Abstract
Purpose
The present study aimed to quantify the level of sleep problems in 5835 breast, prostate, and colorectal cancer survivors, and explore a number of potential determinants of poor sleep quality in the present sample. BMI, diet, and physical activity were of particular interest as potential determinants.
Methods
Participants who completed the ‘Health and Lifestyle after Cancer’ survey were adults who had been diagnosed with breast, prostate, or colorectal cancer (mean time since cancer diagnosis was 35.5 months, SD=13.56). Sleep quality was assessed using the Pittsburgh Sleep Quality Index. BMI was calculated from self-reported height and weight. Participants were categorised as meeting/not meeting the World Cancer Research Fund (WCRF) recommendations for fibre, fruit and vegetables, added sugar, red meat, processed meat, fat, alcohol, and physical activity. Analyses accounted for demographic and clinical factors.
Results
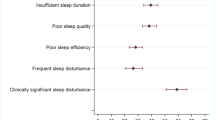
Fifty-seven percent of those with sleep data were classified as poor sleepers (response rate 79%). Being female, having a higher number of cancer treatments, more comorbid conditions, and being more anxious/depressed increased the odds of being a poor sleeper. After adjustment for these factors, there were no associations between diet/alcohol/physical activity and sleep. However, BMI was associated with sleep. Individuals in the overweight and obese categories had 22% and 79% higher odds of being poor sleepers than individuals in the underweight/healthy weight category, respectively.
Conclusions
The findings suggest that there may be a need to develop sleep quality interventions for cancer survivors with obesity. Even after adjustment for multiple clinical and demographic factors, BMI (particularly obesity) was associated with poor sleep. Thus, researchers and health professionals should find ways to support individuals with overweight and obesity to improve their sleep quality.
Implications for Cancer Survivors
The present findings highlight that poor sleep is a common issue in cancer survivors. Interventions seeking to improve outcomes for cancer survivors over the longer term should consider sleep quality.
Similar content being viewed by others
Data availability
The datasets generated during and/or analysed during the current study are available from the corresponding author on reasonable request.
Notes
Items 5d and 5j and 10a–e of the PSQI were omitted for the present survey and scoring was adjusted accordingly.
References
Office for National Statistics. Cancer registration statistics, England: 2017. London: Office for National Statistics; 2019.
Mourouti N, Panagiotakos DB, Kotteas EA, Syrigos KN. Optimizing diet and nutrition for cancer survivors: a review. Maturitas. 2017;105:33–6. https://doi.org/10.1016/j.maturitas.2017.05.012.
Cancer Research UK. Cancer survival statisitcs. Retrieved from: https://www.cancerresearchuk.org/health-professional/cancer-statistics/survival#heading-Zero . Accessed 15 January 2021.
WCRF, AICR. Diet, physical activity and cancer: a global perspective. Continuous Update Project Expert Report 2018. 2019. Retrieved from:https://www.ietandcancerreport.org. Accessed 1 Mar 2021.
Liska TM, Kolen AM. The role of physical activity in cancer survivors’ quality of life. Health Qual Life Outcomes. 2020;18:197. https://doi.org/10.1186/s12955-020-01448-3.
Oberguggenberger A, Meraner V, Sztankay M, Hilbert A, Hubalek M, et al. Health behavior and quality of life outcomes in breast cancer survivors: prevalence rates and predictions. Clin Breast Cancer. 2018;18:38–44. https://doi.org/10.1016/j.clbc.2017.09.008.
Slade AN, Waters MR, Serrano NA. Long-term sleep disturbance and prescription sleep aid use among cancer survivors in the United States. Support Care Cancer. 2019;28(2):551–60. https://doi.org/10.1007/s00520-019-04849-3.
Harrington CB, Hansen JA, Moskowitz M, Todd BL, Feuerstein M. It’s not over when it’s over: long-term symptoms in cancer survivors—a systematic review. Int J Psychiatry Med. 2010;40(2):163–81. https://doi.org/10.2190/PM.40.2.c.
Baker F, Denniston M, Smith T, West MM. Adult cancer survivors: how are they faring? Cancer. 2005;104(11):2565–76. https://doi.org/10.1002/cncr.21488.
Lowery-Allison AE, Passik SD, Cribbet MR, Reinsel RA. Sleep problems in breast cancer survivors 1-10 years posttreatment. Palliat Support Care. 2018;16(3):325–34. https://doi.org/10.1017/S1478951517000311.
Rutherford C, Müller F, Faiz N, King MT, White K. Patient-reported outcomes and experiences from the perspective of colorectal cancer survivors: meta-synthesis of qualitative studies. JPRO. 2020;4:27. https://doi.org/10.1186/s41687-020-00195-9.
Sokol RL, Grummon AH, Lytle LA. Sleep duration and body mass: direction of associations from adolescence to young adulthood. Int J Obes. 2019;44:852–6. https://doi.org/10.1038/s41366-019-0462-5.
De Sousa AGP, Cercato C, Mancini MC, Halpern A. Obesity and obstructive sleep apnea-hypopnea syndrome. Obes Rev. 2008;9:340–54. https://doi.org/10.1111/j.1467-789X.2008.00478.x.
Romero-Corral A, Caples SM, Lopez-Jimenez F, Somers VK. Interactions between obesity and obstructive sleep apnea. Implications for treatment. Chest. 2010;137(3):711–9. https://doi.org/10.1378/chest.09-0360.
Magee L, Hale L. Longitudinal associations between sleep duration and subsequent weight gain: a systematic review. Sleep Med Rev. 2013;16(3):231–41. https://doi.org/10.1016/j.smrv.2011.05.005.
Klyushnenkova EN, Sorkin JD, Gallicchio L. Association of obesity and sleep problems among breast cancer survivors: results from a registry-based survey study. Support Care Cancer. 2015;23(12):3437–45. https://doi.org/10.1007/s00520-015-2692-y.
Greer SM, Goldstein AN, Walker MP. The impact of sleep deprivation on food desire in the human brain. Nat Commun. 2014;4:2259. https://doi.org/10.1038/ncomms3259.
Muscogiuri G, Barrea L, Aprano S, Framondi L, Matteo RD, et al. Sleep quality in obesity: does adherence to the Mediterranean diet matter? Nutrients. 2020;12(5):1364. https://doi.org/10.3390/nu12051364.
Lindseth G, Murray A. Dietary macronutrients and sleep. West J Nurs Res. 2016;38(8):938–58. https://doi.org/10.1177/0193945916643712.
Kechribari I, Kontogianni MD, Georgoulis M, Lamprou K, Mourati I, Vagiakis E, Yiannakouris N. Associations between red meat intake and sleep parameters in patients with obstructive sleep apnea. J Acad Nutr Diet. 2020;120(6):1042–53.
Kim N-H, Song S, Jung S-Y, Lee E, Kim Z, et al. Dietary patterns and health-related quality of life among breast cancer survivors. BMC Womens Health. 2018;18(1):1–10. https://doi.org/10.1186/s12905-018-0555-7.
Zheng D, Yuan X, Ma C, Liu Y, VanEvery H, et al. Alcohol consumption and sleep quality: a community-based study. Public Health Nutr 2020;15:4851–4858. https://doi.org/10.1017/S1368980020004553
Mercier J, Savard J, Bernard P. Exercise interventions to improve sleep in cancer patients: a systematic review and meta-analysis. Sleep Med Rev. 2017;36:43–56. https://doi.org/10.1016/j.smrv.2016.11.001.
Beeken RJ, Croker H, Heinrich M, Heinrich M, Smith L, et al. Study protocol for a randomised controlled trial of brief, habit-based, lifestyle advice for cancer survivors: exploring behavioural outcomes for the Advancing Survivorship Cancer Outcomes Trial (ASCOT). BMJ Open. 2016;6:e011646. https://doi.org/10.1136/bmjopen-2016-011646.
Buysse DJ, Reynolds CF III, Monk TH, Berman SR, Kupfer DJ. The Pittsburgh sleep quality index: a new instrument for psychiatric practice and research. Psychiatry Res. 1989;2:193–213. https://doi.org/10.1016/0165-1781(89)90047-4.
Mollayeva T, Thurairajah P, Burton K, Mollayeva S, Shapiro CM, et al. The Pittsburgh sleep quality index as a screening tool for sleep dysfunction in clinical and non-clinical samples: a systematic reviewand meta-analysis. Sleep Med Rev. 2016;25:52–73. https://doi.org/10.1016/j.smrv.2015.01.009.
Roe L, Strong C, Whiteside C, Neil M, Mant D. Dietary intervention in primary care: validity of the DINE method for diet assessment. Fam Pract. 1994;11:375–81. https://doi.org/10.1093/fampra/11.4.375.
McGowan L, Cooke LJ, Gardner B, Beeken RJ, et al. Healthy feeding habits: efficacy results from a cluster-randomized, controlled exploratory trial of a novel, habit-based intervention with parents. Am J Clin Nutr. 2013;98(3):769–77. https://doi.org/10.3945/ajcn.112.052159.
Cappuccio FP, Rink E, Perkins-Porras L, McKay C, Hilton S, et al. Estimation of fruit and vegetable intake using a two-item dietary questionnaire: a potential tool for primary health care workers. NMCD. 2003;13:12–9. https://doi.org/10.1016/s0939-4753(03)80163-1.
Bush K, Kivlahan DR, McDonell MB, Fihn SD, Bradley KA, et al. The AUDIT alcohol consumption questions (AUDIT-C): an effective brief screening test for problem drinking. Arch Intern Med. 1998;158(16):1789–95. https://doi.org/10.1001/archinte.158.16.1789.
Godin G, Shephard RJ. A simple method to assess exercise behavior in the community. Can J Appl Sport Sci. 1985;10:141–6.
Amireault S, Godin G, Lacombe J. The use of the Godin-Shephard Leisure-Time Physical Activity Questionnaire in oncology research: a systematic review. BMC Med Res Methodol. 2015;15:60. https://doi.org/10.1186/s12874-015-0045-7.
Department of Health and Social Care. UK chief medical officer’s low risk drinking guidelines. 2016. Retrieved from: https://www.gov.uk/government/publications/alcohol-consumption-advice-on-low-risk-drinking. Accessed 1 Mar 2021.
Downing A, Morris EJA, Richards M, Corner J, Wright P, et al. Health-related quality of life after colorectal cancer in England: a patient-reported outcomes study of individuals 12 to 36 months after diagnosis. J Clin Oncol. 2015;33(6):616–24. https://doi.org/10.1200/JCO.2014.56.6539.
EuroQol Group. EuroQol — a new facility for the measurement of health-related quality of life. Health Policy. 1990;16:199–208. https://doi.org/10.1016/0168-8510(90)90421-9.
Pickard AS, Leon MCD, Kohlmann T, Cella D, Rosenbloom SK. Psychometric comparison of the standard EQ-5D to a 5 level version in cancer patients. Med Care. 2007;45(3):259–63. https://doi.org/10.1097/01.mlr.0000254515.63841.81.
Bursac Z, Gauus CH, Williams DK, Hosmer DW. Purposeful selection of variables in logistic regression. Source Code Biol Med. 2008;3:17. https://doi.org/10.1186/1751-0473-3-17.
He Y. Missing data analysis using multiple imputation. Getting to the heart of the matter. Circ. Cardiovasc. Qual. Outcomes. 2010;3(1):98–105. https://doi.org/10.1161/CIRCOUTCOMES.109.875658.
Nock NL, Dimitropoulos A, Zanotti KM, Waggoner S, Nagel C, et al. Sleep, quality of life, and depression in endometrial cancer survivors with obesity seeking weight loss. Support Care Cancer. 2020;28:2311–9. https://doi.org/10.1007/s00520-019-05051-1.
Mitchell LJ, Davidson ZE, Bonham M, O’Driscoll DM, Hamilton GS, et al. Weight loss from lifestyle interventions and severity of sleep apnoea: a systematic review and meta-analysis. Sleep Med. 2014;15(10):1173–83. https://doi.org/10.1016/j.sleep.2014.05.012.
Thomson CA, Morrow KL, Flatt SW, Wertheim BC, Perfect MM, et al. Relationship between sleep quality and quantity and weight loss in women participating in a weight-loss intervention trial. Obesity. 2012;20(7):1419–25. https://doi.org/10.1038/oby.2012.62.
Demark-Wahnefried W, Schmitz KH, Alfano CM, Bail JR, Goodwin PJ, et al. Weight management and physical activity throughout the cancer care continuum. CA: A Cancer J Clin. 2017;68(1):64–89.
Dong Z, Xu X, Wang C, Cartledge S, Maddison R, et al. Association of overweight and obesity with obstructive sleep apnoea: a systematic review and meta-analysis. Obes Med. 2020;17:100185. https://doi.org/10.1016/j.obmed.2020.100185.
Schoeller DA. How accurate is self-reported dietary energy intake? Nutr Rev. 1990;48(10):373–9. https://doi.org/10.1111/j.1753-4887.1990.tb02882.x.
Macdiarmid J, Blundell J. Assessing dietary intake: who, what and why of under-reporting. Nutr Res Rev. 1998;11:231–53. https://doi.org/10.1079/NRR19980017.
Ravelli MN, Schoeller DA. Traditional self-reported dietary instruments are prone to inaccuracies and new approaches are needed. Front Nutr. 2020;7:90. https://doi.org/10.3389/fnut.2020.00090.
Du H, Bennett D, Li L, Whitlock G, Guo Y, et al. Physical activity and sedentary leisure time and their associations with BMI, waist circumference, and percentage body fat in 0.5 million adults: the China Kadoorie Biobank study. Am. J Clin Nutr. 2012;97:487–96. https://doi.org/10.3945/ajcn.112.046854.
Rogers L, Courneya K, Oster R, Anton P, Robbs RS, et al. Physical activity and sleep in breast cancer survivors. A randomized trial. Med Sci Sports Exerc. 2017;49(10):2009–15. https://doi.org/10.1249/MSS.0000000000001327.
Cerin E, Cain KL, Oyeyemi AL, Owen N, Conway TL, et al. Correlates of agreement between accelerometry and self-reported physical activity. Med Sci Sports Exerc. 2016;48(6):1075–84. https://doi.org/10.1249/MSS.0000000000000870.
Phillips KM, Jim HS, Donovan KA, Pinder-Schenck MC, Jacobsen PB. Characteristics and correlates of sleep disturbances in cancer patients. Support Care Cancer. 2011;20:357–65. https://doi.org/10.1007/s00520-011-1106-z.
Irwin MR, Olmstead RE, Ganz PA, Haque R. Sleep disturbance, inflammation and depression risk in cancer survivors. Brain, behave. Immune. 2013;30:s58–67. https://doi.org/10.1016/j.bbi.2012.05.002.
Liu R-Q, Bloom MS, Wu Q-Z, He Z-Z, Qian Z, et al. Association between depressive symptoms and poor sleep quality among Han and Manchu ethnicities in a large, rural, Chinese population. PLoS One. 2019;14(12):e0226562. https://doi.org/10.1371/journal.pone.0226562.
Ramsawh HJ, Stein MB, Belik S-L, Jacobi F, Sareen J. Relationship of anxiety disorders, sleep quality, and functional impairment in a community sample. J Psychiatr Res. 2009;43(10):926–33. https://doi.org/10.1016/j.jpsychires.2009.01.009.
Zhang B, Wing Y-K. Sex differences in insomnia: a meta-analysis. Sleep. 2006;29(1):85–93. https://doi.org/10.1093/sleep/29.1.85.
Otte JL, Carpenter JS, Russell KM, Bigatti S, Champion VL. Prevalence, severity, and correlates of sleep-wake disturbances in long-term breast cancer survivors. J Pain Symptom Manag. 2010;39(3):535–47. https://doi.org/10.1016/j.jpainsymman.2009.07.004.
Martire VL, Caruso D, Palagini L, Zoccoli G, Bastianini S. Stress and sleep: a relationship lasting a life time. Neurosci Biobehav Rev. 2019;117:65–77. https://doi.org/10.1016/j.neubiorev.2019.08.024.
Hoyt MA, Bower JE, Irwin MR, Weierich MR, Stanton AL. Sleep quality and depressive symptoms after prostate cancer: the mechanistic role of cortisol. Behav Neurosci. 2016;130(3):351–6. https://doi.org/10.1037/bne0000107.
Davies A, Wellard-Cole L, Rangan A, Allman-Farinelli M. Validity of self-reported weight and height for BMI classification: a cross-sectional study among young adults. Nutr. 2020;71:110622. https://doi.org/10.1016/j.nut.2019.110622.
Yorkin M, Spaccarotella K, Martin-Biggers J, Quick V, Byrd-Bredbenner C. Accuracy and consistency of weights provided by home bathroom scales. BMC Public Health. 2013;13:1194. https://doi.org/10.1186/1471-2458-13-1194.
Acknowledgements
The authors would like to thank Cancer Research UK for funding the Advancing Survivorship Cancer Outcomes Trial (ASCOT) [22], from which the survey data used in the present study was obtained. The authors also wish to thank the NHS trusts who helped recruit participants and the participants for taking the time to complete the measures.
Funding
ASCOT was funded by Cancer Research UK (grant numbers C43975/A27498, C1418/A14133).
Author information
Authors and Affiliations
Contributions
SA, RC, PL, AR, HC, RB, and AF designed the study; RC, AR, and HC collected the data; SA, RC, and AF analysed the data; SA, RC, PL, RB, and AF interpreted the data. SA drafted the manuscript, and RC, AF, PL, RB, AR, and HC revised it critically for intellectual content and finally approved the version to be submitted.
Corresponding author
Ethics declarations
Ethics approval
Ethical approval was obtained through the National Research Ethics Service Committee South Central—Oxford B (reference number 14/SC/1369).
Consent to participate and consent for publication
The following statement was provided on the questionnaire: ‘By completing this questionnaire you are consenting to your anonymous information being used for research on lifestyle in people diagnosed with cancer’.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rebecca J Beeken and Abigail Fisher are joint senior authors.
Supplementary information
ESM 1
(PDF 446 kb)
Rights and permissions
About this article
Cite this article
Aronsen, S., Conway, R., Lally, P. et al. Determinants of sleep quality in 5835 individuals living with and beyond breast, prostate, and colorectal cancer: a cross-sectional survey. J Cancer Surviv 16, 1489–1501 (2022). https://doi.org/10.1007/s11764-021-01127-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11764-021-01127-2