Abstract
Background
Despite the increased uptake of intravascular lithotripsy (IVL) for treating severely calcified coronary lesions, there is limited patient-level data examining the effect of IVL on quality of life, symptomatology, and outcomes beyond 30 days. We sought to assess demographics, procedural characteristics, outcomes, and impact of IVL on patient-reported angina after a minimum of 6 months follow-up.
Methods
A retrospective single-center study was conducted of patients treated with coronary IVL between January and October 2020. Baseline demographics were obtained from electronic patient records and SYNTAX scores were calculated from index coronary angiograms. Technical success and complications were assessed along with clinical outcomes, which included all-cause mortality, myocardial infarction (MI), target lesion revascularization (TLR), and MACE (composite of death, stroke, MI, and TLR). Canadian Cardiovascular Society (CCS) angina classification was assessed at virtual clinical follow-up.
Results
Forty-seven consecutive patients were included. At a mean follow-up of 306 ± 74 days, the mean CCS angina score was reduced by 53% post-IVL-assisted PCI (2.9 vs 1.4, p < 0.001). Technical and procedural success were high (94% and 92%, respectively). One patient (2%) met the pre-specified criteria for in-hospital MACE and 4 (9%) met pre-specified MACE at follow-up, including 2 deaths and 2 TLR. Procedural complications included coronary dissection (11%) and coronary perforation (6%) and were managed either conservatively or with PCI.
Conclusions
Coronary IVL is a safe and effective adjunctive therapy for treating heavily calcified coronary lesions. This cohort shows high procedural success and a significant reduction in CCS angina at follow-up.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Background
Coronary artery calcification (CAC) is an independent predictor of major cardiovascular events and poses increased technical complexity during percutaneous coronary intervention (PCI) [1,2,3]. Heavily calcified coronary lesions may lead to challenging stent delivery, stent underexpansion and stent malapposition [4]. Consequently, stenting inadequately prepared calcific lesions may lead to peri-procedural and long-term complications such as in-stent restenosis (ISR) and stent thrombosis [4].
Established therapies for CAC include specialty balloons (scoring, cutting, ultra-high pressure), rotational atherectomy (RA), orbital atherectomy, and excimer laser atherectomy [5,6,7,8]. The Shockwave™ intravascular lithotripsy (IVL) system (Shockwave Medical, Santa Clara, CA, USA) was recently added to the armamentarium for CAC lesion preparation. Briefly, it is a balloon-based system which emits sonic pressure waves to selectively fracture calcium in the vessel wall which facilitates lesion expansion and deployment of an appropriately sized stent [9]. IVL is easy to use with a short learning curve and may have benefits over other therapies due to lower-pressure inflations reducing the risk of coronary artery dissection [4, 9]. Early experience of IVL for CAC has demonstrated efficacy with a satisfactory safety profile [4, 9, 10].
Despite the recent widespread uptake in the use of IVL, there is limited patient-level data examining the effect of IVL on quality of life, symptomatology, and clinical follow-up data beyond 30 days. In this review of IVL, we sought to assess the demographics, procedural characteristics, complications and impact of IVL on patient-reported angina scores, and follow-up of these patients beyond 6 months.
Methods
Approval for the study was granted by the hospital research and innovation department (ID: RI7215). All patients treated with the Shockwave™ intracoronary lithotripsy system at our institution between January 2020 and October 2020 were included. Electronic patient records were used to obtain patient demographics, baseline Canadian Cardiovascular Society (CCS) angina classification scores, and estimated cardiovascular risk scores using the EuroSCORE II tool [11, 12]. Index coronary angiograms were independently reviewed to calculate SYNTAX scores, evaluate procedural success, and assess procedural complications [13]. Clinical outcomes including CCS angina scores were assessed at virtual clinical follow-up a minimum of 6 months post-procedure. A Student t-test was used to detect clinical significance between pre- and post-procedure angina scores. Continuous variables are presented as mean ± standard deviation (SD) or median [Q1; Q3]. Categorical data are presented as frequencies and percentages.
Definitions
Technical success was defined as (1) ability to cross the lesion with both a wire and a balloon and successfully open the artery, (2) < 30% residual stenosis, and (3) TIMI III flow in all major branches. Target lesion revascularization (TLR) was defined as any unplanned repeat PCI of the target lesion or bypass of the target vessel performed for in-stent restenosis (ISR) or other complication of the target lesion. Major adverse cardiac events (MACE) were defined as the composite of total death, stroke, TLR, and myocardial infarction. Procedural success was defined as technical success with no in-hospital MACE.
Results
Patient characteristics (Table 1)
Forty-seven consecutive patients were included. The mean age was 69 ± 9 years, with a strong male predominance (83%). Hypertension (77%), smoking history (74%), prior PCI (72%), hyperlipidemia (72%), renal impairment (62%), and diabetes mellitus (43%) were the most common comorbidities. The median EuroSCORE II was 3.4 ± 3. Stable angina (60%) was the most common indication for intervention, followed by NSTEMI (19%) and unstable angina (17%).
Procedural characteristics (Table 2)
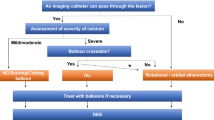
A total of 62 Shockwave™ balloons were used in 47 patients. Two balloons ruptured during inflation. The mean SYNTAX score was 26 ± 15. All patients had circumferential or near-circumferential calcium as determined by coronary angiography and/or intracoronary imaging. Overall, 57% had PCI of a de novo lesion, 32% had PCI to a previously placed underexpanded stent, and 11% had severely calcified in-stent restenosis (ISR). Most patients underwent intracoronary imaging with either intravascular ultrasound (83%) or optical coherence tomography (8%). Adjunctive therapies such as RA (17%) were employed for additional plaque modification if there was difficulty delivering the IVL balloon or if there was inadequate lesion preparation with IVL alone.
Procedural outcomes (Table 3)
Technical and procedural success were high (94% and 92%, respectively). One patient (2%) met the pre-specified criteria for in-hospital MACE due to TLR, and a total of 4 patients (9%) met the pre-specified criteria for MACE at follow-up, including 2 deaths and 2 TLR. Complications included coronary dissection (11%), which were successfully treated with either conservative measures or stenting, and coronary perforation (6%) which were treated with either prolonged balloon inflation or covered stent placement (n = 1). Notably, two of the cases of coronary perforation were attributed to high-pressure inflation with non-compliant balloons and one case was due to “wire-exit perforation” and not as a complication of the IVL balloon per se.
Ten patients had undergone had interval angiography, of whom 5 required additional stenting and 2 had repeat IVL with the Shockwave™ system. One patient had an unplanned diagnostic angiogram for ongoing symptoms but no significant residual stenosis was detected. Six patients (13%) were lost to follow-up. At a mean follow-up of 306 ± 74 days, the mean CCS angina score was reduced by 53% post-IVL-assisted PCI (2.9 vs 1.4, p < 0.001).
Discussion
Our results suggest that IVL-assisted PCI is associated with significantly reduced patient-reported angina scores, and this benefit persists beyond 6 months post-intervention. To our knowledge, this is the first IVL series to include CCS angina classification scores, which represent an important clinical indicator of quality of life in cardiac patients [14]. Our results add to the existing registries which suggest that IVL-assisted PCI with the Shockwave™ system is safe and effective in a real-world population [4, 15,16,17,18,19]. The high EuroSCORE II and SYNTAX scores observed in our cohort reflect the increased clinical and technical complexity in managing patients with severe coronary artery calcification and who are routinely encountered in clinical practice.
Procedural considerations
The Shockwave™ intracoronary system has a number of advantages over specialty balloons and atherectomy devices. As a balloon-based technology, the device does not require specific training and may be introduced via smaller French catheters in comparison to traditional atherectomy devices [20]. In our study, procedural success was achieved via radial access in the majority of patients (66%). Our procedural success rate is comparable to those seen in both the DISRUPT CAD III study (92% vs 92%, respectively) and other major registry datasets (Table 4) [4, 15,16,17,18,19]. Reasons for treatment failure/abandonment in our cohort included (i) intra-procedural ischemia with significant residual stenosis; (ii) prolonged procedural time, ischemia, and resistant stent underexpansion; and (iii) prolonged procedural time and dissection with no-reflow, in the setting of a chronic total occlusion (CTO).
Specialty balloons and atherectomy devices exert their effects through direct tissue debulking or compression; however, the IVL device generates sonic pulses at relatively low balloon inflation pressures, thereby minimizing direct mechanical pressure to the vessel wall [4, 9, 10]. In theory, this should reduce the risk of mechanical complications including dissections and perforations. In our study, coronary dissections were seen in 5 (11%) of patients treated with IVL. This is higher than the 3% dissection rate reported with RA in the ROTAXUS trial [5] and the 7% rate reported with modified balloons in the PREPARE-CALC trial [6, 7]. IVL registries report rates of dissection ranging from 0 to 36% (Table 4). Of the 5 coronary dissections in our series, 3 occurred in patients who had received adjunctive therapy with either RA or laser atherectomy devices and therefore may not be directly attributable to the use of IVL. Furthermore, the frequent use of intravascular imaging in our cohort may have allowed for the detection of more subtle dissections than those reported in previous studies. The rate of coronary perforation (6%) is higher compared to other similar registries (Table 4). The explanation for this is unclear but may be attributable to aggressive calcium modification with larger balloon sizes and high-pressure balloon inflations. The mean IVL balloon size used for calcium modification was 3.5 mm which was similar or larger compared with other similar registries [15,16,17,18,19].
In our cohort, in-hospital MACE (2%) is comparable to other registries (Table 4). However, our study found a higher rate of MACE at follow-up (6.3% at mean 10 months follow-up) than those reported by either Aziz et al. (2.6% at 7 months) [16]. These differences may relate to our definition of MACE, which included total deaths rather than cardiac deaths, as well as our longer duration of follow-up.
Two notable IVL-specific complications have been identified in post-licensing series: (i) IVL induced ventricular capture beats or “Shocktopics” were noted in 77.8% of patients treated with the Shockwave™ system in one series [21]. It has been suggested that these ectopic beats may have the potential to induce ventricular arrhythmia. While our study did not directly assess the prevalence of “Shocktopics,” no patients in our cohort developed peri-procedural ventricular arrhythmias. (ii) Several cases of Shockwave™ balloon rupture causing coronary dissection have been reported and while there were two balloon ruptures in our cohort, neither resulted in vessel dissection [22, 23].
Impact on practice
High procedural success and low complication rates suggest that this is a safe and effective adjunctive therapy for the management of CAC. The cost of IVL remains a major barrier to widespread use of the Shockwave™ system [24]. In our cohort, the median number of pulses required was 80, with a mean of 1.32 balloons used per patient treated. Further research could be garnered to evaluate the cost–benefit analysis of this technology in the context of a publicly funded healthcare system, such as the one in which this cohort is based.
Limitations
The data presented in this study should be interpreted within the inherent limitations of a single-center, retrospective, observational study. Similar to other studies of IVL, the data is not randomized or compared to a control group. Additionally, our study has a relatively high attrition rate, with 6/47 (13%) patients lost to follow-up after 6 months. This may have been related in part to the COVID-19 pandemic, during which the majority of our outpatient services were being conducted virtually. This presented challenges in reliably contacting patients, especially those referred from remote referral centers.
Conclusion
The use of coronary IVL is a safe and effective adjunctive therapy for treating heavily calcified coronary lesions. This cohort shows high procedural success with IVL and a significant reduction in CCS angina class at follow-up.
Abbreviations
- CAC:
-
Coronary artery calcification
- CABG:
-
Coronary artery bypass graft
- CCS:
-
Canadian Cardiovascular Society
- ISR:
-
In-stent restenosis
- IVL:
-
Intravascular lithotripsy
- MACE:
-
Major adverse cardiac events
- PCI:
-
Percutaneous coronary intervention
- RA:
-
Rotational atherectomy
- TIMI:
-
Thrombolysis in myocardial infarction
- TLR:
-
Target lesion revascularization
References
Ferencik M, Pencina KM, Liu T et al (2017) Coronary artery calcium distribution is an independent predictor of incident major coronary heart disease events: results from the Framingham Heart Study. Circ Cardiovasc Imaging 10(10):e006592
Madhavan MV, Tarigopula M, Mintz GS et al (2014) Coronary artery calcification: pathogenesis and prognostic implications. J Am Coll Cardiol 63(17):1703–1714
Barbato E, Shlofmitz E, Milkas A et al (2017) State of the art: evolving concepts in the treatment of heavily calcified and undilatable coronary stenoses - from debulking to plaque modification, a 40-year-long journey. EuroIntervention 13(6):696–705
Hill JM, Kereiakes DJ, Shlofmitz RA et al (2020) Disrupt CAD III Investigators. Intravascular lithotripsy for treatment of severely calcified coronary artery disease J Am Coll Cardiol 76(22):2635–2646
Forero MNT, Daemen J (2019) The coronary intravascular lithotripsy system. Interv Cardiol 14(3):174–181
Abdel-Wahab M, Richardt G, Joachim Büttner H et al (2013) High-speed rotational atherectomy before paclitaxel-eluting stent implantation in complex calcified coronary lesions: the randomized ROTAXUS (Rotational Atherectomy Prior to Taxus Stent Treatment for Complex Native Coronary Artery Disease) trial. JACC Cardiovasc Interv 6(1):10–19
Abdel-Wahab M, Toelg R, Byrne RA et al (2018) High-speed rotational atherectomy versus modified balloons prior to drug-eluting stent implantation in severely calcified coronary lesions. Circ Cardiovasc Interv 11(10):e007415
Bittl JA, Chew DP, Topol EJ, Kong DF, Califf RM (2004) Meta-analysis of randomized trials of percutaneous transluminal coronary angioplasty versus atherectomy, cutting balloon atherotomy, or laser angioplasty. J Am Coll Cardiol 43(6):936–942
Brinton TJ, Ali ZA, Hill JM et al (2019) Feasibility of shockwave coronary intravascular lithotripsy for the treatment of calcified coronary stenoses. Circulation 139(6):834–836
Ali ZA, Nef H, Escaned J et al (2019) Safety and effectiveness of coronary intravascular lithotripsy for treatment of severely calcified coronary stenoses: the Disrupt CAD II study. Circ Cardiovasc Interv 12(10):e008434
Kaul P, Naylor CD, Armstrong PW et al (2009) Assessment of activity status and survival according to the Canadian Cardiovascular Society angina classification. Can J Cardiol 25(7):e225–e231
Nashef SA, Roques F, Sharples LD et al (2012) EuroSCORE II. Eur J Cardiothorac Surg 41(4):734–744; discussion 744–745
Sianos G, Morel MA, Kappetein AP et al (2005) The SYNTAX score: an angiographic tool grading the complexity of coronary artery disease. EuroIntervention 1(2):219–227
Manolis AJ, Ambrosio G, Collins P et al (2019) Impact of stable angina on health status and quality of life perception of currently treated patients. The BRIDGE 2 survey. Eur J Intern Med 70:60–67
El Jattari H, Holvoet W, De Roeck F et al (2022) Intracoronary lithotripsy in calcified coronary lesions: a multicenter observational study. J Invasive Cardiol 34(1):E24–E31 (Epub 2021 Dec 12)
Aziz A, Bhatia G, Pitt M et al (2021) Intravascular lithotripsy in calcified-coronary lesions: a real-world observational. European multicenter study Catheter Cardiovasc Interv 98(2):225–235
Sinclair H, Fan L, Fahy E et al (2021) Intravascular imaging-guided intracoronary lithotripsy: first real-world experience. Health Sci Rep 4(3):e307
Umapathy S, Keh YS, Wong N et al (2021) Real-world experience of coronary intravascular lithotripsy in an Asian population: a retrospective, observational, single-center, all-comers registry. J Invasive Cardiol 33(6):E417–E424 (Epub 2021 Apr 13)
Aksoy A, Salazar C, Becher MU et al (2019) Intravascular lithotripsy in calcified coronary lesions: a prospective, observational, multicenter registry. Circ Cardiovasc Interv 12(11):e008154. https://doi.org/10.1161/CIRCINTERVENTIONS.119.008154. Epub 2019 Nov 11
Sgueglia GA, Gioffrè G, Piccioni F, Gaspardone A (2019) Slender distal radial five French coronary shockwave lithotripsy. Catheter Cardiovasc Interv 94(3):395–398
Wilson SJ, Spratt JC, Hill J et al (2020) Incidence of “shocktopics” and asynchronous cardiac pacing in patients undergoing coronary intravascular lithotripsy. EuroIntervention 15(16):1429–1435
Soriano F, Veas N, Piccinelli E, Oreglia J (2019) Coronary dissection due to intravascular lithoplasty balloon rupture. EuroIntervention 15(6):e558–e559. https://doi.org/10.4244/EIJ-D-19-00383
Lee TJ, Wan Rahimi WFB, Low MY, Nurruddin AA (2021) Type E coronary artery dissection caused by intravascular lithotripsy balloon rupture; vessel anatomy and characteristics in a lithoplasty complication case as detailed by optical coherence tomography: a case report. Eur Heart J Case Rep 5(12):ytab432
Kassimis G, Ziakas A, Didagelos M et al (2021) Shockwave coronary intravascular lithotripsy system for heavily calcified de novo lesions and the need for a cost-effectiveness analysis. Cardiovasc Revasc Med S1553–8389(21):00490–00495
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Buckley, A.J., McCormick, J.P., Carey, J. et al. Intravascular lithotripsy-assisted PCI for severely calcified coronary lesions: evaluating the impact on quality of life and outcomes. Ir J Med Sci 192, 1085–1090 (2023). https://doi.org/10.1007/s11845-022-03077-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11845-022-03077-9