Abstract
Background
The brush-border enzyme intestinal alkaline phosphatase (IAP) functions as a gut mucosal defense factor and detoxifies different toll-like receptor ligands. This study aimed to determine the therapeutic effects of locally administered calf IAP (cIAP) in a cecal ligation and puncture (CLP) model of polymicrobial sepsis.
Methods
C57BL/6 mice underwent CLP followed by intraperitoneal injection of cIAP or normal saline. Blood leukocyte counts, levels of cytokines and liver enzymes, and lung myeloperoxidase activity were determined. Peritoneal lavage fluid (PLF) was assayed for neutrophil infiltration and both aerobic and anaerobic bacterial counts.
Results
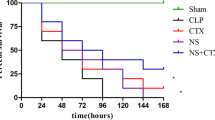
After intraperitoneal injection, cIAP activity in PLF decreased 50% within 15 min with minimal activity evident at 4 h. Compared with irrigation with normal saline, cIAP irrigation increased the 7-day survival rate in mice undergoing CLP, with maximal effects seen at 25 units of cIAP (0% vs. 46% survival rate, respectively; p < 0.001). cIAP treatment reduced lung inflammation, liver damage and levels of tumor necrosis factor alpha and interleukin-6.
Conclusions
Peritoneal irrigation with cIAP significantly enhances survival in a mouse model of peritonitis, likely through reduction of local inflammation and remote organ damage. We suggest that intraperitoneal cIAP irrigation could be a novel therapy for intra-abdominal sepsis.





Similar content being viewed by others
References
Rice TW, Bernard GR. Therapeutic intervention and targets for sepsis. Annu Rev. Med 2005;56:225–248.
Wittmann DH, Schein M, Condon RE. Management of secondary peritonitis. Ann Surg 1996;224:10–18.
Marshall JC, al Naqbi A. Principles of source control in the management of sepsis. Crit Care Clin. 2009;25(4):753–768, viii-ix. Review.
Oğuz M, Bektemir M, Dülger M, Yalin R. Treatment of experimental peritonitis with intraperitoneal povidone-iodine solution. Can J Surg. 1988;31(3):169–171.
van Veen SQ, Levi M, van Vliet AK, Florquin S, van Gulik TM, Boermeester MA. Peritoneal lavage with activated protein C alters compartmentalized coagulation and fibrinolysis and improves survival in polymicrobial peritonitis. Crit Care Med. 2006;34(11):2799–2805.
Brocco MC, Paulo DN, Baptista JF, Ferrari TA, Azevedo TC, Silva AL. Effects of peritoneal lavage with lidocaine on survival of rats with fecal peritonitis. Acta Cir Bras. 2008;23(1):42–47.
Schneider A, Sack U, Rothe K, Bennek J. Peritoneal taurolidine lavage in children with localised peritonitis due to appendicitis. Pediatr Surg Int. 2005;21(6):445–448.
Koyama I., Matsunaga T., Harada T., Hokari S., and Komoda T. Alkaline phosphatase reduce toxicity of lipopolysaccharides in vivo and in vitro through dephosphorylation. Clin Biochem 2002;35: 455–461.
Poelstra K, Bakker WW, Klok PA, Kamps JA, Hardonk MJ, Meijer DK. Dephosphorylation of endotoxin by alkaline phosphatase in vivo. Am J Pathol. 1997; 151(4):1163–9.
Poelstra K, Bakker WW, Klok PA, Hardonk MJ, Meijer DK. A Physiologic function for alkaline phosphatase: endotoxin detoxification. Lab investing 1997;76:319–327.
Bentala H, Verweij WR, Huizinga-Van der Vlag A, van Loenen-Weemaes AM, Meijer DK, Poelstra K. Removal of phosphate from lipid A as a strategy to detoxify lipopolysaccharide. Shock 2002;18(6):561–566.
Beumer C, Wulferink M, Raaben W, Fiechter D, Brands R, and Seinen W. Calf intestinal alkaline phosphatase, a novel therapeutic drug for lipopolysaccharide (LPS) mediated diseases, attenuates LPS toxicity in mice and piglets. J Pharmacol Exp Ther. 2003; 307(2):737–44.
Wichterman KA, Baue AE, Chaudry IH. Sepsis and septic shock-a review of laboratory models and a proposal. J Surg Res 1980;29:189–201.
Hubbard WJ, Choudhry M, Schwacha MG, Kerby JD, Rue LW, Bland KI, Chaudry IH. Cecal Ligation and puncture. Shock 2005;24:52–57.
Van Veen SQ, van Vliet AK, Wulferink M, Brands R, Boermeester MA, van Gulik TM. Bovine intestinal alkaline phosphatase attenuates the inflammatory response in secondary peritonitis in mice. Infect Immun. 2005;73(7):4309–14.
Goldberg RF, Austen WG Jr, Zhang X, Munene G, Mostafa G, Biswas S, McCormack M, Eberlin KR, Nguyen JT, Tatlidede HS, Warren HS, Narisawa S, Millan JL, Hodin RA. Intestinal alkaline phosphatase is a gut mucosal defense factor maintained by enteral nutrition. Proc Natl Acad Sci U S A. 2008 Mar 4;105(9):3551–6.
Doerschug KC, Powers LS, Monick MM, Thorne PS, Hunninghake GW. Antibiotics delay but do not prevent bacteremia and lung injury in murine sepsis. Crit Care Med 2004;32:489–494.
Enoh VT, Lin SH, Etogo A, Lin CY, Sherwood ER. CD4+ T-cell depletion is not associated with alterations in survival, bacterial clearance, and inflammation after cecal ligation and puncture. Shock 2008;29(1);56–64.
Bedirli A, Kerem M, Pasaoglu H, Akyurek N, Tezcaner T, Elbeg S, Memis L, Sakrak O. Beta-glucan attenuates inflammatory cytokine release and prevents acute lung injury in an experimental model of sepsis. Shock 2007;27(4):397–401.
Morath S, Geyer A, Hartung T. Structure-function relationship of cytokine induction by lipoteichoic acid from Staphylococcus aureus. J Exp Med. 2001;193(3):393–397.
Eaves-Pyles T, Murthy K, Liaudet L, Virág L, Ross G, Soriano FG, Szabó C, Salzman AL. Flagellin, a novel mediator of Salmonella-induced epithelial activation and systemic inflammation: I kappa B alpha degradation, induction of nitric oxide synthase, induction of proinflammatory mediators, and cardiovascular dysfunction. J Immunol. 2001;166(2):1248–1260.
Herwald H, Mörgelin M, Olsén A, Rhen M, Dahlbäck B, Müller-Esterl W, Björck L. Activation of the contact-phase system on bacterial surfaces--a clue to serious complications in infectious diseases. Nat Med. 1998;4(3):298–302.
Sparwasser T, Miethke T, Lipford G, Borschert K, Häcker H, Heeg K, Wagner H. Bacterial DNA causes septic shock. Nature. 1997;386(6623):336–337.
Bauer S, Kirschning CJ, Häcker H, Redecke V, Hausmann S, Akira S, Wagner H, Lipford GB. Human TLR9 confers responsiveness to bacterial DNA via species-specific CpG motif recognition. Proc Natl Acad Sci U S A. 2001;98(16):9237–9242.
Chen KT, Malo S, Moss A, et al. Identification of specific targets for the gut mucosal defense factor intestinal alkaline phosphatase. Am J Physiol Gastrointest Liver Physiol. 2010;299(2):G467–75.
Vaishnava S, Hooper LV. Alkaline phosphatase: keeping the peace at the gut epithelial surface. Cell Host Microbe. 2007;2(6):365–367.
Wang P, Ba ZF, Chaudry IH. Mechanism of hepatocellular dysfunction during early sepsis. Key role of increased gene expression and release of proinflammatory cytokines tumor necrosis factor and interleukin-6. Arch Surg 1997;132:364–369.
Xu Z, Foster PA, Gross GJ. A novel role of alkaline phosphatase in protection from immunological liver injury in mice. Liver 2002;22:8–14.
Walley KR, Lukacs NW, Standiford TJ, Strieter RM, Kunkel SL. Balance of inflammatory cytokines related to severity and mortality of murine sepsis. Infect Immun 1996;64(11):4733–4738.
Waage A, Halstensen A, Espevik T. Association between tumor necrosis factor in serum and fatal outcome in patients with meningococcal disease. Lancet 1987;1:355–357.
Ebong S, Call D, Nemzek J, Bolgos G, Newcomb D, Remick D. Immunopathologic alterations in murine models of sepsis of increasing severity. Infect Immun 1999;67(12):6603–6610.
Damas P, Ledoux D, Nys M, Vrindts Y, De Groote D, Franchimont P, Lamy M. Cytokine serum level during severe sepsis in human IL-6 as a marker of severity. Ann Surg 1992;215:356–362.
Baigrie RJ, Lamont PM, Kwiatkowski D, Dallman MJ, Morris PJ. Systemic cytokine response after major surgery. Br J Surg 1992;79:757–760.
Schubert TE, Echtenacher B, Hofstädter F, Männel DN. Failure of interferon-gamma and tumor necrosis factor in mediating anemia of chronic disease in a mouse model of protracted septic peritonitis. Int J Mol Med. 2005 Oct;16(4):753–8.
Doerschug K, Sanlioglu S, Flaherty DM. Wilson RL, Yarovinsky T, Monick MM, Engelhardt JF, Hunninghake GW. First-generation adenovirus vectors shorten survival time in a murine model of sepsis. J Immunol 2002;169(11):6539–6545.
Reinhart K, Menges T, Gardlund B, et al: Randomized, placebo-controlled trial of the anti-tumor necrosis factor antibody fragment afelimomab in hyperinflammatory response during sever sepsis: The RAMSES Study. Crit Care Med 2001;29:765–769.
Martins GA, Da Gloria Da Costa Carvalho M, Rocha Gattass C. Sepsis: a follow up of cytokine production in different phases of septic patients. Int J Mol Med. 2003;11:585–591.
Tuin A, Poelstra K, de Jager-Krikken A, Bok L, Raaben W, Velders MP, Dijkstra G. Role of alkaline phosphatase in colitis in man and rats. Gut 2009;58(3):379–387.
Ramasamy S, Nguyen DD, Eston MA, Nasrin Alam S, Moss AK, Ebrahimi F, Biswas B, Mostafa G, Chen KT, Kaliannan K, Yammine H, Narisawa S, Millán JL, Warren HS, Hohmann EL, Mizoguchi E, Reinecker HC, Bhan AK, Snapper SB, Malo MS, Hodin RA. Intestinal alkaline phosphatase has beneficial effects in mouse models of chronic colitis. Inflamm Bowel Dis. 2011;17(2):532–42
Fielding CA, McLoughlin RM, McLeod L, Colmont CS, Najdovska M, Grail D, Ernst M, Jones SA, Topley N, Jenkins BJ. IL-6 regulates neutrophil trafficking during acute inflammation via STAT3. J Immunol 2008;181(3):2189–2195.
Xiao H, Siddiqui J, Remick DG. Mechanisms of mortality in early and late sepsis. Infect Immun 2006;74(9):5227–5235.
Acknowledgments
This work was supported by NIH grants R01DK050623 and R01DK047186 to RAH, and a Junior Faculty Award from the MGH Department of Surgery and a Grand Challenge Exploration Grant from the Bill and Melinda Gates Foundation to MSM.
Competing interests
The authors declare no conflicting financial interests.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ebrahimi, F., Malo, M.S., Alam, S.N. et al. Local Peritoneal Irrigation with Intestinal Alkaline Phosphatase Is Protective Against Peritonitis in Mice. J Gastrointest Surg 15, 860–869 (2011). https://doi.org/10.1007/s11605-010-1405-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11605-010-1405-6