 (2016). https://doi.org/10.1111/gbb.12308
L.F. Fröhlich, M. Mrakovcic, R. Steinborn, U.-I. Chung, M. Bastepe, H. Juppner, Targeted deletion of the Nesp55 DMR defines another Gnas imprinting control region and provides a mouse model of autosomal dominant PHP-Ib. PNAS 107(20), 9275–9280 (2010)
A. Linglart, M. Bastepe, H. Juppner, Similar clinical and laboratory findings in patients with symptomatic autosomal dominant and sporadic pseudohypoparathyroidism type Ib despite different epigenetic changes at the GNAS locus. Clin. Endocrinol. 67(6), 822–831 (2007). https://doi.org/10.1111/j.1365-2265.2007.02969.x
A. Linglart, S. Maupetit-Mehouas, C. Silve, GNAS-related loss-of-function disorders and the role of imprinting. Horm. Res. Paediatr. 79(3), 119–129 (2013). https://doi.org/10.1159/000348516
S. Mulchandani, E.J. Bhoj, M. Luo, N. Powell-Hamilton, K. Jenny, K.W. Gripp et al., Maternal uniparental disomy of chromosome 20: a novel imprinting disorder of growth failure. Genet. Med. 18(4), 309–315 (2016). https://doi.org/10.1038/gim.2015.103
F.I. Rezwan, R.L. Poole, T. Prescott, J.M. Walker, I. Karen Temple, D.J. Mackay, Very small deletions within the NESP55 gene in pseudohypoparathyroidism type 1b. Eur. J. Hum. Genet. 23(4), 494–499 (2015). https://doi.org/10.1038/ejhg.2014.133
Y. Wang, M. Nie, O. Wang, Y. Li, Y. Jiang, M. Li et al., Genetic screening in a large Chinese cohort of childhood onset hypoparathyroidism by next-generation sequencing combined with TBX1-MLPA. J. Bone Miner. Res. (2019). https://doi.org/10.1002/jbmr.3854
C.M. Williamson, S.T. Ball, W.T. Nottingham, J.A. Skinner, A. Plagge, M.D. Turner et al., A cis-acting control region is required exclusively for the tissue-specific imprinting of Gnas. Nat. Genet. 36(8), 894–899 (2004). https://doi.org/10.1038/ng1398
Acknowledgements
We thank the patient for getting permission to publish relative information.
Funding
This work was supported by the Chinese Academy of Medical Sciences (CAMS) Initiative for Innovative Medicine (CAMS-I2M) (No. 2017-I2M-1–001) and the National Natural Science Foundation of China (No. 81873641).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
All of the procedures performed in the study involving the human participant were approved by the Ethics Committee of Peking Union Medical College Hospital and conducted under the instructions of the principles in the Declaration of Helsinki.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
About this article
Cite this article
Yang, Y., Chu, X., Nie, M. et al. A novel long-range deletion spanning STX16 and NPEPL1 causing imprinting defects of the GNAS locus discovered in a patient with autosomal-dominant pseudohypoparathyroidism type 1B. Endocrine 69, 212–219 (2020). https://doi.org/10.1007/s12020-020-02304-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12020-020-02304-6