Opinion statement
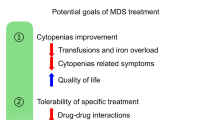
Aging is the most potent of carcinogens, especially for the bone marrow stem cell clonal disorders called myelodysplastic syndromes (MDS). Age-associated changes in the microenvironment or the soil of the bone marrow (BM) as well as in the cell or the seed provide a growth advantage for clonal myeloid cells. Slowly accumulating senescent cells which can no longer divide because they have reached the end of their proliferative life cycle, but which continue to produce metabolic debris, overwhelm the natural autophagy mechanisms resulting in pro-inflammatory changes in the BM soil. In addition, the seed or stem cells acquire passenger mutations with each round of proliferation resulting from DNA copying errors. Some mutations commonly associated with MDS can be found in older, otherwise healthy individuals; however, when combined with other passenger mutations or in the setting of a noxious soil, the result could be a proliferative advantage for one stem cell over others, leading to its clonal expansion and development of the clinical syndrome. When considering therapeutic options for MDS patients, the important considerations are related to both the common co-morbidities of an elderly population along with the heterogeneous passenger mutations and the inflammatory changes in the soil. At present, allogeneic stem cell transplant is the only potentially curative option in MDS. Palliative strategies are directed at improving the quality of life and prolonging survival. Only three drugs are FDA approved, two being the hypomethylating agents azacytidine and decitabine while the third is lenalidomide which is restricted to lower risk MDS patients with deletion 5q. Promising future therapies are directed at reversing the pro-inflammatory changes in the microenvironment (luspatercept) or targeting specific mutations isocitrate dehydrogenase (IDH)1, IDH2, p53, EZH2. More durable responses are to be expected when the seed and soil are targeted simultaneously through a combination of drugs.

Similar content being viewed by others
References and Recommended Reading
Papers of particular interest, published recently, have been highlighted as: • Of importance •• Of major importance
Tefferi A, Vardiman JW. Myelodysplastic syndromes. N Engl J Med. 2009;361:1872–85.
Bejar R, Stevenson K, Abdel-Wahab O, et al. Clinical effect of point mutations in myelodysplastic syndromes. N Engl J Med. 2011;364:2496–506.
Papaemmanuil E, Gerstung M, Malcovati L, et al. Clinical and biological implications of driver mutations in myelodysplastic syndromes. Blood. 2013;122:3616–27.
Haferlach T, Nagata Y, Grossmann V, et al. Landscape of genetic lesions in 944 patients with myelodysplastic syndromes. Leukemia. 2014;28:241–7.
Fenaux P, Mufti GJ, Hellstrom-Lindberg E, et al. Efficacy of azacitidine compared with that of conventional care regimens in the treatment of higher-risk myelodysplastic syndromes: a randomised, open-label, phase III study. Lancet Oncol. 2009;10:223–32.
Kantarjian H, Issa JP, Rosenfeld CS, et al. Decitabine improves patient outcomes in myelodysplastic syndromes: results of a phase III randomized study. Cancer. 2006;106:1794–803.
List A, Kurtin S, Roe DJ, et al. Efficacy of lenalidomide in myelodysplastic syndromes. N Engl J Med. 2005;352:549–57.
De Witte T, Hermans J, Vossen J, et al. Haematopoietic stem cell transplantation for patients with myelodysplastic syndromes and secondary acute myeloid leukaemias: a report on behalf of the Chronic Leukaemia Working Party of the European Group for Blood and Marrow Transplantation (EBMT). Br J Haematol. 2000;110:620–30.
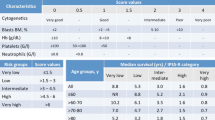
Greenberg PL, Tuechler H, Schanz J, et al. Revised international prognostic scoring system for myelodysplastic syndromes. Blood. 2012;120:2454–65.
Greenberg P, Cox C, LeBeau MM, et al. International scoring system for evaluating prognosis in myelodysplastic syndromes. Blood. 1997;89:2079–88.
Pfeilstöcker M, Tuechler H, Sanz G, et al. Time-dependent changes in mortality and transformation risk in MDS. Blood. 2016;128:902–10.
Kawabata H, Tohyama K, Matsuda A, et al. Validation of the revised International Prognostic Scoring System in patients with myelodysplastic syndrome in Japan: results from a prospective multicenter registry. Int J Hematol. 2017;106:375–84.
• Bejar R, Papaemmanuil E, Haferlach T, et al. Somatic mutations in MDS patients are associated with clinical features and predict prognosis independent of the IPSS-R: analysis of combined datasets from the International Working Group for Prognosis in MDS Molecular Committee. Blood. 2015;126:907 This study validates the prognostic value of mutations in several MDS-associated genes. Furthermore, mutations in several genes retain their prognostic significance after adjustment for IPSS-R risk groups refining risk prognostication. This study will serve as the template with which to build a molecular risk model for MDS.
Gangat N, Mudireddy M, Lasho TL, et al. Mutations and prognosis in myelodysplastic syndromes: karyotype-adjusted analysis of targeted sequencing in 300 consecutive cases and development of a genetic risk model. Am J Hematol. 2018;93:691–69.
Ali AM, Huang Y, Pinheiro RF, et al. Severely impaired terminal erythroid differentiation as an independent prognostic marker in myelodysplastic syndromes. Blood Adv. 2018;2:1393–402.
Moyo V, Lefebvre P, Duh MS, et al. Erythropoiesis-stimulating agents in the treatment of anemia in myelodysplastic syndromes: a meta-analysis. Ann Hematol. 2008;87(7):527–36.
Buckstein R, Balleari E, Wells R, et al. ITACA: a new validated international erythropoietic stimulating agent-response score that further refines the predictive power of previous scoring systems. Am J Hematol. 2017;92(10):1037–46.
Hellström-Lindberg E, Gulbrandsen N, Lindberg G, et al. A validated decision model for treating the anaemia of myelodysplastic syndromes with erythropoietin + granulocyte colony-stimulating factor: significant effects on quality of life. Br J Haematol. 2003;120(6):1037–46.
Park S, Grabar S, Kelaidi C, et al. Predictive factors of response and survival in myelodysplastic syndrome treated with erythropoietin and G-CSF: the GFM experience. Blood. 2008;111:574–82.
Filı C, Malagola M, Follo MY, et al. Prospective phase II study on 5-days azacitidine for treatment of symptomatic and/or erythropoietin unresponsive patients with low/INT-1-risk myelodysplastic syndromes. Clin Cancer Res. 2013;19:3297–308.
Tobiasson M, Dybedahl I, Holm MS, et al. Limited clinical efficacy of azacitidine in transfusion-dependent, growth factor-resistant, low- and Int-1 risk MDS: results from the nordic NMDSG08 phase II trial. Blood Cancer J. 2014;4:e189.
Thépot S, Abdelali RB, Chevret S, et al. A randomized phase II trial of azacitidine þ/ epoetin-b in lower risk myelodysplastic syndromes resistant to erythropoietic stimulating agents. Haematologica. 2016;101:918–25.
Sanchez-Garcia J, Falantes J, Medina Perez A, et al. Prospective randomized trial of 5 days azacitidine versus supportive care in patients with lower-risk myelodysplastic syndromes without 5q deletion and transfusion-dependent anemia. Leuk Lymphoma. 2018;59(5):1095–110.
Garcia-Manero G, Jabbour E, Borthakur G, et al. Randomized open-label phase II study of decitabine in patients with low- or intermediate-risk myelodysplastic syndromes. J Clin Oncol. (2013);31:2548–53.
Raza A, Reeves JA, Feldman EJ, et al. Phase 2 study of lenalidomide in transfusion-dependent, low-risk, and intermediate-1 risk myelodysplastic syndromes with karyotypes other than deletion 5q. Blood. 2008;111(1):86–93.
• Santini V, Almeida A, Giagounidis A, et al. Randomized phase III study of lenalidomide versus placebo in RBC transfusion-dependent patients with lower-risk non-del(5q) myelodysplastic syndromes and ineligible for or refractory to erythropoiesis-stimulating agents. J Clin Oncol. 2016;34(25):2988–96 This study supports the clinical benefits of lenalidomide in lower-risk non-del(5q) MDS.
• List AF, Sun Z, Verma A, et al. Combined treatment with lenalidomide (LEN) and epoetin alfa (EA) is superior to lenalidomide alone in patients with erythropoietin (Epo)-refractory, lower risk (LR) non-deletion 5q [del(5q)] myelodysplastic syndrome (MDS): results of the E2905 Intergroup Study-an ECOG-ACRIN Cancer Research Group Study, Grant CA180820, and the National Cancer Institute of the National Institutes of Health. Blood. 2016;128(22):223 This study shows that combination treatment with lenalidomide and epoetin alfa results in higher response rates without added toxicity. Also describes CD45 isoform as a possible biomarker of response.
Zeidan AM, Al Ali NH, Padron E, et al. Lenalidomide treatment for lower risk nondeletion 5q myelodysplastic syndromes patients yields higher response rates when used before azacitidine. Clin Lymph Myeloma Leuk. 2015;15(11):705–10.
List A, Dewald G, Bennett J, et al. Lenalidomide in the myelodysplastic syndrome with chromosome 5q deletion. N Engl J Med. 2006;355:1456–65.
Fenaux P, Giagounidis A, Selleslag D, et al. A randomized phase 3 study of lenalidomide versus placebo in RBC transfusion-dependent patients with Low-/Intermediate-1-risk myelodysplastic syndromes with del5q. Blood. 2011;118(14):3765–76.
Komrokji RS, List AF. Short- and long-term benefits of lenalidomide treatment in patients with lower-risk del(5q) myelodysplastic syndromes. Ann Oncol. 2016;27(1):62–8.
• Greenberg PL, Stone RM, Al-Kali A, et al. Myelodysplastic syndromes, version 2.2017, NCCN Clinical Practice Guidelines in Oncology. J Natl Compr Cancer Netw. 2017;15:60–87 Newest version of the American MDS clinical practice guidelines.
Grupo Español de Síndromes Mielodisplásicos (GESMD); Sociedad Española de Hematología y Hemoterapia (SEHH): Guías españolas de diagnóstico y tratamiento de los síndromes mielodisplásicos y la leucemia mielomonocítica crónica. http://gesmd.es/pdfs/guias_smd/Haematologia_Guias_SMD.pdf.
Giagounidis A. Current treatment algorithm for the management of lower-risk MDS. Hematology Am Soc Hematol Educ Program 2017;(1):453–59.
Malcovati L, Hellström-Lindberg E, Bowen D, et al. Diagnosis and treatment of primary myelodysplastic syndromes in adults: recommendations from the European LeukemiaNet. Blood. 2013;122(17):2943–64.
Fenaux P, Haase D, Sanz GF, et al. Myelodysplastic syndromes: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann Oncol. 2014;25 Suppl 3:iii57–69.
Kelaidi C, Park S, Brechignac S, et al. Treatment of myelodysplastic syndromes with 5q deletion before the lenalidomide era; the GFM experience with EPO and thalidomide. Leuk Res. 2008;32:1049–53.
Oliva EN, Lauseker M, Aloe Spiriti MA, et al. Early lenalidomide treatment for low and intermediate-1 International Prognostic Scoring System risk myelodysplastic syndromes with del(5q) before transfusion dependence. Cancer Med. 2015;4(12):1789–97.
Jadersten M, Saft L, Smith A, et al. TP53 mutations in low-risk myelodysplastic syndromes with del(5q) predict disease progression. J Clin Oncol. 2011;29(15):1971–9.
Prebet T, Cluzeau T, Park S, et al. Outcome of patients treated for myelodysplastic syndromes with 5q deletion after failure of lenalidomide therapy. Oncotarget. 2017;8(47):81926–35.
Castelli R, Schiavon R, Deliliers GL, et al. The impact of anaemia, transfusion dependency, comorbidities and polypharmacy in elderly patients with low-risk myelodysplastic syndromes. Med Oncol. 2018;35(3):33.
Cappellini MD, Porter J, El-Beshlawy A, et al. EPIC Study Investigators. Tailoring iron chelation by iron intake and serum ferritin: the prospective EPIC study of deferasirox in 1744 patients with transfusion-dependent anemias. Haematologica. 2010;95(4):557–66.
Bennett JM. MDS Foundation’s Working Group on Transfusional Iron Overload. Consensus statement on iron overload in myelodysplastic syndromes. Am J Hematol. 2008;83(11):858–61.
Sloand EM, Yong AS, Ramkissoon S, et al. Granulocyte colony-stimulating factor preferentially stimulates proliferation of monosomy 7 cells bearing the isoform IV receptor. Proc Natl Acad Sci U S A. 2006;103(39):14483–8.
Malcovati L, Germing U, Kuendgen A, et al. Time-dependent prognostic scoring system for predicting survival and leukemic evolution in myelodysplastic syndromes. J Clin Oncol. 2007;25(23):3503–10.
•• Platzbecker U, Germin U, Götze KS, et al. Luspatercept for the treatment of anaemia in patients with lower-risk myelodysplastic syndromes (PACE-MDS): a multicentre, open-label phase 2 dose-finding study with long-term extension study. Lancet Oncol. 2017;18(10):1338–47 This study shows the efficacy of luspatercept in anemic patients with lower-risk MDS, especially in those who have ≥ 15% ring sideroblasts and/or SF3B1 mutation.
Komrokji R, Garcia-Manero G, Ades L, et al. Sotatercept with long-term extension for the treatment of anaemia in patients with lower-risk myelodysplastic syndromes: a phase 2, dose-ranging trial. Lancet Haematol. 2018;5(2):e63–72.
Giagounidis A, Mufti GJ, Fenaux P, et al. Results of a randomized, double-blind study of romiplostim versus placebo in patients with low/intermediate-1-risk myelodysplastic syndrome and thrombocytopenia. Cancer. 2014;120(12):1838–46.
Oliva EN, Alati C, Santini V, et al. Eltrombopag versus placebo for low-risk myelodysplastic syndromes with thrombocytopenia (EQoL-MDS): phase 1 results of a single-blind, randomised, controlled, phase 2 superiority trial. Lancet Haematol. 2017;4(3):e127–36.
Kantarjian HM, Giles FJ, Greenberg PL, et al. Phase 2 study of romiplostim in patients with low- or intermediate-risk myelodysplastic syndrome receiving azacitidine therapy. Blood. 2010;116:3163–70.
Garcia-Manero G, Khoury HJ, Jabbour E, et al. A phase I study of oral ARRY-614, a p38 MAPK/Tie2 dual inhibitor, in patients with low or intermediate-1 risk myelodysplastic syndromes. Clin Cancer Res. 2015;21(5):985–94.
Cutler CS, Lee SJ, Greenberg P, et al. A decision analysis of allogeneic bone marrow transplantation for the myelodysplastic syndromes: delayed transplantation for low-risk myelodysplasia is associated with improved outcome. Blood. 2004;104(2):579–85.
•• Della Porta MG, Jackson CH, Alessandrino EP, et al. Decision analysis of allogeneic hematopoietic stem cell transplantation for patients with myelodysplastic syndrome stratified according to the revised International Prognostic Scoring System. Leukemia. 2017;31:2449–57 This study provides the optimal timing and the benefit of cytorreduction before transplant in lower, intermediate, and higher-risk MDS.
Majhail NS, Brunstein CG, Shanley R, et al. Reduced-intensity hematopoietic cell transplantation in older patients with AML/MDS: umbilical cord blood is a feasible option for patients without HLA-matched sibling donors. Bone Marrow Transplant. 2012;47:494–8.
Scott BL, Pasquini MC, Logan BR, et al. Myeloablative versus reduced-intensity hematopoietic cell transplantation for acute myeloid leukemia and myelodysplastic syndromes. J Clin Oncol. 2017;35(11):1154–61.
Gerds AT, Gooley TA, Estey EH, et al. Pretransplantation therapy with azacitidine vs induction chemotherapy and posttransplantation outcome in patients with MDS. Biol Blood Marrow Transplant. 2012;18(8):1211–8.
Van Gelder M, Schetelig J, Volin L, et al. Monosomal karyotype predicts poor outcome for MDS/sAML patients with chromosome 7 abnormalities after allogeneic stem cell transplantation for MDS/sAML. A study of the MDS subcommittee of the Chronic Leukemia Working Party of the European Group for Blood and Marrow Transplantation (EBMT). Blood. 2009;114:293.
Díez-Campelo M, Lorenzo JI, Itzykson R, et al. Azacitidine improves outcome in higher-risk MDS patients with chromosome 7 abnormalities: a retrospective comparison of GESMD and GFM registries. Br J Haematol. 2018;181(3):350–9.
Lubbert M, Suciu S, Baila L, et al. Low-dose decitabine versus best supportive care in elderly patients with intermediate- or high-risk myelodysplastic syndrome (MDS) ineligible for intensive chemotherapy: final results of the randomized phase III study of the European Organisation for Research and Treatment of Cancer Leukemia Group and the German MDS Study Group. J Clin Oncol. 2011;29(15):1987–96.
Lyons RM, Cosgriff TM, Modi SS, et al. Hematologic response to three alternative dosing schedules of azacitidine in patients with myelodysplastic syndromes. J Clin Oncol. 2009;27(11):1850–6.
Prébet T, Gore SD, Thépot S, et al. Outcome of acute myeloid leukaemia following myelodysplastic syndrome after azacitidine treatment failure. Br J Haematol. 2012;157(6):764–6.
Issa JJ, Roboz G, Rizzieri D, et al. Safety and tolerability of guadecitabine (SGI-110) in patients with myelodysplastic syndrome and acute myeloid leukaemia: a multicentre, randomised, dose-escalation phase 1 study. Lancet Oncol. 2015;16(9):1099–110.
Seiler M, Yoshimi A, Darman R, et al. H3B-8800, an orally available small-molecule splicing modulator, induces lethality in spliceosome-mutant cancers. Nat Med. 2018;24(4):497–504.
Sharma P, Hu-Lieskovan S, Wargo JA, et al. Primary, adaptive, and acquired resistance to cancer immunotherapy. Cell. 2017;168(4):707–23.
Daver N, Boddu P, Garcia-Manero G, et al. Hypomethylating agents in combination with immune checkpoint inhibitors in acute myeloid leukemia and myelodysplastic syndromes. Leukemia. 2018;32(5):1094–105.
Garcia-Manero G, Fenaux P, Al-Kali A, et al. Rigosertib versus best supportive care for patients with high-risk myelodysplastic syndromes after failure of hypomethylating drugs (ONTIME): a randomised, controlled, phase 3 trial. Lancet Oncol. 2016;17:496–508.
•• Stein EM, Fathi AT, DiNardo CD, et al. Enasidenib (AG-221), a potent oral inhibitor of mutant isocitrate dehydrogenase 2 (IDH2) enzyme, induces hematologic responses in patients with myelodysplastic syndromes (MDS). Blood. 2016;128:343 The new oral molecule enasidenib induces responses in more than one half of MDS with IDH2 mutation, which included those patients who failed to HMAs agents.
Wei A, Strickland SA, Roboz GJ, et al. Safety and efficacy of venetoclax plus low-dose cytarabine in treatment-naive patients aged ≥ 65 years with acute myeloid leukemia. Blood. 2016;128:102.
Raza A, Yousuf N, Bohkari SA, et al. In situ cell cycle kinetics in bone marrow biopsies following sequential infusions of IUdR/BrdU in patients with hematopoietic malignancies. Leuk Res. 1992;16(3):299–6.
Parcharidou A1, Raza A, Economopoulos T, et al. Extensive apoptosis of bone marrow cells as evaluated by the in situ end-labelling (ISEL) technique may be the basis for ineffective haematopoiesis in patients with myelodysplastic syndromes. Eur J Haematol. 1999;62(1):19–26.
Mundle SD, Ali A, Cartlidge JD, et al. Evidence for involvement of tumor necrosis factor-alpha in apoptotic death of bone marrow cells in myelodysplastic syndromes. Am J Hematol. 1999;60(1):36–47
Azra Raza, Peter Meyer, Diya Dutt et al. Thalidomide produces transfusion independence in long-standing refractory anemias of patients with myelodysplastic syndromes. Blood 2001 98:958–65
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
Julia Montoro declares that she has no conflict of interest.
Aslihan Yerlikaya declares that she has no conflict of interest.
Abdullah Ali declares that he has no conflict of interest.
Azra Raza has received research funding through grants from Celgene, Kura Oncology, Janssen, and Syros Pharmaceuticals and has received honoraria from Novartis for Educational lectures on MDS.
Human and Animal Rights and Informed Consent
This article does not contain any studies with human or animal subjects performed by any of the authors.
Additional information
This article is part of the Topical Collection on Leukemia
Rights and permissions
About this article
Cite this article
Montoro, J., Yerlikaya, A., Ali, A. et al. Improving Treatment for Myelodysplastic Syndromes Patients. Curr. Treat. Options in Oncol. 19, 66 (2018). https://doi.org/10.1007/s11864-018-0583-4
Published:
DOI: https://doi.org/10.1007/s11864-018-0583-4