Abstract
Purpose of Review
The electrophysiology lab is an important source of growth of anesthetic volume as the indications and evidence for catheter ablations and various cardiac implantable electronic devices improve. Paired with this increase in volume is an increasing number of patients with substantial comorbid conditions presenting for their EP procedures. For these patients, the interaction between their comorbidities and traditional anesthesia practices may create the risk of hemodynamic instability, cardiovascular or respiratory complications, and potential need for prolonged post-operative monitoring negatively impacting length of hospital stay.
Recent Findings
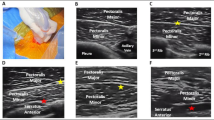
Regional anesthetic techniques, including pectoralis, serratus, and erector spinae plane blocks, offer options for both regional analgesia and surgical anesthesia for a variety of EP procedures. Existing case reports and extrapolations from other areas support these techniques as viable, safe, and effective components of an anesthetic plan.
Summary
In this article, we will review the development and challenges of various EP procedures and how different regional anesthetic techniques can function as a component of the anesthesia plan.


Similar content being viewed by others
Data Availability
Not applicable.
References
Papers of particular interest, published recently, have been highlighted as: • Of importance
• Du AL, Robbins K, Waterman RS, et al. National trends in nonoperating room anesthesia: procedures, facilities, and patient characteristics. Curr Opin Anaesthesiol. 2021;34(4):464–9. Retrospective study demonstrating the substantial growth in nonoperating room procedures and the impact of this growth on anesthesia services.
Lu AC, Wald SH, Sun EC. Into the wilderness?: The growing importance of nonoperating room anesthesia care in the United States. Anesth Analg. 2017;124(4):1044–6.
Kornej J, Borschel CS, Benjamin EJ, et al. Epidemiology of atrial fibrillation in the 21st century: novel methods and new insights. Circ Res. 2020;127(1):4–20.
Parameswaran R, Al-Kaisey AM, Kalman JM. Catheter ablation for atrial fibrillation: current indications and evolving technologies. Nat Rev Cardiol. 2021;18(3):210–25.
Hsu JC, Darden D, Du C, et al. Initial findings from the national cardiovascular data registry of atrial fibrillation ablation procedures. J Am Coll Cardiol. 2023;81(9):867–78.
Ghzally Y, Ahmed I, Gerasimon G. Catheter ablation. StatPearls. Treasure Island (FL) 2023.
Vladinov G, Fermin L, Longini R, et al. Choosing the anesthetic and sedative drugs for supraventricular tachycardia ablations: a focused review. Pacing Clin Electrophysiol. 2018;41(11):1555–63.
Steffen MM, Osborn JS, Cutler MJ. Cardiac implantable electronic device therapy: permanent pacemakers, implantable cardioverter defibrillators, and cardiac resynchronization devices. Med Clin North Am. 2019;103(5):931–43.
Kozlowski B. [Commentary to the article: Moss AJ, Hall WJ, Cannom DS, et al. Cardiac-resynchronization therapy for the prevention of heart-failure events. N Engl J Med 2009; 361: 1329–38]. Kardiol Pol. 2009;67(12):1417–8.
Kober L, Thune JJ, Nielsen JC, et al. Defibrillator implantation in patients with nonischemic systolic heart failure. N Engl J Med. 2016;375(13):1221–30.
Groeneveld PW, Dixit S. Cardiac pacing and defibrillation devices: cost and effectiveness. Annu Rev Med. 2017;68:1–13.
Friedman P, Murgatroyd F, Boersma LVA, et al. Efficacy and safety of an extravascular implantable cardioverter-defibrillator. N Engl J Med. 2022;387(14):1292–302.
Knops RE, Olde Nordkamp LRA, Delnoy PHM, et al. Subcutaneous or transvenous defibrillator therapy. N Engl J Med. 2020;383(6):526–36.
Kleemann T, Becker T, Doenges K, et al. Annual rate of transvenous defibrillation lead defects in implantable cardioverter-defibrillators over a period of >10 years. Circulation. 2007;115(19):2474–80.
Abdin A, Aktaa S. Subcutaneous ICD for more and transvenous ICD for few?! Clin Res Cardiol. 2022;111(5):475–8.
Brouwer TF, Miller MA, Quast AB, et al. Implantation of the subcutaneous implantable cardioverter-defibrillator: an evaluation of 4 implantation techniques. Circ Arrhythm Electrophysiol. 2017;10(1):e004663.
Miller MA, Bhatt HV, Weiner M, et al. Implantation of the subcutaneous implantable cardioverter-defibrillator with truncal plane blocks. Heart Rhythm. 2018;15(7):1108–11.
• Zhang Y, Min J, Chen S. Analgesic efficacy of regional anesthesia of the hemithorax in patients undergoing subcutaneous implantable cardioverter-defibrillator placement. J Cardiothorac Vasc Anesth. 2021;35(11):3288–93. First randomized trial to demonstrate the clinical utility of regional anesthesia in the anesthetic management of patients in the EP lab.
Shariat A, Ghia S, Gui JL, et al. Use of serratus anterior plane and transversus thoracis plane blocks for subcutaneous implantable cardioverter-defibrillator (S-ICD) implantation decreases intraoperative opioid requirements. J Cardiothorac Vasc Anesth. 2021;35(11):3294–8.
Yang JK, Char DS, Motonaga KS, et al. Pectoral nerve blocks decrease postoperative pain and opioid use after pacemaker or implantable cardioverter-defibrillator placement in children. Heart Rhythm. 2020;17(8):1346–53.
Mavarez AC, Ripat CI, Suarez MR. Pectoralis plane block for pacemaker insertion: a successful primary anesthetic. Front Surg. 2019;6:64.
Elders J, AlHashimi H, Gomes M, et al. Subcutaneous ICD implantation under ultrasound-guided serratus anterior plane block: single-center experience in the Netherlands. Int J Cardiol Heart Vasc. 2022;38:100949.
Droghetti A, Basso Ricci E, Scimia P, et al. Ultrasound-guided serratus anterior plane block combined with the two-incision technique for subcutaneous ICD implantation. Pacing Clin Electrophysiol. 2018;41(5):517–23.
Krishna SN, Chauhan S, Bhoi D, et al. Bilateral erector spinae plane block for acute post-surgical pain in adult cardiac surgical patients: a randomized controlled trial. J Cardiothorac Vasc Anesth. 2019;33(2):368–75.
Zhang J, He Y, Wang S, et al. The erector spinae plane block causes only cutaneous sensory loss on ipsilateral posterior thorax: a prospective observational volunteer study. BMC Anesthesiol. 2020;20(1):88.
• Hasak L, Jagielak D, Lango R, et al. Pain control with ultrasound-guided ilioinguinal-iliohypogastric nerve block compared with local infiltration anesthesia in patients undergoing transfemoral transcatheter aortic valve implantation: a prospective randomized trial. Journal of Cardiothoracic and Vascular Anesthesia. 2019;33:S86-S7. Randomized trial showing the benefit to femoral nerve blocks for transcatheter procedures requiring a femoral artery approach.
Funding
This work was supported by the Department of Anesthesiology and Perioperative Medicine, Tufts Medical Center, Boston, MA.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
None.
Human and Animal Rights and Informed Consent
This article does not contain any studies with human or animal subjects performed by any of the authors.t
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Oh, D., Stapleton, G. & Brovman, E.Y. Utilization of Regional Anesthesia in the Electrophysiology Lab: A Narrative Review. Curr Pain Headache Rep 27, 455–459 (2023). https://doi.org/10.1007/s11916-023-01147-w
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11916-023-01147-w