Abstract
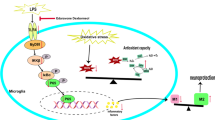
Microglia-mediated neuroinflammation is known to play a pivotal role in the pathogenesis of different neurological diseases. Gastrodin, a phenolic glucoside, has been reported to exert anti-inflammatory effects in activated microglia challenged with lipopolysaccharide (LPS); however, the underlying mechanism has remained obscure. The present study aimed to ascertain if Gastrodin would regulate the Notch signaling pathway involved in microglia activation. We show here that LPS increased the expression of various members of the Notch-1 pathway, including intracellular Notch receptor domain (NICD), recombining binding protein suppressor of hairless (RBP-Jκ) and transcription factor hairy and enhancer of split-1 (Hes-1) in microglia in postnatal rat brain and in BV-2 microglia. Remarkably, Gastrodin was found to markedly attenuate the expression of the above various biomarkers both in vivo and in vitro. Moreover, increased phosphorylation level of ERK, JNK and P38 induced by LPS was attenuated with pretreatment of Notch-1 signaling inhibitor, N-[N-(3,5-difluorophenacetyl)-1-alany1-Sphenyglycinet-butylester (DAPT) as well as Gastrodin. Gastrodin mimicked the effects of DAPT by inhibiting the LPS-induced expression of IL-1β, IL-6, IL-23, TNF-α and NO. Moreover, lentivirus transfection mediated NICD overexpression inhibited the anti-inflammatory effects of Gastrodin. Furthermore, the activation of Notch-1 signaling promoted microglia migration and Gastrodin could inhibit the migration of activated BV-2 microglia by regulating the Notch-1 signaling pathway. In light of the above, our results indicate that Notch-1 signaling pathway is involved in the anti-inflammatory effects of Gastrodin against LPS-induced microglia activation. These findings provide a new biological target of Gastrodin for the treatment of neuroinflammatory disorders.








Similar content being viewed by others
Data Availability
All data generated or analyzed during this study are included in this published article.
Abbreviations
- AD:
-
Alzheimer’s disease
- ADAM:
-
A Disintegrin and Metalloproteinase
- CNS:
-
Central nervous system
- COX-2:
-
Cyclooxygenase-2
- DAPT:
-
N-[N-(3,5-Difluorophenacetyl)-1-alany1-sphenygl-ycinet-butylester
- DMEM:
-
Dulbecco’s Modified Eagle Medium
- ECL:
-
Enhanced chemiluminescence
- ELISA:
-
Enzyme-linked immunosorbent assay
- ERK:
-
Extracellular signal-regulated kinases
- FBS:
-
Fetal bovine serum
- Hes-1:
-
Hairy and enhancer of split-1
- IL-10:
-
Interleukin-10
- IL-1β:
-
Interleukin-1β
- IL-23:
-
Interleukin-23
- IL-6:
-
Interleukin-6
- iNOS:
-
Inducible nitric oxide synthase
- JNK:
-
C-Jun N-terminal kinase
- LPS:
-
Lipopolysaccharide
- MAPK:
-
Mitogen-activated protein kinase
- MCP-1:
-
Monocyte chemotactic protein-1
- NO:
-
Nitric oxide
- PD:
-
Parkinson’s disease
- PVDF membranes:
-
Polyvinylidene fluoride membranes
- RBP-Jκ:
-
Recombining binding protein suppressor of hairless
- ROS:
-
Reactive oxygen species
- RT-PCR:
-
Real time polymerase chain reactions
- TBS:
-
Tris-buffered saline
- TNF-α:
-
Tumor necrosis factor-α
References
Ajmone-Cat, M. A., De Simone, R., Nicolini, A., & Minghetti, L. (2003). Effects of phosphatidylserine on p38 mitogen activated protein kinase, cyclic AMP responding element binding protein and nuclear factor-kappaB activation in resting and activated microglial cells. Journal of Neurochemistry, 84(2), 413–416.
Ando, K., Kanazawa, S., Tetsuka, T., Ohta, S., Jiang, X., Tada, T., et al. (2003). Induction of Notch signaling by tumor necrosis factor in rheumatoid synovial fibroblasts. Oncogene, 22(49), 7796–7803.
Ankeny, D. P., & Popovich, P. G. (2009). Mechanisms and implications of adaptive immune responses after traumatic spinal cord injury. Neuroscience, 158(3), 1112–1121.
Asnaghi, L., Lin, M. H., Lim, K. S., Lim, K. J., Tripathy, A., Wendebornet, M., et al. (2014). Hypoxia promotes uveal melanoma invasion through enhanced notch and MAPK activation. PLoS ONE, 9(8), e105372.
Campbell, A. (2010). Inflammation, neurodegenerative diseases, and environmental exposures. Annals of the New York Academy of Sciences, 1035, 117–132.
Cao, Q., Li, P., Lu, J., Dheen, S. T., Kaur, C., & Ling, E. A. (2010). Nuclear factor-κB/p65 responds to changes in the Notch signaling pathway in murine BV-2 cells and in amoeboid microglia in postnatal rats treated with the γ-secretase complex blocker DAPT. Journal of Neuroscience Research, 88(12), 2701–2714.
Chen, P. Z., Jiang, H. H., Wen, B., Ren, S. C., Chen, Y., Ji, W. G., et al. (2014). Gastrodin suppresses the amyloid β-induced increase of spontaneous discharge in the entorhinal cortex of rats. Neural Plasticity, 2014, 320937.
Choi, Y., Lee, M. K., Lim, S. Y., Sung, S. H., & Kim, Y. C. (2009). Inhibition of inducible NO synthase, cyclooxygenase-2 and interleukin-1beta by torilin is mediated by mitogen-activated protein kinases in microglial BV-2 cells. British Journal of Pharmacology, 156(6), 933–940.
Dai, J. N., Zong, Y., Zhong, L. M., Li, Y. M., Zhang, W., Bian, L. G., et al. (2011). Gastrodin inhibits expression of inducible NO synthase, cyclooxygenase-2 and proinflammatory cytokines in cultured LPS-stimulated microglia via MAPK pathways. PLoS ONE, 6(7), e21891.
Dheen, S. T., Jun, Y., Yan, Z., Tay, S. S., & Ling, E. A. (2005). Retinoic acid inhibits expression of TNF-alpha and iNOS in activated rat microglia. Glia, 50(1), 21–31.
Fontana, A., Gast, H., Reith, W., Recher, M., Birchler, T., & Bassetti, C. L. (2010). Narcolepsy: Autoimmunity, effector T cell activation due to infection, or T cell independent, major histocompatibility complex class II induced neuronal loss? Brain, 133(Pt 5), 1300–1311.
Hara, H., Kimoto, D., Kajita, M., Takada, C., Kamiya, T., & Adachi, T. (2017). Apomorphine prevents LPS-induced IL-23 p19 mRNA expression via inhibition of JNK and ATF4 in HAPI cells. European Journal of Pharmacology, 795, 108–114.
Huo, Y., Rangarajan, P., Ling, E. A., & Dheen, S. T. (2011). Dexamethasone inhibits the Nox-dependent ROS production via suppression of MKP-1-dependent MAPK pathways in activated microglia. BMC Neuroscience, 12, 49.
Jin, R., Yang, G., & Li, G. (2010). Inflammatory mechanisms in ischemic stroke: Role of inflammatory cells. Journal of Leukocyte Biology, 87(5), 779–789.
Kaur, C., Ling, E. A., & Wong, W. C. (1985). Transformation of amoeboid microglial cells into microglia in the corpus callosum of the postnatal rat brain: An electron microscopical study. Archivum Histologicum Japonicum, 48(1), 17–25.
Kelly, D. F., Lake, R. J., Walz, T., & Artavanis-Tsakonas, S. (2007). Conformational variability of the intracellular domain of Drosophila Notch and its interaction with Suppressor of Hairless. Proceedings of the National Academy of Sciences of the United States of America, 104(23), 9591–9596.
Kharitonov, S. A., & Sjöbring, U. (2007). Lipopolysaccharide challenge of humans as a model for chronic obstructive lung disease exacerbations. Contributions to Microbiology, 14, 83–100.
Kopan, R. (2002). Notch: A membrane-bound transcription factor. Journal of Cell Science, 115(Pt 6), 1095–1097.
Krämer, H. (2000). RIPping notch apart: a new role for endocytosis in signal transduction? Science’s STKE, 2000(29), pe1.
Lee, Y. G., Lee, J., Byeon, S. E., Yoo, D. S., Kim, M. H., Lee, S. Y., et al. (2011). Functional role of Akt in macrophage-mediated innate immunity. Frontiers in Bioscience, 16, 517–530.
Ling, E. A., & Wong, W. C. (1993). The origin and nature of ramified and amoeboid microglia: A historical review and current concepts. Glia, 7(1), 9–18.
Livak, K. J., & Schmittgen, T. D. (2001). Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) Method. Methods, 25(4), 402–408.
Mendsaikhan, A., Tooyama, I., & Walker, D. G. (2019). Microglial progranulin: Involvement in Alzheimer’s disease and neurodegenerative diseases. Cells, 8(3), 230.
Mittal, S., Subramanyam, D., Dey, D., Kumar, R. V., & Rangarajan, A. (2009). Cooperation of notch and Ras/MAPK signaling pathways in human breast carcinogenesis. Molecular Cancer, 8, 128–140.
Nakajima, K., & Kohsaka, S. (1998). Functional roles of microglia in the central nervous system. Human Cell, 11(3), 141–155.
Nam, K. N., Koketsu, M., & Lee, E. H. (2008). 5-Chloroacetyl-2-amino-1,3-selenazoles attenuate microglial inflammatory responses through NF-kappaB inhibition. European Journal of Pharmacology, 589(1–3), 53–57.
Neher, J. J., Neniskyte, U., Hornik, T., & Brown, G. C. (2014). Inhibition of UDP/P2Y6 purinergic signaling prevents phagocytosis of viable neurons by activated microglia in vitro and in vivo. Glia, 62(9), 1463–1475.
Neumann, H., Kotter, M. R., & Franklin, R. J. M. (2009). Debris clearance by microglia: An essential link between degeneration and regeneration. Brain, 132(Pt 2), 288–295.
Porcelli, L., Mazzotta, A., Garofoli, M., Fonte, R. D., Guida, G., Guida, M., et al. (2021). Active notch protects MAPK activated melanoma cell lines from MEK inhibitor cobimetinib. Biomedicine & Pharmacotherapy, 133, 111006. https://doi.org/10.1016/j.biopha.2020.111006
Selkoe, D., & Kopan, R. (2003). Notch and presenilin: Regulated intramembrane proteolysis links development and degeneration. Annual Review of Neuroscience, 26, 565–597.
Semple, B. D., Kossmann, T., & Morganti-Kossmann, M. C. (2010). Role of chemokines in CNS health and pathology: A focus on the CCL2/CCR2 and CXCL8/CXCR2 networks. Journal of Cerebral Blood Flow and Metabolism, 30(3), 459–473.
Siebel, C., & Lendahl, U. (2017). Notch signaling in development, tissue homeostasis, and disease. Physiological Review, 97(4), 1235–1294.
Sivakumar, V., Ling, E. A., Lu, J., & Kaur, C. (2010). Role of glutamate and its receptors and insulin-like growth factors in hypoxia induced periventricular white matter injury. Glia, 58(5), 507–523.
Thomas, W. E. (1992). Brain macrophages: Evaluation of microglia and their functions. Brain Research Reviews, 17(1), 61–74.
Wang, M. J., Huang, H. Y., Chen, W. F., Chang, H. F., & Kuo, J. S. (2010). Glycogen synthase kinase-3β inactivation inhibits tumor necrosis factor-α production in microglia by modulating nuclear factor κB and MLK3/JNK signaling cascades. Journal of Neuroinflammation, 7, 99.
Wang, X. L., Xing, G. H., Hong, B., Li, X. M., Zou, Y., Zhang, X. J., et al. (2014). Gastrodin prevents motor deficits and oxidative stress in the MPTP mouse model of Parkinson’s disease: Involvement of ERK1/2-Nrf2 signaling pathway. Life Sciences, 114(2), 77–85.
Wang, Y., Wu, Z., Liu, X., & Fu, Q. (2013). Gastrodin ameliorates Parkinson’s disease by downregulating connexin 43. Molecular Medicine Reports, 8(2), 585–590.
Xu, X., Yang, C., Chen, J., Liu, J., Li, P., Shi, Y., et al. (2018). Interleukin-23 promotes the migration and invasion of gastric cancer cells by inducing epithelial-to-mesenchymal transition via the STAT3 pathway. Biochemical and Biophysical Research Communications, 499(2), 273–278.
Yao, L., Kan, E. M., Kaur, C., Dheen, S. T., Hao, A., Lu, J., et al. (2013). Notch-1 signaling regulates microglia activation via NF-κB pathway after hypoxic exposure in vivo and in vitro. PLoS ONE, 8(11), e78439.
Zhang, Q., Wang, C., Liu, Z., Liu, X., Han, C., Cao, X., et al. (2012). Notch signal suppresses Toll-like receptor-triggered inflammatory responses in macrophages by inhibiting extracellular signal-regulated kinase 1/2-mediated nuclear factor κB activation. Journal of Biological Chemistry, 287(9), 6208–6217.
Zhu, C., Xiong, Z., Chen, X., Peng, F., Hu, X., Chen, Y., et al. (2012). Artemisinin attenuates lipopolysaccharide-stimulated proinflammatory responses by inhibiting NF-κB pathway in microglia cells. PLoS ONE, 7(4), e35125.
Funding
This study was supported by the National Natural Sciences Foundation of China (Grants Nos. 81960251, 31760292, 81460210, 81360200), Department project of Science and Technology of Yunnan Province (Grants Nos. 2017FA035, 2019ZF011-2, 2018IA048, 2018FE001(-029), 202001AY070001-174) and the Department project of Education of Yunnan Province (Grants No. 2019J1214).
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Material preparation, data collection and analysis were performed by Y.Y., R.L., Y.Z. and J.G.; the first draft of the manuscript was written by Y.Y. and Y.G.; D.L. and L.Z. designed experiments and obtained science grant support. All authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that there was no conflict of interest.
Ethical Approval
All animal experiments were designed to minimize suffering and the number of animals used. All procedures were carried out in accordance with the National Institute of Health Guide for the Care and Use of Laboratory Animals (NIH Publications number 80-23). The project was approved by the Department of Science and Technology, Yunnan Province, China.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Yao, Yy., Li, R., Guo, Yj. et al. Gastrodin Attenuates Lipopolysaccharide-Induced Inflammatory Response and Migration via the Notch-1 Signaling Pathway in Activated Microglia. Neuromol Med 24, 139–154 (2022). https://doi.org/10.1007/s12017-021-08671-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12017-021-08671-1