Abstract
Objective
To observe the effect of Quyu Chencuo Formula (去菀陈莝方, QCF) on renal fibrosis in rats with obstructive nephropathy.
Methods
Twenty-four rats were randomly divided into three groups, 4 for sham operation as the control group, 10 for unilateral ureteral obstruction (UUO) model group, and the rest 10 for QCF treating UUO model group. All rats were sacrificed under 3% pentobarbital (50 mg/kg) anesthesia on the 14th day after surgery, then the right kidney samples of rats were harvested for hematoxylin eosin (HE) staining and Masson staining to observe the renal pathological changes. Immunohistochemistry and Western blotting were used to examine the expression of transforming growth factor β1 (TGF-β1), and real-time polymerase chain reaction (RT-PCR) was employed to examine the expressions of TGF-β1, α-smooth muscle actin (α-SMA) and E-cadherin mRNA.
Results
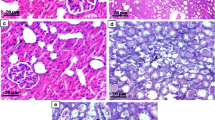
HE and Masson staining showed that the renal interstitial of the rats in the control group had no significant fibrotic lesion; in the model group, there were obvious interstitial fibrosis; for the QCF group, there were epithelial cell necrosis, infiltration of lymphocytes and mononuclear cells, aggravated interstitial fibrosis in varied degrees, but the pathological changes were less in the QCF group than in the model group. The immunohistochemistry and Western blotting results showed that the TGF-β1 expression was increased significantly in the model group, while decreased significantly in the QCF group (P<0.05); RT-PCR showed that the mRNA expression of α-SMA and TGF-β1 increased significantly in the model group, while both were significantly decreased in the QCF group compared with the model group (P<0.05). The mRNA expression of E-cadherin was decreased significantly in the model group, and it was significantly increased in the QCF group as compared with the model group (P<0.05).
Conclusion
QCF may improve renal fibrosis by regulating the expressions of TGF-β1, α-SMA and E-cadherin, and prevent the progress of kidney fibrosis.
Similar content being viewed by others
References
Kisseleva T, Brenner DA. Mechanisms of fibrogenesis. Exp Biol Med 2008;233:109–122.
Levey AS, Atkins R, Coresh J, Cohen EP, Collins AJ, Eckardt KU, et al. Chronic kidney disease as a global public health problem: approaches and initiatives—a position statement from Kidney Disease Improving Global Outcomes. Kidney Int 2007;72:247–259.
Liu Y. Epithelial to mesenchymal transition in renal fibrogenesis: pathologic significance, molecular mechanism, and therapeutic intervention. J Am Soc Nephrol 2004;15:1–12.
Carew R M, Wang B, Kantharidis P. The role of EMT in renal fibrosis. Cell Tissue Res 2012;347:103–116.
Park SH, Choi MJ, Song IK, Choi SY, Nam JO, Kim CD, et al. Erythropoietin decreases renal fibrosis in mice with ureteral obstruction: role of inhibiting TGF–beta–induced epithelial–to–mesenchymal transition. J Am Soc Nephrol 2007;18:1497–1507.
Lamouille S, Xu J, Derynck R. Molecular mechanisms of epithelial–mesenchymal transition. Nature Rev Mol Cell Biol 2014;15:178–196.
Zhou Q, Zeng R, Xu C, Liu L, Chen L, Kou P, et al. Erbin inhibits TGF–β1–induced EMT in renal tubular epithelial cells through an ERK–dependent pathway. J Mol Med (Berl) 2012;90:563–574.
Hinz B. Formation and function of the myofibroblast during tissue repair. J Investig Dermatol 2007;127:526–537.
Qi W, Chen X, Poronnik P, Pollock CA. The renal cortical fibroblast in renal tubulointerstitial fibrosis. Internat J Biochem Cell Boil 2006;38:1–5.
Gao Z, Ku BQ, Qin YF, Liu JG. Effect of Paidu Mixture on fibrosis parameters and renal function of patients with chronic renal failure. J New Chin Med (Chin) 2007;39:90–92.
Ku BQ, Liu JG. Clinical study on change in level of t–lymphocyte subpopulation of the patients with chronic renal failure belongs in blood stasis syndrome and intervening action by Yishen Huoxue Decoction. Hubei J Tradit Chin Med (Chin) 2007;29:5–7.
Zhang B, Liu JG. Clinical efficacy observation of "Jixue Paidu Tang" in treating 33 cases of chronic renal failure. J Sichuan Tradit Chin Med (Chin) 2007;25:47–49.
Zhang B, Wang QS, Zhu R, Wang YH, Zhang LX, Xu M, et al. Expression of serum–and glucocorticoid–induced kinase 1 in renal glomeruli of patients with obstructive nephropathy and its response to curcumin. J Med Postgrad (Chin) 2008;21:245–257.
Eddy AA, Neilson EG. Chronic kidney disease progression. J Am Soc Nephrol 2006;17:2964–2966.
Yang J, Liu Y. Dissection of key events in tubular epithelial to myofibroblast transition and its implications in renal interstitial fibrosis. Am J Pathol 2001;159:1465–1475.
Burns W C, Kantharidis P, Thomas M C. The role of tubular epithelial–mesenchymal transition in progressive kidney disease. Cells Tissues Organs 2007;185:222–231.
Ning X, Ge X, Cui Y, An HX. Ulinastatin inhibits unilateral ureteral obstruction–induced renal interstitial fibrosis in rats via transforming growth factor β (TGF–β)/Smad signalling pathways. Int Immunopharmacol 2013;15:406–413.
Cernaro V, Lacquaniti A, Donato V, Fazio MR, Buemi A, Buemi M. Fibrosis, regeneration and cancer: what is the link? Nephrol Dial Transplant 2012;27:21–27.
Dudas PL, Argentieri RL, Farrell FX. BMP–7 fails to attenuate TGF–β1–induced epithelial–to–mesenchymal transition in human proximal tubule epithelial cells. Nephrol Dialysis Transplant 2009;24:1406–1416.
Author information
Authors and Affiliations
Corresponding author
Additional information
Supported by the National Natural Science Foundation of China (No. 30973834 and No. 81102692)
Rights and permissions
About this article
Cite this article
Zhu, R., Du, Xg., Yang, Sl. et al. Effect of Quyu Chencuo Formula (去菀陈莝方) on Renal Fibrosis in Obstructive Nephropathy Rats. Chin. J. Integr. Med. 25, 190–196 (2019). https://doi.org/10.1007/s11655-015-2133-2
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11655-015-2133-2