Abstract
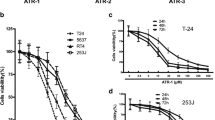
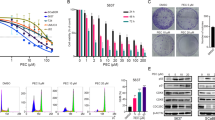
Catalpol is an iridoid glucoside extracted from the traditional Chinese medicinal plant called Rehmannia glutinosa, and it is reported to possess a variety of pharmacological activities. The goal of this research was to explore the effect of catalpol on the human bladder cancer cells T24. The cells were treated for different durations with various concentrations of catalpol. Then the viability, mobility, and invasiveness of the cells were determined by MTT assay and flow cytometry, respectively. Catalpol was found to significantly suppress the proliferation, migration, and invasiveness of T24 bladder cancer cells in a dose- and time-dependent manner. The results also revealed that catalpol promoted apoptosis and caused the cell-cycle arrest at G2/M phase. Furthermore, the catalpol-mediated apoptosis was found to involve the modulation of PI3K/Akt pathway and inhibit the expression of anti-apoptotic B cell lymphoma-2 (Bcl-2) family proteins. Also, the expressions of Bcl-2 proapoptotic effectors, Bcl-2-associated X protein, and Bcl-2 associated death promoter were up-regulated in the cells treated with catalpol. The enzymes involved in the ultimate process of apoptosis, active caspase-3 and poly ADP ribose polymerase were elevated, and the latter was observed to be cleaved, indicating that catalpol-accelerated cell apoptosis involved a caspase-dependent pathway. These results suggest that catalpol might serve to be a promising therapeutic agent for the treatment of human bladder cancer.






Similar content being viewed by others
References
Dong, Z., & Chen, C. X. (2013). Effect of catalpol on diabetic nephropathy in rats. Phytomedicine, 20, 1023–1029.
Feng, Y., Liu, Z., Peng, Y., et al. (2013). Validated LC-MS method for simultaneous quantitation of catalpol and harpagide in rat plasma: Application to a comparative pharmacokinetic study in normal and diabetic rats after oral administration of Zeng-Ye-Decoction. Biomedical Chromatography, 27, 1503–1510.
Chen, C., Chen, Z., Xu, F., et al. (2013). Radio-protective effect of catalpol in cultured cells and mice. Journal of Radiation Research, 54, 76–82.
Chen, W., Li, X., Jia, L. Q., et al. (2013). Neuroprotective activities of catalpol against CaMKII-dependent apoptosis induced by LPS in PC12 cells. British Journal of Pharmacology, 169, 1140–1152.
Choi, H. J., Jang, H. J., Chung, T. W., et al. (2013). Catalpol suppresses advanced glycation end-products-induced inflammatory responses through inhibition of reactive oxygen species in human monocytic THP-1 cells. Fitoterapia, 86, 19–28.
Bi, J., Jiang, B., Zorn, A., et al. (2013). Catalpol inhibits LPS plus IFN-gamma-induced inflammatory response in astrocytes primary cultures. Toxicology in Vitro, 27, 543–550.
Wan, D., Xue, L., Zhu, H., et al. (2013). Catalpol induces neuroprotection and prevents memory dysfunction through the cholinergic system and BDNF. Evidence-Based Complementary and Alternative Medicine, 2013, 134852.
Zhang, X., Jin, C., Li, Y., et al. (2013). Catalpol improves cholinergic function and reduces inflammatory cytokines in the senescent mice induced by d-galactose. Food and Chemical Toxicology, 58, 50–55.
Huang, C., Cui, Y., Ji, L., et al. (2013). Catalpol decreases peroxynitrite formation and consequently exerts cardioprotective effects against ischemia/reperfusion insult. Pharmaceutical Biology, 51, 463–473.
Saracoglu, I., & Harput, U. S. (2012). In vitro cytotoxic activity and structure activity relationships of iridoid glucosides derived from Veronica species. Phytotherapy Research, 26, 148–152.
Saracoglu, I., Oztunca, F. H., Nagatsu, A., et al. (2011). Iridoid content and biological activities of Veronica cuneifolia subsp. cuneifolia and V. cymbalaria. Pharmaceutical Biology, 49, 1150–1157.
Pungitore, C. R., Leon, L. G., Garcia, C., et al. (2007). Novel antiproliferative analogs of the Taq DNA polymerase inhibitor catalpol. Bioorganic & Medicinal Chemistry Letters, 17, 1332–1335.
Nguyen, A. T., Fontaine, J., Malonne, H., et al. (2005). A sugar ester and an iridoid glycoside from Scrophularia ningpoensis. Phytochemistry, 66, 1186–1191.
Pungitore, C. R., Ayub, M. J., Borkowski, E. J., et al. (2004). Inhibition of Taq DNA polymerase by catalpol. Cellular and Molecular Biology (Noisy-Le-Grand), 50, 767–772.
Zhu, Y., Mao, Y., Chen, H., et al. (2013). Apigenin promotes apoptosis, inhibits invasion and induces cell cycle arrest of T24 human bladder cancer cells. Cancer Cell International, 13, 54.
Shen, Z., Shen, T., Wientjes, M. G., O’Donnell, M. A., & Au, J. L. (2008). Intravesical treatments of bladder cancer. Pharmaceutical Research, 25(7), 1500–1510.
Bai, L., Chen, J., McEachern, D., Liu, L., Zhou, H., et al. (2014). BM-1197: A novel and specific Bcl-2/Bcl-xL inhibitor inducing complete and long-lasting tumor regression in vivo. PLoS One, 9(6), e99404.
Zhou, B., Li, Y., Deng, Q., et al. (2013). SRPK1 contributes to malignancy of hepatocellular carcinoma through a possible mechanism involving PI3K/Akt. Molecular and Cellular Biochemistry, 379, 191–199.
Jiang, S., Zhu, W., Li, C., et al. (2013). alpha-Lipoic acid attenuates LPS-induced cardiac dysfunction through a PI3K/Akt-dependent mechanism. International Immunopharmacology, 16, 100–107.
Chen, X., Wang, H., Ou-Yang, X. N., et al. (2013). Research on drug resistance mechanism of trastuzumab caused by activation of the PI3K/Akt signaling pathway. Contemporary Oncology (Poznan), 17, 363–369.
Ma, Y., Lu, C., Li, C., et al. (2013). Overexpression of HSPA12B protects against cerebral ischemia/reperfusion injury via a PI3K/Akt-dependent mechanism. Biochimica et Biophysica Acta, 1832, 57–66.
FrankeTF, Hornik C. P., Segev, L., Shostak, G. A., & Sugimoto, C. (2003). PI3K/Akt and apoptosis: Size matters. Oncogene, 22, 8983–8998.
Lee, K. M., Kang, H. A., Park, M., et al. (2012). Interleukin-24 suppresses the growth of vascular smooth muscle cells by inhibiting H(2)O(2)-induced reactive oxygen species production. Pharmacology, 90, 332–341.
Miyake, N., Chikumi, H., Takata, M., et al. (2012). Rapamycin induces p53-independent apoptosis through the mitochondrial pathway in non-small cell lung cancer cells. Oncology Reports, 28, 848–854.
Noori, S., & Hassan, Z. M. (2012). Tehranolide inhibits proliferation of MCF-7 human breast cancer cells by inducing G0/G1 arrest and apoptosis. Free Radical Biology and Medicine, 52, 1987–1999.
Ma, Y., Hao, X., Zhang, S., et al. (2012). The in vitro and in vivo effects of human umbilical cord mesenchymal stem cells on the growth of breast cancer cells. Breast Cancer Research and Treatment, 133, 473–485.
Lu, J., Wang, Y., Zhao, W., et al. (2014). Effects of catalpol, l-shikonin and paeonol extracted from radix rehmanniae, radix arnebiaeand cortex moutan on KGF-induced HaCaT cell proliferation. Zhonghua Yi Xue Za Zhi, 94, 1265–1269.
Garcia, C., Leon, L. G., Pungitore, C. R., et al. (2010). Enhancement of antiproliferative activity by molecular simplification of catalpol. Bioorganic and Medicinal Chemistry, 18, 2515–2523.
Alnemri, E. S. (1997). Mammalian cell death proteases: A family of highly conserved aspartate specific cysteine proteases. Journal of Cellular Biochemistry, 64(1), 33–42.
Majeed, R., Hamid, A., Sangwan, P. L., Chinthakindi, P. K., Koul, S., Rayees, S., et al. (2014). Inhibition of phophotidylinositol-3 kinase pathway by a novel naphthol derivative of betulinic acid induces cell cycle arrest and apoptosis in cancels of different origin. Cell Death and Disease, 5, e1459.
Hsu, Y. Y., Liu, C. M., Tsai, H. H., et al. (2010). KMUP-1 attenuates serum deprivation-induced neurotoxicity in SH-SY5Y cells: Roles of PKG, PI3K/Akt and Bcl-2/Bax pathways. Toxicology, 268, 46–54.
Moon, D. O., Kim, M. O., Choi, Y. H., et al. (2008). Beta-Sitosterol induces G2/M arrest, endoreduplication, and apoptosis through the Bcl-2 and PI3K/Akt signaling pathways. Cancer Letters, 264, 181–191.
Jin, Y. P., Fishbein, M. C., Said, J. W., et al. (2004). Anti-HLA class I antibody-mediated activation of the PI3K/Akt signaling pathway and induction of Bcl-2 and Bcl-xL expression in endothelial cells. Human Immunology, 65, 291–302.
Isabelle, M., Moreel, X., Gagné, J. P., Rouleau, M., Ethier, C., Gagné, P., et al. (2010). Investigation of PARP-1, PARP-2, and PARG interactomes by affinity-purification mass spectrometry. Proteome Science, 8, 22.
Pawlowski, J., & Kraft, A. S. (2000). Bax-induced apoptotic cell deat. Proceedings of National Academy of Sciences, 97(2), 529–531.
Hu, W., Shen, T., & Wang, M. H. (2011). Cell cycle arrest and apoptosis induced by methyl 3,5-dicaffeoyl quinate in human colon cancer cells: Involvement of the PI3K/Akt and MAP kinase pathways. Chemico-Biological Interactions, 194, 48–57.
Roy, S. K., Srivastava, R. K., & Shankar, S. (2010). Inhibition of PI3K/AKT and MAPK/ERK pathways causes activation of FOXO transcription factor, leading to cell cycle arrest and apoptosis in pancreatic cancer. Journal of Molecular Signaling, 5, 10.
Author information
Authors and Affiliations
Corresponding author
Additional information
Dr. Di Jin and Dr. Ming Cao have contributed equally to this study.
Rights and permissions
About this article
Cite this article
Jin, D., Cao, M., Mu, X. et al. Catalpol Inhibited the Proliferation of T24 Human Bladder Cancer Cells by Inducing Apoptosis Through the Blockade of Akt-Mediated Anti-apoptotic Signaling. Cell Biochem Biophys 71, 1349–1356 (2015). https://doi.org/10.1007/s12013-014-0355-0
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12013-014-0355-0