Opinion statement
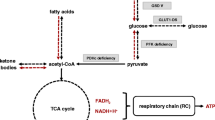
GLUT1 deficiency syndrome (GLUT1DS) results from impaired glucose transport into the brain: awareness of its wide phenotypic spectrum is a prerequisite in order to ensure an early diagnosis, treating the patients is the subsequent challenge to allow prompt compensation for the brain’s lack of fuel. The ketogenic diet (KD) plays a primary role in the treatment of GLUT1DS because it provides ketone bodies as an alternative source to meet the demands of energy of the brain. Therefore, we recommend early initiation of the KD based on the assumption that early diagnosis and treatment improves the long term neurological outcome: the classic KD (4:1 or 3:1) at the present time is the most proven and effective in GLUT1DS. A KD should be continued at least until adolescence, although there are reports of good tolerability even in adulthood, possibly with a less rigorous ratio; in our experience seizure and movement disorder control can be achieved by a 2:1 ketogenic ratio but the relationship between ketosis and neurodevelopmental outcome remains undetermined. Other types of KDs can, therefore, be considered. The Modified Atkins diet, for example, is also well tolerated and provides effective symptom control; furthermore, this diet has the advantage of being easy to prepare and more palatable, which are important requirements for good compliance. Nevertheless, about 20 % of these patients have compliance trouble or the same diet loses its effectiveness over time; for these reasons, new therapeutic strategies are currently under investigation but further studies on pathophysiological mechanisms and potential effects of novel “diets” or “therapies” are needed for this new pathology.


Similar content being viewed by others
References and Recommended Reading
Papers of particular interest, published recently, have been highlighted as: • Of importance •• Of major importance
Vannucci SJ, Maher F, Simpson IA. Glucose transporter proteins in brain: delivery of glucose to neurons and glia. Glia. 1997;21:2–21.
De Vivo DC, Trifiletti RR, Jacobson RI, Ronen GM, Behman RA, Harik SI. Defective glucose transport across the blood-brain barrier as a cause of persistent hypoglycorrhachia, seizures, and developmental delay. N Engl J Med. 1991;325:703–9.
Wang D, Pascual JM, Yang H, Engelstad K, Jhung S, Sun RP, et al. Glut-1 deficiency syndrome: clinical, genetic, and therapeutic aspects. Ann Neurol. 2005;57:111–8.
Brockmann K. The expanding phenotype of GLUT1-deficiency syndrome. Brain and Development. 2009;31:545–52.
Leen WG, Klepper J, Verbeek MM, Leferink M, Hofste T, van Engelen BG, et al. Glucose transporter-1 deficiency syndrome: the expanding clinical and genetic spectrum of a treatable disorder. Brain. 2010;133(Pt 3):655–70.
Suls A, Dedeken P, Goffin K, Van Esch H, Dupont P, Cassiman D, et al. Paroxysmal exerciseinduced dyskinesia and epilepsy is due to mutations in SLC2A1, encoding the glucose transporter GLUT1. Brain. 2008;131:1831–44.
Pons R, Collins A, Rotstein M, Engelstad K, De Vivo DC. The spectrum of movement disorders in Glut-1 deficiency. Mov Disord. 2010;25:275–81.
Klepper J, Scheffer H, Elsaid MF, Jamsteeg E-J, Leferink M, Ben-Omran T. Autosomal recessive inheritance of GLUT1 deficiency syndrome. Neuropediatrics. 2009;40:207–10.
Rotstein M, Engelstad K, Yang H, Wang D, Levy B, Chung WK, et al. Glut1 deficiency: inheritance pattern determined by haploinsufficiency. Ann Neurol. 2010;68:955–8.
De Giorgis V, Veggiotti P. GLUT1 deficiency syndrome 2013: current state of the art. Seizure. 2013;22(10):803–11. This recent review clearly details the different GLUT1DS phenotypes, highlights the latest diagnostic and therapeutic procedures and it provides a handy and useful diagnostic flow chart.
Ramm-Pettersen A, Nakken KO, Skogseid IM, Randby H, Skei EB, Bindoff LA, et al. Good outcome in patients with early dietary treatment of GLUT-1 deficiency syndrome: results from a retrospective Norwegian study. Dev Med Child Neurol. 2013;55(5):440–7.
Gras D, Roze E, Caillet S, Méneret A, Doummar D, Billette de Villemeur T, et al. GLUT1 deficiency syndrome: an update. Rev Neurol (Paris). 2013 Nov 20.
Veggiotti P, Teutonico F, Alfei E, Nardocci N, Zorzi G, Tagliabue A, et al. Glucose transporter type 1 deficiency: ketogenic diet in three patients with atypical phenotype. Brain Dev. 2010;32:404–8.
Pong AW, Geary BR, Engelstad KM, Natarajan A, Yang H, De Vivo DC. Glucose transporter type I deficiency syndrome: epilepsy phenotypes and outcome. Epilepsia. 2012;53(9):503–10. This retrospective study details the epilepsy phenotypes and treatment response to the ketogenic diet and anticonvulsant mediations in 87 patients with Glut1DS.
Klepper J, Scheffer H, Leiendecker B, Gertsen E, Binder S, Leferink M, et al. Seizure control and acceptance of the ketogenic diet in GLUT1 deficiency syndrome: a 2- to 5-year follow-up of 15 children enrolled prospectively. Neuropediatrics. 2005;36(5):302–8.
Tzadok M, Nissenkorn A, Porper K, Matot I, Marcu S, Anikster Y, et al. The many faces of glut1 deficiency syndrome. J Child Neurol. 2014;29(3):349–59. doi:10.1177/0883073812471718.
Klepper J, Leiendecker B. Glut1 deficiency syndrome and novel ketogenic diets. J Child Neurol. 2013;28(8):1045–8. It is a clear, concise and detailed paper on the current data on novel ketogenic diets and faces the implications on the use of these diets in regard to GLUT1DS.
Klepper J. GLUT1 deficiency syndrome in clinical practice. Epilepsy Res. 2012;100:272–7. This accurate and precise review is useful for a practical approach in the suspicion of GLUT1DS. It faces the complexity of symptoms’ recognition, the genetic heterogeneity and diagnostic difficulties about cut-offs for hypoglycorrhachia.
Kim DW, Kang HC, Park JC, Kim HD. Benefits of the nonfasting ketogenic diet compared with the initial fasting ketogenic diet. Pediatrics. 2004;114:1627–30.
Kossoff EH, McGrogan JR, Bluml RM. A modified Atkins diet is effective for the treatment of intractable pediatric epilepsy. Epilepsia. 2006;47:421–4.
Kossoff EH, Cervenka MC, Henry BJ, Haney CA, Turner Z. A decade of the modified Atkins diet (2003–2013): results, insights, and future directions. Epilepsy Behav. 2013;29(3):437–42. This review discusses the past decade of experience with the modified Atkins diet with results demonstrating similar efficacy to the classical ketogenic diet and its tolerability.
Haberlandt E, Karall D, Jud V, Baumgartner SS, Zotter S, Rostasy K, et al. Glucose transporter type 1 deficiency syndrome effectively treated with modified Atkins diet. Neuropediatrics. 2013 Jul 25.
Pfeifer HH, Thiele EA. Low-glycemic-index treatment: a liberalized ketogenic diet for treatment of intractable epilepsy. Neurology. 2005;5:1810–2.
Kossoff EH, Freeman JM, Turner Z, Rubenstein JE. Ketogenic diets: treatments for epilepsy and other disorders. 5th ed. New York NY: Demos Medical; 2011. Clear and complete overview on the ketogenic diets, with particular regard to the practical management in the different clinical pictures.
Yang H, Wang D, Engelstad K, Bagay L, Wei Y, Rotstein M, et al. Glut1 deficiency syndrome and erythrocyte glucose uptake assay. Ann Neurol. 2011;70(6):996–1005.
Klepper J, Leiendecker B. GLUT1 deficiency syndrome – 2007 update. Dev Med Child Neurol. 2007;49:707–16.
Weber YG, Storch A, Wuttke TV, Brockmann K, Kempfle J, Maljevic S, et al. GLUT1 mutations are a cause of paroxysmal exertion-induced dyskinesias and induce hemolytic anemia by a cation leak. J Clin Invest. 2008;118(6):2157–68.
Urbizu A, Cuenca-Leon E, Raspall-Chaure M, Gratacòs M, Conill J, Redecillas S, et al. Paroxysmal exercise-induced dyskinesia, writer’s cramp, migraine with aura and absence epilepsy in twin brothers with a novel SLC2A1 missense mutation. J Neurol Sci. 2010;295:110–3.
Friedman JR, Thiele EA, Wang D, Levine KB, Cloherty EK, Pfeifer HH. Atypical GLUT1 deficiency with prominent movement disorder responsive to ketogenic diet. Mov Disord. 2006;21(2):241–5.
Gramer G, Wolf NI, Vater D, Bast T, Santer R, Kamsteeg EJ, et al. Glucose transporter-1 (GLUT1) deficiency syndrome: diagnosis and treatment in late childhood. Neuropediatrics. 2012;43(3):168–71. This paper interestingly describes symptoms, diagnostic results, and effects of therapy in two GLUT1-DS patients aged 10 and 15 years.
Ito S, Oguni H, Ito Y, Ishigaki K, Ohinata J, Osawa M. Modified Atkins diet therapy for a case with glucose transporter type 1 deficiency syndrome. Brain Dev. 2008;30:226–8.
Coman DJ, Sinclair KG, Burke CJ, Appleton DB, Pelekanos JT, O'Neil CM, et al. Seizures, ataxia, developmental delay and the general paediatrician: glucose transporter 1 deficiency syndrome. J Paediatr Child Health. 2006;42:263–7.
Ito Y, Oguni H, Ito S, Oguni M, Osawa M. A modified Atkins diet is promising as a treatment for glucose transporter type 1 deficiency syndrome. Dev Med Child Neurol. 2011;53(7):658–63.
Kitamura Y, Okumura A, Hayashi M, Mori H, Takahashi S, Yanagihara K, et al. Oxidative stress markers and phosphorus magnetic resonance spectroscopy in a patient with GLUT1 deficiency treated with modified Atkins diet. Brain Dev. 2012;34(5):372–5.
Pearson TS, Akman C, Hinton VJ, Engelstad K, De Vivo DC. Phenotypic spectrum of glucose transporter type 1 deficiency syndrome (Glut1 DS). Curr Neurol Neurosci Rep. 2013;13(4):342.
Klepper J, Fischbarg J, Vera JC, Wang D, De Vivo DC. GLUT1- deficiency: barbiturates potentiate haploinsufficiency in vitro. Pediatr Res. 1999;46(6):677–83.
von Moers A, Brockmann K, Wang D, Korenke CG, Huppke P, De Vivo DC, et al. EEG features of glut-1 deficiency syndrome. Epilepsia. 2002;43(8):941–5.
Cano A, Ticus I, Chabrol B. Glucose transporter type 1 (GLUT-1) deficiency. Rev Neurol (Paris). 2008;164(11):896–901.
Verrotti A, D’Egidio C, Agostinelli S, Gobbi G. Glut1 deficiency: when to suspect and how to diagnose? Eur J Paediatr Neurol. 2012;16:3–9.
Anheim M, Maillart E, Vuillaumier-Barrot S, Flamand-Rouvière C, Pineau F, Ewenczyk C, et al. Excellent response to acetazolamide in a case of paroxysmal dyskinesias due to GLUT1-deficiency. J Neurol. 2011;258:316–7.
De Vivo DC, Leary L, Wang D. Glucose transporter 1 deficiency syndrome and other glycolytic defects. J Child Neurol. 2002;17 Suppl 3:S15–23.
Mochel F, DeLonlay P, Touati G, Brunengraber H, Kinman RP, Rabier D, et al. Pyruvate carboxylase deficiency: clinical and biochemical response to anaplerotic diet therapy. Mol Genet Metab. 2005;84(4):305–12.
Kinman RP, Kasumov T, Jobbins KA, Thomas KR, Adams JE, Brunengraber LN, et al. Parenteral and enteral metabolism of anaplerotic triheptanoin in normal rats. Am J Physiol Endocrinol Metab. 2006;291(4):E860–6.
Deng S, Zhang GF, Kasumov T, Roe CR, Brunengraber H. Interrelations between C4 ketogenesis, C5 ketogenesis, and anaplerosis in the perfused rat liver. J Biol Chem. 2009;284(41):27799–807.
Borges K, Sonnewald U. Triheptanoin–a medium chain triglyceride with odd chain fatty acids: a new anaplerotic anticonvulsant treatment? Epilepsy Res. 2012;100(3):239–44.
Willis S, Stoll J, Sweetman L, Borges K. Anticonvulsant effects of a triheptanoin diet in two mouse chronic seizure models. Neurobiol Dis. 2010;40(3):565–72.
Thomas NK, Willis S, Sweetman L, Borges K. Triheptanoin in acute mouse seizure models. Epilepsy Res. 2012;99(3):312–7.
Kim TH, Borges K, Petrou S, Reid CA. Triheptanoin reduces seizure susceptibility in a syndrome-specific mouse model of generalized epilepsy. Epilepsy Res. 2013;103(1):101–5.
Veggiotti P, Burlina A, Coppola G, Cusmai R, De Giorgis V, Guerrini R, et al. The ketogenic diet for Dravet syndrome and other epileptic encephalopathies: an Italian consensus. Epilepsia. 2011;52 Suppl 2:83–9. This is an interesting review that provides a consensus statement regarding the clinical management, patient selection, counseling and follow-up of the ketogenic diet in the Italian experience.
Chambon R, Vuillaumier-Barrot S, Seta N, Wagner S, Sarret C. Partial effectiveness of acetazolamide in a mild form of GLUT1 deficiency: a pediatric observation. Mov Disord. 2013;28(12):1749–51.
Brockmann K. Towards a more palatable treatment for Glut1 deficiency syndrome. Dev Med Child Neurol. 2011;53(7):580–1.
Acknowledgment
The authors wish to thank A.I.E.F. ONLUS Foundation for its support.
Compliance with Ethics Guidelines
Conflict of Interest
This work has been supported by FONDAZIONE CARIPLO, grant n°8 2010-0759.
Pierangelo Veggiotti and Valentina De Giorgis declare that they have no conflict of interest.
Human and Animal Rights and Informed Consent
This article does not contain any studies with human or animal subjects performed by any of the authors.
Author information
Authors and Affiliations
Corresponding author
Additional information
This article is part of the Topical Collection on Pediatric Neurology
Rights and permissions
About this article
Cite this article
Veggiotti, P., De Giorgis, V. Dietary Treatments and New Therapeutic Perspective in GLUT1 Deficiency Syndrome. Curr Treat Options Neurol 16, 291 (2014). https://doi.org/10.1007/s11940-014-0291-8
Published:
DOI: https://doi.org/10.1007/s11940-014-0291-8