Abstract
Purpose
This study was conducted to compare the in vitro biomechanical properties of tensile strength and gap resistance of a double grasping loop (DGL) flexor tendon repair with the established four-strand cross-locked cruciate (CLC) flexor tendon repair, both with an interlocking horizontal mattress (IHM) epitendinous suture. The hypothesis is that the DGL-IHM method which utilizes two looped core sutures, grasping and locking loops, and a single intralesional knot will have greater strength and increased gap resistance than the CLC-IHM method.
Methods
Forty porcine tendons were evenly assigned to either the DGL-IHM or CLC-IHM group. The tendon repair strength, 2-mm gap force and load to failure, was measured under a constant rate of distraction. The stiffness of tendon repair was calculated and the method of repair failure was analyzed.
Results
The CLC-IHM group exhibited a statistically significant greater resistance to gapping, a statistically significant higher load to 2-mm gapping (62.0 N), and load to failure (99.7 N) than the DGL-IHM group (37.1 N and 75.1 N, respectively). Ninety percent of CLC-IHM failures were a result of knot failure whereas 30 % of the DGL-IHM group exhibited knot failure.
Conclusions
This study demonstrates that the CLC-IHM flexor tendon repair method better resists gapping and has a greater tensile strength compared to the experimental DGL-IHM method. The authors believe that while the DGL-IHM provides double the number of sutures at the repair site per needle pass, this configuration does not adequately secure the loop suture to the tendon, resulting in a high percentage of suture pullout and inability to tolerate loads as high as those of the CLC-IHM group.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
In 1918, Sterling Bunnell coined the term “no man’s land” for flexor tendon injuries which occurred between the proximal margin of the A-1 pulley and the distal margin of the A-4 pulley [8]. In the 1970s, Kleinert et al. [33] demonstrated that flexor tendon repair was indeed possible in no man’s land using a meticulous surgical technique and early active extension of the finger. This was successful because active extension of the finger resulted in relaxation of the flexor tendon [34] and minimal force across the suture line at the site of tendon repair while allowing excursion of the tendon through the sheath, thereby minimizing adhesion formation and secondary loss of motion. More recent research has focused on flexor tendon repair techniques that allow early active flexion as well as extension. An ideal repair method is one that provides sufficient strength to allow early active finger flexion, is resistant to gap formation in order to limit scarring, contributes minimally to bulk, and is easily reproducible without tendon damage [53]. The repair should restore tendon function by maximizing motion [18] while limiting peritendinous adhesion formation [19], and allow for early protected rehabilitation [3]. Early protected motion protocols have been shown to benefit tendon healing, with decreased adhesions, and therefore improve functional results [3, 16, 56, 64]. Due to these benefits, repair techniques attempt to attain the strength to withstand at least 30–51 N of force [16, 26, 48] of early rehabilitation.
The combination suture repair method of a cross-locked cruciate (CLC) 3-0 FiberWire (Arthrex, Naples, FL) core suture with an interlocking horizontal mattress (IHM) 6-0 PROLENE (Ethicon, Somerville, NJ) circumferential suture was used as comparison in this study for its demonstrated superior performance and strength as documented in the literature [11, 13, 54]. It is suggested that the Adelaide repair technique (CLC), first reported by Sandow and McMahon [46], is more favorable than other selected four-strand techniques in terms of strength, gap formation, and simplicity [2, 4, 6, 11, 39]. FiberWire was chosen because it has been shown to provide greater strength than similar low-profile braided sutures [36, 41]. The additional reinforcement of a circumferential suture method was used given that it has been shown to contribute significantly to tendon repair by limiting gap formation and providing up to 50 % of the strength of repair and thereby reducing the rate of repair rupture [12, 32, 50]. The IHM suture method, originally described by Dona et al. in 2003, was selected as it has been found to be superior to a simple running or a cross-locked circumferential suture [13].
The success of a tendon repair can be predicted based on the ultimate tensile strength and the ability to resist gapping, which are influenced by the suture material used and the surgical technique, including the configuration and strand number [1, 3, 4, 42, 43, 49, 56, 68]. The recent development of looped sutures using a braided polyblend suture (FiberWire) provides the potential for combining the one of the strongest suture material for flexor tendon repair with two strands of suture for each pass of the needle [17, 31]. Although Brockardt et al. [7] demonstrated that using a looped suture in a traditional manner adds no benefit over two separate suture passes, Cao and Tang [9] reveal greater holding power using looped sutures when the repair method is optimized by utilizing the loop to lock anchors to the tendon forming tendon-suture junctions.
In this study, the authors propose a “3-double-stranded double grasping loop” (DGL) suture method utilizing two 4-0 looped FiberWire (Arthrex, Naples, FL) core sutures with an IHM 6-0 PROLENE (Ethicon, Somerville, NJ) circumferential suture. Our hypothesis is that the DGL technique with double the number of strands of suture per pass of the needle with a single knot at the site of tendon repair will provide added strength and resistance to gapping without the added complexity of multiple needle passes. The purpose of this study was to compare the in vitro biomechanical properties of tensile strength in terms of load to 2-mm gapping and load to failure, as well as gap resistance in terms of stiffness, with those of the established CLC-IHM repair method. The hypothesis was that the DGL-IHM method will have a greater load to failure and increased gap resistance.
Materials and Methods
Experimental Design
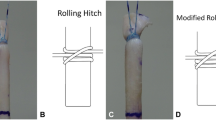
Forty flexor digitorum profundus tendons from 110- to 130-lb Yorkshire pigs were used for this in vitro study. Porcine tendons have been established in previous biomechanical studies as a standard experimental model given the similarity to human flexor digitorum profundus tendons and ease of availability [11, 42, 48, 51]. Two tendons from each foot were harvested, separated, and randomly allocated into one of two groups: control and experimental, to avoid selection bias. Throughout the harvest, repair, and testing processes, the tendons were kept moist using sterile saline. Tendons were transected transversely with a no. 15 scalpel into two equal segments simulating a zone 2 injury. The four-strand Adelaide cross-locked cruciate (CLC) core (3-0 FiberWire) suture method [46] was employed for the control group (Fig. 1a), and the experimental group utilized a novel double grasping loop (DGL) core (two 4-0 looped Fiberwire) suture method (Fig. 1b). The cross locks of the CLC method were set at 10 mm from the tendon division line. Both study groups included a circumferential running interlocking horizontal mattress (IHM) epitendinous (6-0 PROLENE) suture repair method (Fig. 1c). To minimize data variability, repairs of each group were evenly performed by two surgeons using ×2.5 loupe magnification without any deviations made from the described techniques. To prevent impact on tendon tensile properties, tendons were refrigerated, wrapped in saline-soaked gauze after repair prior to biomechanical testing [28].
Surgical Technique
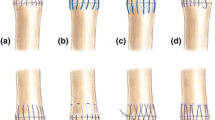
The authors have termed this flexor tendon repair method a double grasping loop repair for its configuration using two looped sutures (Fig. 2a-g). Each looped suture is initiated 10 mm from the laceration site on each tendon segment incorporating 25 % of the tendon width prior to being passed through the suture loop to “grasp” the tendon. Then, the needle is advanced through the grasped portion of the tendon into the laceration site and the opposite segment with 10-mm purchase length. The suture is then passed through the side wall to a central point prior to being locked in a fashion similar to a modified Kessler repair with a pass exiting the laceration site centrally. The second looped suture is initiated on the contralateral wall of the opposite tendon segment and passed simultaneously in a mirrored fashion. The two suture ends are then knotted centrally within the repair site.
Double grasping loop flexor tendon repair method: looped suture is initiated (a); passed through suture, looped, and tightened (b); advanced through grasped tendon portion to laceration site (c), attaining 10-mm purchase length of opposite segment (d); passed from lateral wall to central point (e); locked as inserted centrally returning to laceration site (f); and knotted within repair site with mirrored suture of opposite segment (g)
For both groups, the core suture purchase length was standardized at 10 mm from the tendon division to maximize strength [10, 57] with the loops incorporating 25 % of the tendon, attempting to minimize gapping [14], and the knots were placed within the transection site [45]. All knots were tied using a surgeon’s knot, followed by three alternating half hitches. The IHM repair was standardized as well with both groups utilizing 12 loops and 2-mm purchase, 6 loops on each side of the laceration [13, 40].
Mechanical Testing
To investigate tensile strength and load to 2-mm gapping of the repaired tendons, each tendon was secured into a Tinius Olsen electromechanical materials testing machine (Horsham, PA) using standard specimen non-slip clamps connected to a 200-lb load cell force transducer. The clamps were distracted at a rate of 2/100th of an inch per minute. Every 30 s throughout the distraction, tendons and an adjacent fixed linear scale for calibration were photographed using two digital cameras. The minimum and maximum amounts of gapping visualized from each camera were calculated. At these same time points, force measurements were collected. From this data, the load at which the maximum amount of gapping visualized on either camera equaled 0.5, 1, 2, and 4 mm was recorded. As well, the load at which ultimate failure occurred was recorded, defined as the point at which the peak force occurred just before the force across the tendon suddenly decreased. Repair site stress was then calculated using the measured forces and cross-sectional areas of each tendon (avg. 27.9 ± 3.84 mm2). Tendon stiffness was calculated from the tangent of the linear region of the load displacement curve, representing the resistance to gap formation. After testing, the tendons were analyzed at ×10 power under a light microscope (American Optical Stereo Star Model 569, Buffalo, NY) for the method of failure. Failure mechanisms of both the core suture and the epitendinous suture were categorized as suture breakage, knot failure, or suture pullout. Two modes of core suture failure were identified: suture pullout and knot failure. Suture pullout was defined as a repair in which the core suture maintained continuity with its knot remaining visibly intact. Knot failure, on the other hand, was defined as a repair in which the knot of the core suture was unraveled with two ends of the suture visible and no knot present.
Statistical Analysis
Measurements obtained from tendon testing were evaluated using independent t tests. These data were analyzed with statistical software, SPSS 12.0 (Statistical Package for the Social Sciences, SPSS Inc., Chicago, IL). Data were graphed as means with 95 % confidence intervals. Findings were considered to be statistically significant with a p value of less than 0.05.
Results
The CLC-IHM group demonstrated statistically significant higher loads to attain gapping compared to the DGL-IHM group for all measured gap intervals: 0.5, 1.0, 2.0, and 4.0 mm. Figure 3 demonstrates that the CLC-IHM group had a statistically significant higher load to 2-mm gapping, 62.0 N compared to 37.1 N (p ≤ 0.025), and a statistically significant higher load to failure, 99.7 N compared to 75.1 N (p ≤ 0.025). As well, the CLC-IHM group had a statistically significant greater resistance to gapping with a stiffness of 16.9 MPa compared to 12.9 MPa for the DGL-IHM group (p ≤ 0.0001) (Fig. 4). Resistance to gapping was significantly higher in the CLC-IHM group for all gap measurements.
Ninety percent of the CLC-IHM group failed due to core suture knot failure, which was statistically significant when compared to only 30 % of the DGL-IHM group (p < 0.01), with the remaining 10 and 70 % of each respective group failing due to core suture pullout (Fig. 5). In specifically examining epitendinous suture failure, the results were not statistically significant: 75 % of the CLC-IHM group and 60 % of the DGL-IHM group failed due to suture rupture near the knot within the laceration site. The remaining CLC-IHM epitendinous suture failures occurred 10 % by suture pullout and 15 % by knot failure, whereas the remaining DGL-IHM epitendinous suture failures occurred 10 % by suture pullout and 30 % by knot failure (Fig. 6).
Discussion
Numerous studies have examined suture methods and the many variables involved in an attempt to determine the best flexor tendon repair method. Attention has been paid to the biomechanical factors of gap strength and load to failure as studies have demonstrated gap formation to be associated with poor results [22, 50]. Currently, the strongest technique described involves the use of stainless steel wire held together with a metal crimp [25]. Other techniques, including the CLC-IHM, are able to withstand loads of 30–51 N of early rehabilitation [16, 26, 48] and therefore allow the patient to achieve the many benefits of early active mobilization, including decreased adhesions, improved range of motion, increased grip strength, and enhanced tendon nutrition and healing [20, 21, 52]. It can be concluded that an ideal flexor tendon repair is one that is strong enough to tolerate early motion with limited gapping.
For the control group, this study utilized the CLC Adelaide method in combination with an IHM circumferential epitendinous suture due to the proven biomechanical properties of this suture method [2, 4, 6, 11, 39]. This study proved that the CLC-IHM flexor tendon repair method better resists gapping with a greater tensile strength than the DGL-IHM flexor tendon repair method. The superiority of this repair is attributed to the many biomechanical attributes that are incorporated into the repair. The CLC-IHM repair method utilizes the strength of the FiberWire core suture [36, 41]; the improved strength and decreased risk of gapping with locked loops [4, 6, 29, 48, 62, 63, 66, 67]; the improved strength with reduced number of points of tensile weakness, unraveling, and bulk of a single intralesional knot [41]; and the improved performance with limited complexity of four strands [15]. The four-strand CLC repair technique has been suggested to be more favorable than other four-strand techniques in terms of strength, gap formation, and simplicity [2, 4, 6, 11] and was therefore used as a control in this study. As well, a circumferential suture, which has been shown to contribute markedly to tendon repair by limiting gap formation, providing up to 50 % of the strength of repair, and reducing repair rupture rate [12, 32, 50], was employed. It was hypothesized that the DGL-IHM repair method would prove to be superior as this repair also incorporated three strands of doubled suture crossing the repair site, effectively providing six strands of suture, for proposed strength and gap resistance without added complexity and additional grasping loops along with the same biomechanical attributes of a FiberWire core suture, locked loops, an intralesional knot, and a circumferential epitenon suture. This method is also in accordance with the recommendations of Cao and Tang [9], in that it fully utilizes the looped design of the suture in its locked anchoring. It is important to note, however, that the 4-0 looped FiberWire core suture was employed in the experimental group to complement the 3-0 FiberWire used in the control group as this more closely resembles the total core suture cross-sectional area (DGL 0.402 mm2 and CLC 0.332 mm2) according to calculations based on the measurements of Scherman [47]. This is reinforced by the fact that 90 % of the failure of the control group was attributed to suture pullout rather than suture breakage.
We believe that the high percentage of failure by suture pullout in the DGL-IHM group is due to the smaller amount of tendon enclosed within the loops of the repair when compared to the CLC-IHM repair. Because the DGL-IHM repair was unable to maintain its position within the tendon, it was therefore unable to tolerate loads as high as those tolerated by the CLC-IHM group. On the contrary, a majority of the CLC-IHM group failed by knot failure, likely due to the superiority of the repair method, the concentration of tensile loads on a single intralesional knot, and the higher propensity of FiberWire to fail by unraveling at lower loads, specifically with less than six-knot throws [30, 35].
The DGL-IHM method was designed to provide the biomechanical strength and gap resistance one would expect from having an increased number of crossing sutures per needle pass [4, 5, 37, 60, 65]. Not only are there fewer needle passes, but the authors found the DGL-IHM technique relatively easy to perform with little change in the shape of the tendon [3, 37, 39, 53]. As these six crossing sutures are not individual strands, they are likely to have different biomechanical properties than a traditional six-strand repair. Other studies have demonstrated that an increased number of strands compared to passes in various repair methods are able to provide improved strength and gap resistance [5, 24, 55, 61]. However, due to the failure of the DGL-IHM group prior to obtaining loads high enough to cause failure of the suture or the knot, the potential benefit of having two crossing sutures for each pass of the needle is not clearly established for this repair.
One of the primary limitations of this study is that it is an in vitro animal model study. This study used pig tendons which, among animal tendons, have been preferred for flexor tendon repair studies [51]. However, an in vitro study is unable to evaluate adhesion formation, tendon healing, and the effect of post-operative changes on the biomechanical properties of the tendon. Findings from this study are the result of a time 0 study and the correlation with clinical results is difficult to ascertain. The tensile load and gapping measurements of this study were obtained using static load, rather than cyclic loading, which may not provide an accurate representation of the repetitive loads associated with rehabilitation as cyclic loading has been shown to be more physiologic and lead to earlier gap formation compared to static loading [23, 44].
In an attempt to limit the variability of repair, the two groups were assigned equally to two performing surgeons. Nonetheless, repairs were handmade with variable positioning of the suture and suture tension at the time of repair. These human error variables should be distributed throughout both groups evenly and should not affect one repair more than another. Anatomic variations within the porcine tendons should be considered as well. An attempt to account for this was made by dividing two tendons from each foot and randomly allocating one tendon to the control group and the other to the experimental group. Another limitation to this study may be the number of throws for each knot as studies have shown that FiberWire produces more bulk and requires more throws than other sutures to create a secure knot [30]. All knots in this study were tied using a surgeon’s knot, followed by three alternating half hitches, in an attempt to account for unraveling and bulk. Some studies have suggested that a minimum of six-knot throws is required to prevent FiberWire unraveling [38, 58].
Since repair strength cannot exceed the load to failure of the suture and knot [27, 59], gap formation rather than load of ultimate failure becomes the most important criterion in assessing repair strength, specifically for repairs that failed by knot unraveling. This study utilized a maximum gapping measurement of 2 mm on one of two viewpoints as the threshold given the findings of Gelberman et al. [22] that 1 mm minimally affects resistance whereas 3 mm may prevent motion.
This study has identified that the CLC-IHM group has required not only a greater load to ultimate failure but also a greater load to 2-mm gapping with a statistically significant greater resistance to gapping (stiffness) compared to the DGL-IHM group. Given these results, the authors are able to reject their hypothesis and cannot recommend the DGL-IHM flexor tendon repair method without further improvement and investigation.
References
Amadio PC. Friction of the gliding surface. Implications for tendon surgery and rehabilitation. J Hand Ther. 2005;18:112–9.
Angeles JG, Heminger H, Mass DP. Comparative biomechanical performances of 4-strand core suture repairs for zone II flexor tendon lacerations. J Hand Surg [Am]. 2002;27:508–17.
Aoki M, Kubota H, Pruitt DL, et al. Biomechanical and histologic characteristics of canine flexor tendon repair using early postoperative mobilization. J Hand Surg [Am]. 1997;22:107–14.
Barrie KA, Tomak SL, Cholewicki J, et al. The role of multiple strands and locking sutures on gap formation of flexor tendon repairs during cyclical loading. J Hand Surg [Am]. 2000;25:714–20.
Barrie KA, Wolfe SW, Shean C, et al. A biomechanical comparison of multistrand flexor tendon repairs using an in situ testing model. J Hand Surg [Am]. 2000;25:499–506.
Barrie KA, Tomak SL, Cholewicki J, et al. Effect of suture locking and suture caliber on fatigue strength of flexor tendon repairs. J Hand Surg [Am]. 2001;26:340–6.
Brockardt CJ, Sullivan LG, Watkins BE, et al. Evaluation of simple and looped suture and new material for flexor tendon repair. J Hand Surg Eur Vol. 2009;34:329–32.
Bunnell S. Repair of tendons in the fingers and description of two new instruments. Surg Gynecol Obstet. 1918;26:103–10.
Cao Y, Tang JB. Biomechanical evaluation of a four-strand modification of the Tang method of tendon repair. J Hand Surg (Br). 2005;30:374–8.
Cao Y, Zhu B, Xie RG, et al. Influence of core suture purchase length on strength of four-strand tendon repairs. J Hand Surg Am. 2006;31:107–12.
Croog A, Goldstein R, Nasser P, et al. Comparative biomechanic performances of locked cruciate four-strand flexor tendon repairs in an ex vivo porcine model. J Hand Surg Am. 2007;32:225–32.
Diao E, Hariharan JS, Soejima O, et al. Effect of peripheral suture depth on strength of tendon repairs. J Hand Surg Am. 1996;21:234–9.
Dona E, Turner AW, Gianoutsos MP, et al. Biomechanical properties of four circumferential flexor tendon suture techniques. J Hand Surg Am. 2003;28:824–31.
Dona E, Gianoutsos MP, Walsh WR. Optimizing biomechanical performance of the 4-strand cruciate flexor tendon repair. J Hand Surg Am. 2004;29:571–80.
Elliot D, Moiemen NS, Flemming AF, et al. The rupture rate of acute flexor tendon repairs mobilized by the controlled active motion regimen. J Hand Surg (Br). 1994;19:607–12.
Evans RB, Thompson DE. The application of force to the healing tendon. J Hand Ther. 1993;6:266–84.
Gan AW, Neo PY, He M, et al. A biomechanical comparison of 3 loop suture materials in a 6-strand flexor tendon repair technique. J Hand Surg [Am]. 2012;37:1830–4.
Gelberman RH, Woo SL, Lothringer K, et al. Effects of early intermittent passive mobilization on healing canine flexor tendons. J Hand Surg [Am]. 1982;7:170–5.
Gelberman RH, Manske PR. Factors influencing flexor tendon adhesions. Hand Clin. 1985;1:35–42.
Gelberman RH, Woo SL, Amiel D, et al. Influences of flexor sheath continuity and early motion on tendon healing in dogs. J Hand Surg Am. 1990;15:69–77.
Gelberman RH, Nunley 2nd JA, Osterman AL, et al. Influences of the protected passive mobilization interval on flexor tendon healing a prospective randomized clinical study. Clin Orthop Relat Res. 1991;264:189–96.
Gelberman RH, Boyer MI, Brodt MD, et al. The effect of gap formation at the repair site on the strength and excursion of intrasynovial flexor tendons. An experimental study on the early stages of tendon-healing in dogs. J Bone Joint Surg Am. 1999;81:975–82.
Gibbons CE, Thompson D, Sandow MJ. Flexor tenorrhaphy tensile strength: reduction by cyclic loading: in vitro and ex vivo porcine study. Hand (N Y). 2009;4:113–8.
Gill RS, Lim BH, Shatford RA, et al. A comparative analysis of the six-strand double-loop flexor tendon repair and three other techniques: a human cadaveric study. J Hand Surg Am. 1999;24:1315–22.
Gordon L, Matsui J, McDonald E, et al. Analysis of a knotless flexor tendon repair using a multifilament stainless steel cable-crimp system. J Hand Surg Am. 2013;38:677–83.
Greenwald D, Shumway S, Allen C, et al. Dynamic analysis of profundus tendon function. J Hand Surg Am. 1994;19:626–35.
Herrmann JB. Tensile strength and knot security of surgical suture materials. Am Surg. 1971;37:209–17.
Hirpara KM, Sullivan PJ, O'Sullivan ME. The effects of freezing on the tensile properties of repaired porcine flexor tendon. J Hand Surg Am. 2008;33:353–8.
Hotokezaka S, Manske PR. Differences between locking loops and grasping loops: effects on 2-strand core suture. J Hand Surg Am. 1997;22:995–1003.
Ilahi OA, Younas SA, Ho DM, et al. Security of knots tied with ethibond, fiberwire, orthocord, or ultrabraid. Am J Sports Med. 2008;36:2407–14.
Karjalainen T, He M, Chong AK, et al. An analysis of the pull-out strength of 6 suture loop configurations in flexor tendons. J Hand Surg Am. 2012;37:217–23.
Kim PT, Aoki M, Tokita F, et al. Tensile strength of cross-stitch epitenon suture. J Hand Surg (Br). 1996;21:821–3.
Kleinert HE, Kutz JE, Atasoy E, et al. Primary repair of flexor tendons. Orthop Clin North Am. 1973;4:865–76.
Kleinert HE, Kutz JE, Cohen MJ. Primary repair of zone 2 flexor tendon lacerations. In AAOS Symposium on Tendon Surgery in the Hand. St. Louis: C.V. Mosby, pp. 91–104, 1975.
Komatsu F, Mori R, Uchio Y. Optimum surgical suture material and methods to obtain high tensile strength at knots: problems of conventional knots and the reinforcement effect of adhesive agent. J Orthop Sci. 2006;11:70–4.
Lawrence TM, Davis TR. A biomechanical analysis of suture materials and their influence on a four-strand flexor tendon repair. J Hand Surg Am. 2005;30:836–41.
Lawrence TM, Woodruff MJ, Aladin A, et al. An assessment of the tensile properties and technical difficulties of two- and four-strand flexor tendon repairs. J Hand Surg (Br). 2005;30:294–7.
Le SV, Chiu S, Meineke RC, et al. Number of suture throws and its impact on the biomechanical properties of the four-strand cruciate locked flexor tendon repair with FiberWire. J Hand Surg Eur Vol. 2012;37:826–31.
McLarney E, Hoffman H, Wolfe SW. Biomechanical analysis of the cruciate four-strand flexor tendon repair. J Hand Surg Am. 1999;24:295–301.
Merrell GA, Wolfe SW, Kacena WJ, et al. The effect of increased peripheral suture purchase on the strength of flexor tendon repairs. J Hand Surg Am. 2003;28:464–8.
Miller B, Dodds SD, deMars A, et al. Flexor tendon repairs: the impact of fiberwire on grasping and locking core sutures. J Hand Surg Am. 2007;32:591–6.
Mishra V, Kuiper JH, Kelly CP. Influence of core suture material and peripheral repair technique on the strength of Kessler flexor tendon repair. J Hand Surg (Br). 2003;28:357–62.
Momose T, Amadio PC, Zhao C, et al. The effect of knot location, suture material, and suture size on the gliding resistance of flexor tendons. J Biomed Mater Res. 2000;53:806–11.
Pruitt DL, Manske PR, Fink B. Cyclic stress analysis of flexor tendon repair. J Hand Surg [Am]. 1991;16:701–7.
Pruitt DL, Aoki M, Manske PR. Effect of suture knot location on tensile strength after flexor tendon repair. J Hand Surg [Am]. 1996;21:969–73.
Sandow MJ, McMahon MM. Single-cross grasp six-strand repair for acute flexor tenorrhaphy: modified Savage technique. Atlas Hand Clin. 1996;1:41–64.
Scherman P, Haddad R, Scougall P, et al. Cross-sectional area and strength differences of fiberwire, prolene, and ticron sutures. J Hand Surg [Am]. 2010;35:780–4.
Schuind F, Garcia-Elias M, Cooney 3rd WP, et al. Flexor tendon forces: in vivo measurements. J Hand Surg [Am]. 1992;17:291–8.
Shaieb MD, Singer DI. Tensile strengths of various suture techniques. J Hand Surg (Br). 1997;22:764–7.
Silfverskiöld KL, Andersson CH. Two new methods of tendon repair: an in vitro evaluation of tensile strength and gap formation. J Hand Surg [Am]. 1993;18:58–65.
Smith AM, Forder JA, Annapureddy SR, et al. The porcine forelimb as a model for human flexor tendon surgery. J Hand Surg (Br). 2005;30:307–9.
Strickland JW, Glogovac SV. Digital function following flexor tendon repair in zone II: a comparison of immobilization and controlled passive motion techniques. J Hand Surg [Am]. 1980;5:537–43.
Strickland JW. Flexor tendon injuries: I foundations of treatment. J Am Acad Orthop Surg. 1995;3:44–54.
Strickland JW. Development of flexor tendon surgery: twenty-five years of progress. J Hand Surg [Am]. 2000;25:214–35.
Tang JB, Shi D, Gu YQ, et al. Double and multiple looped suture tendon repair. J Hand Surg (Br). 1994;19:699–703.
Tang JB, Wang B, Chen F, et al. Biomechanical evaluation of flexor tendon repair techniques. Clin Orthop Relat Res. 2001;386:252–9.
Tang JB, Zhang Y, Cao Y, et al. Core suture purchase affects strength of tendon repairs. J Hand Surg [Am]. 2005;30:1262–6.
Tidwell JE, Kish VL, Samora JB, et al. Knot security: how many throws does it really take? Orthopedics. 2012;35:e532–7.
Trail IA, Powell ES, Noble J. An evaluation of suture materials used in tendon surgery. J Hand Surg (Br). 1989;14:422–7.
Viinikainen A, Göransson H, Huovinen K, et al. A comparative analysis of the biomechanical behaviour of five flexor tendon core sutures. J Hand Surg (Br). 2004;29:536–43.
Viinikainen A, Göransson H, Huovinen K, et al. The strength of the 6-strand modified Kessler repair performed with triple-stranded or triple-stranded bound suture in a porcine extensor tendon model: an ex vivo study. J Hand Surg [Am]. 2007;32:510–7.
Viinikainen A, Göransson H, Ryhänen J. Primary flexor tendon repair techniques. Scand J Surg. 2008;97:333–40.
Wada A, Kubota H, Hatanaka H, et al. The mechanical properties of locking and grasping suture loop configurations in four-strand core suture techniques. J Hand Surg (Br). 2000;25:548–51.
Wada A, Kubota H, Miyanishi K, et al. Comparison of postoperative early active mobilization and immobilization in vivo utilising a four-strand flexor tendon repair. J Hand Surg (Br). 2001;26:301–6.
Winters SC, Gelberman RH, Woo SL, et al. The effects of multiple-strand suture methods on the strength and excursion of repaired intrasynovial flexor tendons: a biomechanical study in dogs. J Hand Surg [Am]. 1998;23:97–104.
Xie RG, Tang JB. Investigation of locking configurations for tendon repair. J Hand Surg [Am]. 2005;30:461–5.
Xie RG, Xue HG, Gu JH, et al. Effects of locking area on strength of 2- and 4-strand locking tendon repairs. J Hand Surg [Am]. 2005;30:455–60.
Zhao C, Amadio PC, Tanaka T, et al. Effect of gap size on gliding resistance after flexor tendon repair. J Bone Joint Surg Am. 2004;86:2482–8.
Conflict of Interest
C. Liam Dwyer declares that he has no conflict of interest.
D. Dean Dominy declares that he has no conflict of interest.
Timothy E. Cooney declares that he has no conflict of interest.
Richard Englund declares that he has no conflict of interest.
Leonard Gordon declares that he has no conflict of interest.
John D. Lubahn declares that he has no conflict of interest.
Statement of Human and Animal Rights
All institutional and national guidelines for the care and use of laboratory animals were followed.
Statement of Informed Consent
Not applicable in the study.
Disclaimer
Grant support was provided by Arthrex, Inc. for reimbursement for suture materials and test fixture. The authors have no affiliation with or disclosures related to the manufacturer.
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Dwyer, C.L., Dominy, D.D., Cooney, T.E. et al. Biomechanical comparison of double grasping repair versus cross-locked cruciate flexor tendon repair. HAND 10, 16–22 (2015). https://doi.org/10.1007/s11552-014-9728-5
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11552-014-9728-5