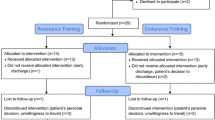
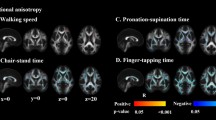
Abstract
Bilateral coordination of the lower extremities is an essential component of mobility. The corpus callosum bridges the two hemispheres of the brain and is integral for the coordination of such complex movements. The aim of this project was to assess structural integrity of the transcallosal sensorimotor fiber tracts and identify their associations with gait coordination using novel methods of ecologically valid mobility assessments in persons with multiple sclerosis and age−/gender-matched neurotypical adults. Neurotypical adults (n = 29) and persons with multiple sclerosis (n = 27) underwent gait and diffusion tensor imaging assessments; the lower limb coordination via Phase Coordination Index, and radial diffusivity, an indirect marker of myelination, were applied as the primary outcome measures. Persons with multiple sclerosis possessed poorer transcallosal white matter microstructural integrity of sensorimotor fiber tracts compared to the neurotypical adults. Further, persons with multiple sclerosis demonstrated significantly poorer bilateral coordination of the lower limbs during over-ground walking in comparison to an age and gender-matched neurotypical cohort. Finally, bilateral coordination of the lower limbs was significantly associated with white matter microstructural integrity of the dorsal premotor and primary motor fiber bundles in persons with multiple sclerosis, but not in neurotypical adults. This analysis revealed that persons with multiple sclerosis exhibit poorer transcallosal microstructural integrity than neurotypical peers. Furthermore, these structural deficits were correlated to poorer consistency and accuracy of gait in those with multiple sclerosis. Together, these results, emphasize the importance of transcallosal communication for gait coordination in those with multiple sclerosis.



Similar content being viewed by others

Data availability
Not applicable.
Code availability
Not applicable.
References
Basser, P. J., & Pierpaoli, C. (1996). Microstructural and physiological features of tissues elucidated by quantitative-diffusion-tensor MRI. Journal of Magnetic Resonance. Series B, 111(3), 209–219.
Beaulieu, C. (2011). What makes diffusion anisotropic in the nervous system? In D. K. Jones (Ed.), Diffusion MRI: Theory, methods, and applications (pp. 92–109). Oxford University Press.
Boisgontier, M. P., Wittenberg, G. F., Fujiyama, H., Levin, O., & Swinnen, S. P. (2014). Complexity of central processing in simple and choice multilimb reaction-time tasks. PLoS One, 9(2), e90457. https://doi.org/10.1371/journal.pone.0090457
Bonzano, L., Tacchino, A., Roccatagliata, L., Abbruzzese, G., Mancardi, G. L., & Bove, M. (2008). Callosal contributions to simultaneous bimanual finger movements. The Journal of Neuroscience, 28(12), 3227–3233. https://doi.org/10.1523/JNEUROSCI.4076-07.2008
Bowen, J., Gibbons, L., Gianas, A., & Kraft, G. H. (2001). Self-administered expanded disability status scale with functional system scores correlates well with a physician-administered test. Multiple Sclerosis, 7, 201–206.
Chouinard, P. A., & Paus, T. (2006). The primary motor and premotor areas of the human cerebral cortex. Neuroscientist, 12(2), 143–152. https://doi.org/10.1177/1073858405284255
Cohen, J. A., & Alexander, R. (2012). Hankbook of multiple sclerosis (2nd ed.). Springer Healthcare.
Comber, L., Galvin, R., & Coote, S. (2017). Gait deficits in people with multiple sclerosis: A systematic review and meta-analysis. Gait & Posture, 51, 25–35. https://doi.org/10.1016/j.gaitpost.2016.09.026
Cook, R. D. (1977). Detection of influential observations in linear regression. Technometrics, 19(15–18).
Croft, J. L., Schroeder, R. T., & Bertram, J. E. A. (2019). The landscape of movement control in locomotion: Cost, strategy, and solution. Frontiers in Psychology, 10, 716. https://doi.org/10.3389/fpsyg.2019.00716
Debaere, F., Wenderoth, N., Sunaert, S., Van Hecke, P., & Swinnen, S. P. (2004). Cerebellar and premotor function in bimanual coordination: Parametric neural responses to spatiotemporal complexity and cycling frequency. Neuroimage, 21(4), 1416–1427. https://doi.org/10.1016/j.neuroimage.2003.12.011
Dilokthornsakul, P., Valuck, R. J., Nair, K. V., Corboy, J. R., Allen, R. R., & Campbell, J. D. (2016). Multiple sclerosis prevelence in the United States commercially insured population. Neurology, 86, 1014–1021.
Fling, B. W., & Seidler, R. D. (2012). Fundamental differences in callosal structure, neurophysiologic function, and bimanual control in young and older adults. Cerebral Cortex, 22(11), 2643–2652. https://doi.org/10.1093/cercor/bhr349
Franchignoni, F., Horak, F., Godi, M., Nardone, A., & Giordano, A. (2010). Using psychometric techniques to improve the balance evaluation systems test: The mini-BESTest. Journal of Rehabilitation Medicine, 42(4), 323–331. https://doi.org/10.2340/16501977-0537
Franchignoni, F., Godi, M., Guglielmetti, S., Nardone, A., & Giordano, A. (2015). Enhancing the usefulness of the Mini-BESTest for measuring dynamic balance: A Rasch validation study. European Journal of Physical and Rehabilitation Medicine, 51(4), 429–437.
Fujiyama, H., Van Soom, J., Rens, G., Cuypers, K., Heise, K. F., Levin, O., & Swinnen, S. P. (2016). Performing two different actions simultaneously: The critical role of interhemispheric interactions during the preparation of bimanual movement. Cortex, 77, 141–154. https://doi.org/10.1016/j.cortex.2016.02.007
Gazzaniga, M. S. (2000). Cerebral specialization and interhemispheric communication: Does the corpus callosum enable the human condition. Brain, 123, 1293–1326.
Godi, M., Franchignoni, F., Caligari, M., Giordano, A., Turcato, A. M., & Nardone, A. (2013). Comparison of reliability, validity, and responsiveness of the mini-BESTest and berg balance scale in patients with balance disorders. Physical Therapy, 93(2), 158–167. https://doi.org/10.2522/ptj.20120171
Goldenberg, M. M. (2012). Multiple sclerosis review. P & T: a peer-reviewed journal for formulary management, 37(3), 175–184.
Gooijers, J., & Swinnen, S. P. (2014). Interactions between brain structure and behavior: The corpus callosum and bimanual coordination. Neuroscience and Biobehavioral Reviews, 43, 1–19. https://doi.org/10.1016/j.neubiorev.2014.03.008
Gooijers, J., Caeyenberghs, K., Sisti, H. M., Geurts, M., Heitger, M. H., Leemans, A., & Swinnen, S. P. (2013). Diffusion tensor imaging metrics of the corpus callosum in relation to bimanual coordination: Effect of task complexity and sensory feedback. Human Brain Mapping, 34(1), 241–252. https://doi.org/10.1002/hbm.21429
Gulani, V., Webb, A. G., Duncan, I. D., & Lauterbur, P. C. (2001). Apparent diffusion tensor measurements in myelin-deficient rat spinal cords. Magnetic Resonance in Medicine, 45(2), 191–195 Retrieved from http://www.ncbi.nlm.nih.gov/pubmed/11180424
Han, S. H., Kim, C. O., Kim, K. J., Jeon, J., Chang, H., Kim, E. S., & Park, H. (2019). Quantitative analysis of the bilateral coordination and gait asymmetry using inertial measurement unit-based gait analysis. PLoS One, 14(10), e0222913. https://doi.org/10.1371/journal.pone.0222913
Hoyt, D. F., & Taylor, C. R. (1981). Gait and the energetics of locomotion in horses. Nature, 292(16), 239–240.
Hughlings, J. (1958). On the anatomical and physiological localization of movements in the brain (J. Taylor Ed.). : Basic Books.
Inglese, M., & Bester, M. (2010). Diffusion imaging in multiple sclerosis: Research and clinical implications. NMR in Biomedicine, 23(7), 865–872. https://doi.org/10.1002/nbm.1515
Jacobs, J. V., & Kasser, S. L. (2012). Balance impairment in people with multiple sclerosis: Preliminary evidence for the balance evaluation systems test. Gait & Posture, 36(3), 414–418. https://doi.org/10.1016/j.gaitpost.2012.03.026
Kalron, A. (2016). Gait variability across the disability spectrum in people with multiple sclerosis. Journal of the Neurological Sciences, 361, 1–6. https://doi.org/10.1016/j.jns.2015.12.012
Kurtzke, J. F. (1983). Rating neurologic impairment in multiple sclerosis: An expanded disability status scale (EDSS). Neurology, 33, 1444–1452.
Laidet, M., Herrmann, F. R., Armand, S., Assal, F., Lalive, P. H., & Allali, G. (2017). Does fear of falling predict gait variability in multiple sclerosis? Journal of the Neurological Sciences, 380, 212–214. https://doi.org/10.1016/j.jns.2017.07.034
Larocca, N. (2011). Impact of walking impairment in multiple sclerosis: Perspectives of patients and care partners. The Patient: Patient-Centered Outcomes Research, 4(3), 189–201.
Leddy, A. L., Crowner, B. E., & Earhart, G. M. (2011). Utility of the Mini-BESTest, BESTest, and BESTest sections for balance assessments in individuals with Parkinson disease. Journal of Neurologic Physical Therapy, 35(2), 90–97. https://doi.org/10.1097/NPT.0b013e31821a620c
Leemans, A., Jeurissen, B., & Sijbers, J. (2009). ExploreDTI: a graphical toolbox for processing, analyzing, and visualizing diffusion MR. Paper presented at the 17th Annual Meeting of Intl Soc Mag Reson Med., Hawaii, USA.
Levine, D., Richards, J., & Whittle, M. W. (2012). Whittle’s gait analysis (Fifth ed.). Elsevier Ltd..
McNeill Alexander, R. (2002). Energetics and optimization of human walking and running: The 2000 Raymond pearl memorial lecture. American Journal of Human Biology, 14(5), 641–648. https://doi.org/10.1002/ajhb.10067
Mirelman, A., Shema, S., Maidan, I., & Hausdorff, J. M. (2018). Gait. Handbook of Clinical Neurology, 159, 119–134. https://doi.org/10.1016/B978-0-444-63916-5.00007-0
Peterson, E. W., Cho, C. C., von Koch, L., & Finlayson, M. L. (2008). Injurious falls among middle aged and older adults with multiple sclerosis. Archives of Physical Medicine and Rehabilitation, 89(6), 1031–1037. https://doi.org/10.1016/j.apmr.2007.10.043
Plotnik, M., Giladi, N., & Hausdorff, J. M. (2007). A new measure for quantifying the bilateral coordination of human gait: Effects of aging and Parkinson's disease. Experimental Brain Research, 181(4), 561–570. https://doi.org/10.1007/s00221-007-0955-7
Plotnik, M., Giladi, N., & Hausdorff, J. M. (2008). Bilateral coordination of walking and freezing of gait in Parkinson's disease. The European Journal of Neuroscience, 27(8), 1999–2006. https://doi.org/10.1111/j.1460-9568.2008.06167.x
Plotnik, M., Giladi, N., & Hausdorff, J. M. (2009). Bilateral coordination of gait and Parkinson's disease: The effects of dual tasking. Journal of Neurology, Neurosurgery, and Psychiatry, 80(3), 347–350. https://doi.org/10.1136/jnnp.2008.157362
Pokryszko-Dragan, A., Banaszek, A., Nowakowska-Kotas, M., Jezowska-Jurczyk, K., Dziadkowiak, E., Gruszka, E., ... Bladowska, J. (2018). Diffusion tensor imaging findings in the multiple sclerosis patients and their relationships to various aspects of disability. Journal of Neuroscience, 391, 127–133.
Richmond, S. B., & Fling, B. W. (2019). Transcallosal control of bilateral actions. Exercise and Sport Sciences Reviews, 47(4), 251–257. https://doi.org/10.1249/JES.0000000000000202
Richmond, S. B., Swanson, C. W., Peterson, D. S., & Fling, B. W. (2020). A temporal analysis of bilateral gait coordination in people with multiple sclerosis. Multiple Sclerosis and Related Disorders, 45. https://doi.org/10.1016/j.msard.2020.102445
Ruddy, K. L., Leemans, A., & Carson, R. G. (2017). Transcallosal connectivity of the human cortical motor network. Brain Structure & Function, 222(3), 1243–1252. https://doi.org/10.1007/s00429-016-1274-1
Salkind, N. J. (2017). Statistics for people who (think they) hate statistics (6th ed.). SAGE Publications.
Serbruyns, L., Gooijers, J., Caeyenberghs, K., Meesen, R. L., Cuypers, K., Sisti, H. M., ... Swinnen, S. P. (2015). Bimanual motor deficits in older adults predicted by diffusion tensor imaging metrics of corpus callosum subregions. Brain Structure & Function, 220(1), 273–290. https://doi.org/10.1007/s00429-013-0654-z.
Shah, V. V., McNames, J., Mancini, M., Carlson-Kuhta, P., Spain, R. I., Nutt, J. G., ... Horak, F. B. (2020). Quantity and quality of gait and turning in people with multiple sclerosis, Parkinson's disease and matched controls during daily living. Journal of Neurology, 267(4), 1188–1196. https://doi.org/10.1007/s00415-020-09696-5.
Socie, M. J., & Sosnoff, J. J. (2013). Gait variability and multiple sclerosis. Multiple Sclerosis International, 2013, 645197. https://doi.org/10.1155/2013/645197
Solopova, I. A., Selionov, V. A., Kazennikov, O. V., & Ivanenko, Y. P. (2014). Effects of transcranial magnetic stimulation during voluntary and non-voluntary stepping movements in humans. Neuroscience Letters, 579, 64–69. https://doi.org/10.1016/j.neulet.2014.07.015
Sparrow, W. A., & Newell, K. M. (1998). Metabolic energy expenditure and the regulation of movement economy. Psychonomic Bulletin & Review, 5, 173–196.
Swanson, C. W., & Fling, B. W. (2018). Associations between gait coordination, variability and motor cortex inhibition in young and older adults. Experimental Gerontology, 113, 163–172. https://doi.org/10.1016/j.exger.2018.10.002
Tournier, J. D., Calamante, F., & Connelly, A. (2007). Robust determination of the fibre orientation distribution in diffusion MRI: Non-negativity constrained super-resolved spherical deconvolution. Neuroimage, 35(4), 1459–1472. https://doi.org/10.1016/j.neuroimage.2007.02.016
van Emmerik, R. E., Jones, S. L., Busa, M. A., Remelius, J. G., & Averill, J. L. (2014). Enhancing postural stability and adaptability in multiple sclerosis. Advances in Experimental Medicine and Biology, 826, 251–276. https://doi.org/10.1007/978-1-4939-1338-1_15
Van Hecke, W., Emsell, L., & Sunaert, S. (Eds.). (2016). Diffusion tensor imaging: A practical handbook (1st ed.). Springer Science+Buisness Media.
Vanderah, T. W., & Gould, D. J. (2016). Nolte's the human brain: An introduction to its functional anatomy (7th ed.). Elsevier.
Wahl, M., Hubers, A., Lauterbach-Soon, B., Hattingen, E., Jung, P., Cohen, L. G., & Ziemann, U. (2011). Motor callosal disconnection in early relapsing-remitting multiple sclerosis. Human Brain Mapping, 32(6), 846–855. https://doi.org/10.1002/hbm.21071
Winter, D. A., Patla, A. E., & Frank, J. S. (1990). Assessment of balance control in humans. Medical Progress through Technology, 16, 31–51.
Wise, S. P. (1985). The primate premotor cortex: Past, present, and preparatory. Annual Review of Neuroscience (Palo Alto, CA), 8, 1–19.
Xu, L., Chang, S. H., Yang, L., & Zhang, L. J. (2020). Quantitative evaluation of callosal abnormalities in relapsing-remitting multiple sclerosis using diffusion tensor imaging: A systemic review and meta-analysis. Clinical Neurology and Neurosurgery, 201, 106442. https://doi.org/10.1016/j.clineuro.2020.106442
Zivari Adab, H., Chalavi, S., Monteiro, T. S., Gooijers, J., Dhollander, T., Mantini, D., & Swinnen, S. P. (2020). Fiber-specific variations in anterior transcallosal white matter structure contribute to age-related differences in motor performance. Neuroimage, 209, 116530. https://doi.org/10.1016/j.neuroimage.2020.116530
Acknowledgements
The authors would like to thank, Clayton W. Swanson, Tyler T. Whittier, Arianna D. Odom, Moriah R. Hanson, Patrick G. Monaghan, and Andrew S. Monaghan for their assistance in the acquisition of data for this project.
Funding
This work was supported by the Dana Foundation and the National Multiple Sclerosis Society [PP-1708-29077] that facilitated the research presented in this manuscript.
Author information
Authors and Affiliations
Contributions
Author contributions included conception and study design (SBR, DSP, BWF), data collection or acquisition (SBR), statistical analysis (SBR), interpretation of results (SBR, DSP, BWF), drafting the manuscript work or revising it critically for important intellectual content (SBR, DSP, BWF) and approval of final version to be publishedand agreement to be accountable for the integrity and accuracy of all aspects of the work (SBR, DSP, BWF).
Corresponding author
Ethics declarations
Ethics approval
I verify that appropriate Institutional Review Board (IRB), Institutional Animal Care and Use Committee (IACUC), and/or ethics committee approval has been obtained for the use of human or animal subjects.
Consent to participate
I verify that written informed consent was obtained from all research particpants.
Consent for publication
Not applicable.
Conflicts of interest/Competing interests
None of the authors have any conflicts of interest to declare.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Richmond, S.B., Peterson, D.S. & Fling, B.W. Bridging the callosal gap in gait: corpus callosum white matter integrity’s role in lower limb coordination. Brain Imaging and Behavior 16, 1552–1562 (2022). https://doi.org/10.1007/s11682-021-00612-7
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11682-021-00612-7