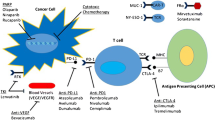
Opinion statement
Research into novel therapies for gynecologic cancers is underfunded, and as a result, we are still playing catchup with other solid tumors in the realm of immune checkpoint inhibition. This is despite the fact that two of the most common gynecologic cancers in the USA have strong biologic rationales for response to these agents. Work is now underway to demonstrate safe and effective therapies for our patients. As we better understand the immune system, and more specifically the tumor microenvironment, we will be able to achieve complete responses. The immune system can learn, adapt, and provide ongoing surveillance; if only we could mimic its abilities.
Similar content being viewed by others
References and Recommended Reading
Papers of particular interest, published recently, have been highlighted as: • Of importance •• Of major importance
Lynch HT, Lynch J. Lynch syndrome: genetics, natural history, genetic counseling, and prevention. J Clin Oncol. 2000;18(21 Suppl):19S–31S.
Lynch HT, Casey MJ, Lynch J, White TE, Godwin AK. Genetics and ovarian carcinoma. Semin Oncol. 1998;25(3):265–80.
Barrow E, Robinson L, Alduaij W, Shenton A, Clancy T, Lalloo F, et al. Cumulative lifetime incidence of extracolonic cancers in Lynch syndrome: a report of 121 families with proven mutations. Clin Genet. 2009;75(2):141–9.
Schmeler KM, Soliman PT, Sun CC, Slomovitz BM, Gershenson DM, Lu KH. Endometrial cancer in young, normal-weight women. Gynecol Oncol. 2005;99(2):388–92.
Dudley JC, Lin MT, Le DT, Eshleman JR. Microsatellite instability as a biomarker for PD-1 blockade. Clin Cancer Res. 2016;22(4):813–20.
Boussiotis VA. Molecular and biochemical aspects of the PD-1 checkpoint pathway. N Engl J Med. 2016;375(18):1767–78.
Iwai Y, Hamanishi J, Chamoto K, Honjo T. Cancer immunotherapies targeting the PD-1 signaling pathway. J Biomed Sci. 2017;24(1):26.
Pietzner K, Nasser S, Alavi S, Darb-Esfahani S, Passler M, Muallem MZ, et al. Checkpoint-inhibition in ovarian cancer: rising star or just a dream? J Gynecol Oncol. 2018;29(6):e93.
Hamanishi J, Mandai M, Matsumura N, Abiko K, Baba T, Konishi I. PD-1/PD-L1 blockade in cancer treatment: perspectives and issues. Int J Clin Oncol. 2016;21(3):462–73.
Siegel RL, Miller KD, Jemal A. Cancer statistics, 2019. CA Cancer J Clin. 2019;69(1):7–34.
Cronin KA, Lake AJ, Scott S, Sherman RL, Noone AM, Howlader N, et al. Annual report to the nation on the status of cancer, part I: national cancer statistics. Cancer. 2018;124(13):2785–800.
McMeekin DS, Filiaci VL, Thigpen JT, Gallion HH, Fleming GF, Rodgers WH, et al. The relationship between histology and outcome in advanced and recurrent endometrial cancer patients participating in first-line chemotherapy trials: a gynecologic oncology group study. Gynecol Oncol. 2007;106(1):16–22.
Bokhman JV. Two pathogenetic types of endometrial carcinoma. Gynecol Oncol. 1983;15(1):10–7.
• Cancer Genome Atlas Research N, Kandoth C, Schultz N, Cherniack AD, Akbani R, Liu Y, et al. Integrated genomic characterization of endometrial carcinoma. Nature. 2013;497(7447):67–73 Landmark paper that shifted our understanding of how to classify endometrial cancers.
Zou W, Wolchok JD, Chen L. PD-L1 (B7-H1) and PD-1 pathway blockade for cancer therapy: mechanisms, response biomarkers, and combinations. Sci Transl Med. 2016;8(328):328rv4.
• Le DT, Uram JN, Wang H, Bartlett BR, Kemberling H, Eyring AD, et al. PD-1 blockade in tumors with mismatch-repair deficiency. N Engl J Med. 2015;372(26):2509–20 Demonstrated the potential of checkpoint inhibitors in dMMR tumors, including gynecologic cancers.
Herzog TJ, Arguello D, Reddy SK, Gatalica Z. PD-1, PD-L1 expression in 1599 gynecological cancers: implications for immunotherapy. Gynecol Oncol. 2015;137:204–5.
Eggink FA, Van Gool IC, Leary A, Pollock PM, Crosbie EJ, Mileshkin L, et al. Immunological profiling of molecularly classified high-risk endometrial cancers identifies POLE-mutant and microsatellite unstable carcinomas as candidates for checkpoint inhibition. Oncoimmunology. 2017;6(2):e1264565.
Ott PA, Bang YJ, Berton-Rigaud D, Elez E, Pishvaian MJ, Rugo HS, et al. Safety and antitumor activity of pembrolizumab in advanced programmed death ligand 1-positive endometrial cancer: results from the KEYNOTE-028 study. J Clin Oncol. 2017;35(22):2535–41.
•• Le DT, Durham JN, Smith KN, Wang H, Bartlett BR, Aulakh LK, et al. Mismatch repair deficiency predicts response of solid tumors to PD-1 blockade. Science. 2017;357(6349):409–13 Established checkpoint inhibitors as a therapeutic option for dMMR tumors, regardless of tissue of origin.
Lemery S, Keegan P, Pazdur R. First FDA approval agnostic of Cancer site - when a biomarker defines the indication. N Engl J Med. 2017;377(15):1409–12.
Santin AD, Bellone S, Buza N, Choi J, Schwartz PE, Schlessinger J, et al. Regression of chemotherapy-resistant polymerase epsilon (POLE) ultra-mutated and MSH6 hyper-mutated endometrial tumors with nivolumab. Clin Cancer Res. 2016;22(23):5682–7.
Fuh K, Lomonosova E, Guo L, Fantl W, Pachynski R, Malkova O, et al. Identifying a potential biomarker for anti-PD-1 immunotherapy in patients with advanced stage, surgically-resectable endometrial cancer. ASCO Annual Meeting; June 1, 2019; Chicago, IL. J Clin Oncol. 2019;37(suppl; abstr 5590).
Secord AA. A study of pembrolizumab on the tumoral immunoprofile of gynecologic cancers. NCT02728830. ClinicalTrials.gov: NCI; [Available from: https://clinicaltrials.gov/ct2/show/NCT02728830. Accessed 14 June 2019.
Santin AD. Pembrolizumab in ultramutated and hypermutated endometrial cancer. NCT02899793. ClinicalTrials.gov: NCI; [Available from: https://clinicaltrials.gov/ct2/show/NCT02899793?term=02899793&rank=1. Accessed 14 June 2019.
Makker V, Rasco D, Vogelzang NJ, Brose MS, Cohn AL, Mier J, et al. Lenvatinib plus pembrolizumab in patients with advanced endometrial cancer: an interim analysis of a multicentre, open-label, single-arm, phase 2 trial. Lancet Oncol. 2019;20(5):711–8.
Makker V, Herraez A, Aghajanian C, Fujiwara K, Pignata S, Penson RT, et al. A phase 3 trial evaluating efficacy and safety of lenvatinib in combination with pembrolizumab in patients with advanced endometrial cancer. ASCO Annual Meeting; June 1, 2019; Chicago, IL. J Clin Oncol. 2019;37(suppl; abstr TPS5607).
Eskander RN, NRG-Oncology. Testing the addition of the immunotherapy drug pembrolizumab to the usual chemotherapy treatment (paclitaxel and carboplatin) in stage iii-iv or recurrent endometrial cancer, NCT03914612. clinicaltrials.gov: NCI; [Available from: https://clinicaltrials.gov/ct2/show/NCT03914612?term=03914612&rank=1. Accessed 14 June 2019.
Willis BC, Sloan EA, Atkins KA, Stoler MH, Mills AM. Mismatch repair status and PD-L1 expression in clear cell carcinomas of the ovary and endometrium. Mod Pathol. 2017;30(11):1622–32.
Al-Hussaini M, Lataifeh I, Jaradat I, Abdeen G, Otay L, Badran O, et al. Undifferentiated endometrial carcinoma, an immunohistochemical study including PD-L1 testing of a series of cases from a single cancer center. Int J Gynecol Pathol. 2018;37(6):564–74.
Antill Y, Kok P, Robledo K, Barnes E, Friedlander M, Baron-Hay S, et al. Activity of durvalumab in advanced endometrial cancer (AEC) according to mismatch repair (MMR) status: The phase II PHAEDRA trial (ANZGOG1601). ASCO Annual Meeting; June 3, 2019; Chicago, IL. J Clin Oncol. 2019;37(suppl; abstr 5501).
Konstantinopoulos PA, Liu JF, Luo W, Krasner CN, Ishizuka J, Gockley A, et al. Phase 2, two-group, two-stage study of avelumab in patients (pts) with microsatellite stable (MSS), microsatellite instable (MSI), and polymerase epsilon (POLE) mutated recurrent/persistent endometrial cancer (EC). ASCO Annual Meeting; June 3, 2019; Chicago, IL. J Clin Oncol. 2019;37(suppl;abstr 5502).
Colombo N, Barretina-Ginesta M, Beale P, Harano K, Hudson E, Marme F, et al. AtEnd/ENGOT-en7: A multicenter phase III double-blind randomized controlled trial of atezolizumab in combination with paclitaxel and carboplatin in women with advanced/recurrent endometrial cancer. . ASCO Annual Meeting; June 1, 2019; Chicago, IL. J Clin Oncol. 2019;37(suppl; abst TPS5608).
Rubinstein M, Caird I, Zhou Q, Iasonos A, Friedman C, Cadoo K, et al. A phase II trial of durvalumab with or without tremelimumab in patients with persistent or recurrent endometrial carcinoma and endometrial carcinosarcoma. . ASCO Annual Meeting June 1, 2019; Chicago, IL J Clin Oncol. 2019;37(suppl; abstr 5582).
Oh MS, Chae YK. Deep and durable response with combination CTLA-4 and PD-1 blockade in mismatch repair (MMR)-proficient endometrial cancer. J Immunother. 2019;42(2):51–4.
Antomarchi J, Ambrosetti D, Cohen C, Delotte J, Chevallier A, Karimdjee-Soilihi B, Ngo-Mai M, Schmid-Alliana A, Schmid-Antomarchi H. Immunosuppressive tumor microenvironment status and histological grading of endometrial carcinoma. Cancer Microenviron. 2019.
Winer I, Jones NL, Xiu J, Ellerbrock A, Brown J, Herzog T. Mutational burden, tumor immune checkpoint expression, and microsatellite instability in gynecologic malignancies: implications for immune therapy. Gynecol Oncol. 2018;149:45.
Jones NL, Xiu J, Herzog T, Winer I. Immune checkpoint expression, microsatellite instability, and mutational burden: identifying immune biomarker phenotypes in uterine cancer. Gynecol Oncol. 2018;149:44.
Yamashita H, Nakayama K, Ishikawa M, Nakamura K, Ishibashi T, Sanuki K, et al. Microsatellite instability is a biomarker for immune checkpoint inhibitors in endometrial cancer. Oncotarget. 2018;9(5):5652–64.
Moore M, Ring KL, Mills AM. TIM-3 in endometrial carcinomas: an immunotherapeutic target expressed by mismatch repair-deficient and intact cancers. Mod Pathol. 2019;32:1168–79.
Asaka S, Yen TT, Wang TL, Shih IM, Gaillard S. T cell-inflamed phenotype and increased Foxp3 expression in infiltrating T-cells of mismatch-repair deficient endometrial cancers. Mod Pathol. 2019;32(4):576–84.
Wortman BG, Bosse T, Nout RA, Lutgens LCHW, van der Steen-Banasik EM, Westerveld H, et al. Molecular-integrated risk profile to determine adjuvant radiotherapy in endometrial cancer: evaluation of the pilot phase of the PORTEC-4a trial. Gynecol Oncol. 2018;151(1):69–75.
Lee L, Matulonis U. Immunotherapy and radiation combinatorial trials in gynecologic cancer: a potential synergy? Gynecol Oncol. 2019.
Son CH, Fleming GF, Moroney JW. Potential role of radiation therapy in augmenting the activity of immunotherapy for gynecologic cancers. Cancer Manag Res. 2017;9:553–63.
Reijnen C, Küsters-Vandevelde HVN, Prinsen CF, Massuger LFAG, Snijders MPML, Kommoss S, et al. Mismatch repair deficiency as a predictive marker for response to adjuvant radiotherapy in endometrial cancer. Gynecol Oncol. 2019.
Talhouk A, McConechy MK, Leung S, Yang W, Lum A, Senz J, et al. Confirmation of ProMisE: a simple, genomics-based clinical classifier for endometrial cancer. Cancer. 2017;123(5):802–13.
Britton H, Huang L, Lum A, Leung S, Shum K, Kale M, et al. Molecular classification defines outcomes and opportunities in young women with endometrial carcinoma. Gynecol Oncol. 2019;153(3):487–95.
•• Talhouk A, Derocher H, Schmidt P, Leung S, Milne K, Gilks CB, et al. Molecular subtype not immune response drives outcomes in endometrial carcinoma. Clin Cancer Res. 2019;25(8):2537–48 A remarkable paper that further demonstrated the utility of the ProMisE classification of endometrial cancers, as well as the significance of TILs in the microenvironment.
Mullen MM, Mutch DG. Endometrial tumor immune response: predictive biomarker of response to immunotherapy. Clin Cancer Res. 2019;25(8):2366–8.
Bookman MA, Brady MF, McGuire WP, Harper PG, Alberts DS, Friedlander M, et al. Evaluation of new platinum-based treatment regimens in advanced-stage ovarian cancer: a phase III trial of the gynecologic cancer InterGroup. J Clin Oncol. 2009;27:1419–25.
• Hamanishi J, Mandai M, Ikeda T, Minami M, Kawaguchi A, Murayama T, et al. Safety and antitumor activity of anti-PD-1 antibody, nivolumab, in patients with platinum-resistant ovarian cancer. J Clin Oncol. 2015;33(34):4015–22 Demonstrated the use of checkpoint inhibitors in ovarian cancer.
Hamanishi J, Murakami R, Baba T, Yamaguchi K, Abiko K, Mandai M. Passenger fusion genes are correlated to antitumor effect of anti-PD-1 antibody nivolumab for ovarian cancer. 2019 SGO annual meeting; march 2019; Honolulu, HI.
Varga A, Piha-Paul S, Ott PA, Mehnert JM, Berton-Rigaud D, Morosky A, et al. Pembrolizumab in patients with programmed death ligand 1-positive advanced ovarian cancer: analysis of KEYNOTE-028. Gynecol Oncol. 2019;152(2):243–50.
Bellone S, Buza N, Choi J, Zammataro L, Gay L, Elvin J, et al. Exceptional response to pembrolizumab in a metastatic, chemotherapy/radiation-resistant ovarian cancer patient harboring a PD-L1-genetic rearrangement. Clin Cancer Res. 2018;24(14):3282–91.
Disis ML, Taylor MH, Kelly K, Beck JT, Gordon M, Moore KM, et al. Efficacy and safety of avelumab for patients with recurrent or refractory ovarian cancer: phase 1b results from the JAVELIN solid tumor trial. JAMA Oncol. 2019;5:393–401.
Emens LA, Kok M, Ojalvo LS. Targeting the programmed cell death-1 pathway in breast and ovarian cancer. Curr Opin Obstet Gynecol. 2016;28(2):142–7.
Zhang G, Liu C, Bai H, Cao G, Cui R, Zhang Z. Combinatorial therapy of immune checkpoint and cancer pathways provides a novel perspective on ovarian cancer treatment. Oncol Lett. 2019;17(3):2583–91.
Pujade-Lauraine E, Fujiwarab K, Ledermann JA, Oza A, Kristeleit R, Ray-Coquard I, et al. Avelumab alone or in combination with pegylated liposomal doxorubicin versus pegylated liposomal doxorubicin alone in platinum-resistant or refractory epithelial ovarian cancer: primary and biomarker analysis of the phase III JAVELIN Ovarian 200 trial. 2019 SGO Annumal meeting; march 17, 2019; Honolulu, HI.
Liao JB, Gwin W, Urban R, Hitchcock-Bernhardt K, Coveler A, Higgins D, et al. Pembrolizumab with low dose carboplatin for recurrent platinum resistant ovarian, fallopian tube, and primary peritoneal cancer-interim results. 2019 ASCO annual meeting; Chicago, IL. J Clin Oncol. 2019;37(suppl; abstr 5519).
Moore KN, Pignata S. Trials in progress: IMagyn050/GOG 3015/ENGOT-OV39. A phase III, multicenter, randomized study of atezolizumab versus placebo administered in combination with paclitaxel, carboplatin, and bevacizumab to patients with newly-diagnosed stage III or stage IV ovarian, fallopian tube, or primary peritoneal cancer. Int J Gynecol Cancer. 2019.
Liu YL, Zamarin D. Combination immune checkpoint blockade strategies to maximize immune response in gynecological cancers. Curr Oncol Rep. 2018;20(12):94.
Penn C, Lester J, Bohrer K, Moon C, Yearley J, Karlan BY, et al. PD-1/PD-L1 expression in BRCA1/2 mutated ovarian cancers. 2019 SGO annual meeting march 2019; Honolulu, HI.
Konstantinopoulos PA, Waggoner S, Vidal GA, Mita M, Moroney JW, Holloway R, et al. Single-arm phases 1 and 2 trial of niraparib in combination with pembrolizumab in patients with recurrent platinum-resistant ovarian carcinoma. JAMA Oncol. 2019;5:1141.
Monk BJ, Coleman RL, Fujiwara K, Oza AM, Westin SN, O’Malley DM, et al. ATHENA (GOG-3020/ENGOT-ov45): a randomised, double-blind, placebo-controlled, phase 3 study of rucaparib + nivolumab following front-line platinum-based chemotherapy in ovarian cancer. 2018 International Gynecologic Cancer Society Biennial Meeting; Kyoto, Japan.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare they have no conflict of interest.
Human and Animal Rights and Informed Consent
This article does not contain any studies with human or animal subjects performed by any of the authors.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This article is part of the Topical Collection on Gynecologic Cancers
Rights and permissions
About this article
Cite this article
Ferriss, J.S., Williams-Brown, M.Y. Immunotherapy: Checkpoint Inhibitors in Lynch-Associated Gynecologic Cancers. Curr. Treat. Options in Oncol. 20, 75 (2019). https://doi.org/10.1007/s11864-019-0676-8
Published:
DOI: https://doi.org/10.1007/s11864-019-0676-8