Abstract
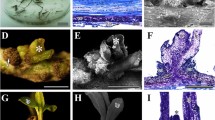
A highly efficient regeneration system of Anthurium andraeanum Linden was established using root segments as explants of four genotypes, Arizona, SWD, OYH, and Mississippi. The root explants were firstly incubated on callus induction media containing NH4NO3 level of 206 mg L−1 or 825 mg L−1, and the calluses were formed from both ends of the root segments after 1 mo of culture. Root-derived calluses were then transferred to shoot regeneration media with NH4NO3 level of 412 mg L−1 or 1650 mg L−1, and numbers of shoot were successfully produced from the surface of calluses after 2 mo of culture. Medium containing 206 mg L−1 NH4NO3 induced more callus compared to 825 mg L−1. In contrast, the number of shoots per callus was the same between the above two shoot regeneration media indicating that NH4NO3 level had little effect on shoot regeneration. Long-termin vitro maintenance of the callus-bearing shoot initials was possible without compromising shoot regeneration and subsequent rooting. Histological analysis showed that early cell division was initiated from the inner cortical parenchyma cells near the vascular tissue and from the vascular parenchyma cells adjacent to the xylem vessel. The primary callus mainly comprised inner parenchyma cells with scattered procambial-like cells and xylem elements and two different meristematic zones beneath epidermal layers. Transfer of callus to the regenerative medium caused meristemoid formation and subsequent shoot primordia production. Calcium oxalate crystals and tannin-like deposits were visible on the outer callus layer or within the cotyledon-like structure of shoot primordium throughout callus development. Starch grains were abundant in root explants, but disappeared in the profusely growing primary callus, and were mobilized in the shoot primordium, indicating energy requirements of these two developmental stages. Flow cytometry analysis demonstrated that ploidy levels were maintained during in vitro cultures, including long-term regenerated plants. The inter-simple sequence repeat analysis indicated genomic DNA stability of Arizona and OYH but occasional genetic variation in primary regenerates of SWD and Mississippi.






Similar content being viewed by others
References
Abhishek S, Sreetama B, Maumita B (2016) Novel nuclei isolation buffer for flow cytometric genome size estimation of Zingiberaceae: a comparison with common isolation buffers. Ann Bot 118:1057–1070. https://doi.org/10.1093/aob/mcw173
Alan AR, Zeng H, Assani A, Shi WL, Mcrae HE, Murch SJ, Saxena PK (2007) Assessment of genetic stability of the germplasm lines of medicinal plant Scutellaria baicalensis Georgi (Huang-qin) in long-term, in vitro maintained cultures. Plant Cell Rep 26:1345–1355. https://doi.org/10.1007/s00299-007-0332-9
Arachchi D, Henny R, Guy C, Li QB (2001) DNA fingerprinting to identify nine Anthurium pot plant cultivars and examine their genetic relationship. Hortscience 36(4): 758-760. https://10.21273/`HORTSCI.36.4.758
Bairu MW, Aremu AO, Staden JV (2011) Somaclonal variation in plants: causes and detection methods. Plant Growth Regul 63:147–173. https://doi.org/10.1007/s10725-010-9554-x
Bejoy M, Sumitha VR, Anish NP (2008) Foliar regeneration in Anthurium andraeanum Hort. cv Agnihothri. Biotechnology 7:134–138
Bhattacharya C, Dam A, Karmakar J, Bandyopadhyay TK (2015) Efficient organogenesis from the induced meristemoid of Anthurium andraeanum Linden cv. Tinora. Plant Sci Today 2:82–86. https://doi.org/10.14719/pst.2015.2.2.110
Bhattacharya C, Dam A, Karmakar J, Bandyopadhyay TK (2016) Direct somatic embryogenesis and genetic homogeneity assessment of regenerated plants of Anthurium andraeanum Linden cv. Fantasia. In Vitro Cell Dev Biol - Plant 52:512–519. https://doi.org/10.1007/s11627-016-9763-8
Bliss BJ, Suzuki JY (2012) Genome size in Anthurium evaluated in the context of karyotypes and phenotypes. AoB Plants 2012:pls006. https://doi.org/10.1093/aobpla/pls006
Cardoso JC, Habermann G (2014) Adventitious shoot induction from leaf segments in Anthurium andraeanumis affected by age of explant, leaf orientation and plant growth regulator. Hortic Environ Biotechnol 55:56–62. https://doi.org/10.1007/s13580-014-0022-9
Chen FC, Kuehnle AR (1996) Obtaining transgenic Anthurium through Agrobacterium-mediated transformation of etiolated internodes. J Amer Soc Hortic Sci 121:47–51
Chen FC, Kuehnle AR, Sugii N (1997)Anthurium roots for micropropagation and Agrobacterium tumefaciens-mediated gene transfer. Plant Cell Tiss Org Cult 49:71–74
Feder N, O’Brine TP (1968) Plant microtechnique: some principles and new methods. Am J Bot 55:123–142
Fiuk A, Bednarek PT, Rybczyński JJ (2010) Flow cytometry, HPLC-RP, and metAFLP analyses to assess genetic variability in somatic embryo-derived plantlets of Gentiana pannonica Scop. Plant Mol Biol Rep 28:413–420. https://doi.org/10.1007/s11105-009-0167-3
Gantait S, Mandal N, Bhattacharyya S, Das PK (2008) In vitro mass multiplication with pure genetic identity in Anthurium andreanum Lind. Plant Tiss Org Cult Biotech 18:113–122. https://doi.org/10.3329/ptcb.v18i2.3361
Gantait S, Sinniah UR (2011) Morphology, flow cytometry and molecular assessment of ex-vitro grown micropropagated Anthurium in comparison with seed germinated plants. Afr J Biotechnol 10:13991–13998. https://doi.org/10.5897/AJB11.1855
Gantait S, Sinniah UR, Mandal N, Das PK (2012) Direct induction of protocorm-like bodies from shoot tips, plantlet formation, and clonal fidelity analysis in Anthurium andreanum cv. CanCan. Plant Growth Regul 67:257–270. https://doi.org/10.1007/s10725-012-9684-4
Geier T (1986) Factors affecting plant regeneration from leaf segments of Anthurium scherzerianum Schott (Araceae) cultured in vitro. Plant Cell Tiss Org Cult 6:115–125
Ghimire BK, Chang YY, Chung IM (2012) Direct shoot organogenesis and assessment of genetic stability in regenerants of Solanum aculeatissimum Jacq. Plant Cell Tiss Org Cult 108:455–464. https://doi.org/10.1007/s11240-011-0057-x
Hosein FN, Lennon AM, Umaharan P (2012) Optimization of an Agrobacterium-mediated transient assay for gene expression studies in Anthurium andraeanum. J Amer Soc Hortic Sci 137:263–272. https://doi.org/10.21273/JASHS.137.4.263
Huang WJ, Ning GG, Liu GF, Bao MZ (2009) Determination of genetic stability of long-term micropropagated plantlets of Platanus acerifolia using ISSR markers. Biol Plant 53:159–163. https://doi.org/10.1007/s10535-009-0025-z
Jaccard P (1908) Nouvelles recherches sur la distribution florale.- Bul.Soc. Vaudoise. SciNat. 44:223–270
Jahan MT, Islam MR, Khan R, Mamun ANK, Hakim L (2014)In vitro clonal propagation of Anthurium (Anthurium andraeanum L.) using callus culture. Plant Tiss Org Cult Biotech 19:61–69. https://doi.org/10.3329/ptcb.v19i1.4961
Kuehnle AR, Chen FC, Sugii N (1992) Somatic embryogenesis and plant regeneration in Anthurium andraeanum hybrids. Plant Cell Rep 11:438–442
Kuehnle AR, Sugii N (1991) Callus induction and plantlet regeneration in tissue cultures of Hawaiian Anthuriums. Hortscience 26:919–921
Lata H, Chandra S, Techen N, Khan I, Elsohly M (2009) Assessment of the genetic stability of micropropagated plants of Cannabis sativa by ISSR markers. Planta Med 76:97–100. https://doi.org/10.1055/s-0029-1185945
Loureiro J, Rodriguez E, Dolezel J, Santos C (2007) Two new nuclear isolation buffers for plant DNA flow cytometry: a test with 37 species. Ann Bot 100:875–888. https://doi.org/10.1093/aob/mcm152
Mallón R, Rodríguez-Oubiña J, González ML (2010)In vitro propagation of the endangered plant Centaurea ultreiae: assessment of genetic stability by cytological studies, flow cytometry and RAPD analysis. Plant Cell Tiss Org Cult 101:31–39. https://doi.org/10.1007/s11240-009-9659-y
Martin KP, Joseph D, Madasser J, Philip VJ (2003) Direct shoot regeneration from lamina explants of two commercial cut flower cultivars of Anthurium andraeanum Hort. In Vitro Cell Dev Biol - Plant 39:500–504
Matsumoto TK, Kuehnle AR (1998) Zygotic embryogenesis in Anthurium (Araceae). Am J Bot 85:1560–1568
Matsumoto TK, Webb DT, Kuehnle AR (1996) Histology and origin of somatic embryos derived from Anthurium andraeanum Linden ex André Lamina. J Amer Soc Hortic Sci 121:404–407. https://doi.org/10.21273/JASHS.121.3.404
Murashige T, Skoog F (1962) A revised medium for rapid growth and bioassays with tobacco tissue culture. Physiol Plant 15:473–497
Neto JDS, Soares TCB, Motta LB, Cabral PDS, Silva JA (2014) Molecular characterization of Anthurium genotypes by using DNA fingerprinting and SPAR markers. Genet Mol Res 13:4766–4775. https://doi.org/10.4238/2014.July.2.6
Paris AM, Pais MS (2000) Organogenesis from internode-derived nodules of Humulus lupulus var. Nugget (Cannabinaceae): histological studies and changes in the starch content. Am J Bot 87:971–979. https://doi.org/10.2307/2656996
Pierik RLM (1976)Anthurium andraeanum plantlets produced from callus tissues cultivated in vitro. Physiol Plant 37:80–82. https://doi.org/10.1111/j.1399-3054.1976.tb01876.x
Pierik RLM, Steegmans HHM, Van Der Meys JAJ (1974) Plantlet formation in callus tissues of Anthurium andraeanum Lind. Sci Hortic 2:193–198
Pinheiro MVM, Martins FB, Cruz ACFD, Carvalho ACPPD, Ventrella MC, Otoni WC (2013) Maturation of Anthurium andraeanum cv. Eidibel somatic embryos from nodal segments. In Vitro Cell Dev Biol - Plant 49:304–312. https://doi.org/10.1007/s11627-013-9522-z
Pinto G, Silva S, Neves L, Araújo C, Santos CO (2010) Histocytological changes and reserve accumulation during somatic embryogenesis in Eucalyptus globulus. Trees 24:763–769. https://doi.org/10.1007/s00468-010-0446-5
Rocha DI, Vieira LM, Tanaka FAO, Silva LCD, Otoni WC (2012) Somatic embryogenesis of a wild passion fruit species Passiflora cincinnata Masters: histocytological and histochemical evidences. Protoplasma 249:747–758
Tang QY, Zhang CX (2013) Data Processing System (DPS) software with experimental design, statistical analysis and data mining developed for use in entomological research. Insect Sci 20:254–260
Teixeira DSJA, Dobránszki J, Winarto B, Zeng SJ (2015)Anthurium in vitro: a review. Sci Hortic 186:266–298. https://doi.org/10.1016/j.scienta.2014.11.024
Varshney A, Sangapillai R, Patil MS, Johnson TS (2011) Histological evidence of morphogenesis from various explants of Jatropha curcas L. Trees 25:689–694. https://doi.org/10.1007/s00468-011-0546-x
Venkat SK, Bommisetty P, Patil MS, Reddy L, Chennareddy A (2014) The genetic linkage maps of Anthurium species based on RAPD, ISSR and SRAP markers. Sci Hortic 178:132–137. https://doi.org/10.1016/j.scienta.2014.08.017
Vujović T, Cerović R, Ružić D (2012) Ploidy level stability of adventitious shoots of sour cherry ‘aanski Rubin’ and Gisela 5 cherry rootstock. Plant Cell Tiss Org Cult 111:323–333. https://doi.org/10.1007/s11240-012-0197-7
Wang GL, Chen C, Li ZX, Li W (2005) Establishment of rapid propagation system of aerial root segments of Anthurium andraeanum Linden in vitro. Plant Physiol Commun 41:297–301 (in Chinese)
Yang X, Hou Z, Ji J (2008) Effect of culture medium and temperature on the ratio of callus of Anthurium leaf. J Shenyang Agri Univ 39:15–18. https://doi.org/10.1007/s10499-007-9164-4 (in Chinese)
Ziegenhagen B, Scholz PG (1993) A procedure for mini-preparations of genomic DNA from needles of silver fir (Abies alba mill.). Plant Mol Biol Rep 11:117–121. https://doi.org/10.1007/BF02670469
Funding
The Natural Science Foundation of Tangshan Normal College (Grant No. 09A01) supported this study.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare no conflict of interest.
Additional information
Editor: Ming Cheng
Rights and permissions
About this article
Cite this article
Zhang, H., Wang, G., Qiao, Y. et al. Plant regeneration from root segments of Anthurium andraeanum and assessment of genetic fidelity of in vitro regenerates. In Vitro Cell.Dev.Biol.-Plant 57, 954–964 (2021). https://doi.org/10.1007/s11627-021-10172-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11627-021-10172-6