Abstract
Purpose
To evaluate the ability of an artificial intelligence (AI) tool in magnetic resonance imaging (MRI) assessment of degenerative pathologies of lumbar spine using radiologist evaluation as a gold standard.
Methods
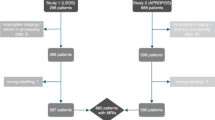
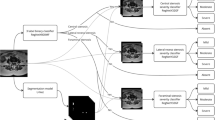
Patients with degenerative pathologies of lumbar spine, evaluated with MRI study, were enrolled in a retrospective study approved by local ethical committee. A comprehensive software solution (CoLumbo; SmartSoft Ltd., Varna, Bulgaria) designed to label the segments of the lumbar spine and to detect a broad spectrum of degenerative pathologies based on a convolutional neural network (CNN) was employed, utilizing an automatic segmentation. The AI tool efficacy was compared to data obtained by a senior neuroradiologist that employed a semiquantitative score.
Chi-square test was used to assess the differences among groups, and Spearman’s rank correlation coefficient was calculated between the grading assigned by radiologist and the grading obtained by software. Moreover, agreement was assessed between the value assigned by radiologist and software.
Results
Ninety patients (58 men; 32 women) affected with degenerative pathologies of lumbar spine and aged from 60 to 81 years (mean 66 years) were analyzed. Significant correlations were observed between grading assigned by radiologist and the grading obtained by software for each localization. However, only when the localization was L2–L3, there was a good correlation with a coefficient value of 0.72. The best agreements were obtained in case of L1–L2 and L2–L3 localizations and were, respectively, of 81.1% and 72.2%. The lowest agreement of 51.1% was detected in case of L4–L5 locations. With regard canal stenosis and compression, the highest agreement was obtained for identification of in L5–S1 localization.
Conclusions
AI solution represents an efficacy and useful toll degenerative pathologies of lumbar spine to improve radiologist workflow.




Similar content being viewed by others
Data availability
Data are available at link https://zenodo.org/records/10643131.
References
Chea P, Mandell JC (2020) Current applications and future directions of deep learning in musculoskeletal radiology. Skeletal Radiol 49(2):183–197. https://doi.org/10.1007/s00256-019-03284-z
Mazurowski MA, Buda M, Saha A, Bashir MR (2019) Deep learning in radiology: an overview of the concepts and a survey of the state of the art with focus on MRI. J Magn Reson Imaging 49(4):939–954. https://doi.org/10.1002/jmri.26534
Piantadosi G, Sansone M, Fusco R, Sansone C (2020) Multi-planar 3D breast segmentation in MRI via deep convolutional neural networks. Artif Intell Med 103:101781. https://doi.org/10.1016/j.artmed.2019.101781
Fusco R, Grassi R, Granata V, Setola SV, Grassi F, Cozzi D, Pecori B, Izzo F, Petrillo A (2021) Artificial intelligence and COVID-19 using chest CT scan and chest X-ray images: machine learning and deep learning approaches for diagnosis and treatment. J Pers Med 11(10):993. https://doi.org/10.3390/jpm11100993
Ueno M, Yoshida K, Takamatsu A, Kobayashi T, Aoki T, Gabata T (2023) Deep learning-based automatic detection for pulmonary nodules on chest radiographs: the relationship with background lung condition, nodule characteristics, and location. Eur J Radiol 166:111002. https://doi.org/10.1016/j.ejrad.2023.111002
Herrmann J, Gassenmaier S, Keller G, Koerzdoerfer G, Almansour H, Nickel D, Othman A, Afat S, Werner S (2023) Deep learning MRI reconstruction for accelerating turbo spin echo hand and wrist imaging: a comparison of image quality, visualization of anatomy, and detection of common pathologies with standard imaging. Acad Radiol S1076–6332(22):00698–5. https://doi.org/10.1016/j.acra.2022.12.042
Kruskal JB, Berkowitz S, Geis JR, Kim W, Nagy P, Dreyer K (2017) Big data and machine learning-strategies for driving this bus: a summary of the 2016 intersociety summer conference. J Am Coll Radiol 14(6):811–817. https://doi.org/10.1016/j.jacr.2017.02.019
Yang L, Ene IC, Arabi Belaghi R, Koff D, Stein N, Santaguida PL (2022) Stakeholders’ perspectives on the future of artificial intelligence in radiology: a scoping review. Eur Radiol 32(3):1477–1495. https://doi.org/10.1007/s00330-021-08214-z
Huisman M, Ranschaert E, Parker W, Mastrodicasa D, Koci M, Pinto de Santos D, Coppola F, Morozov S, Zins M, Bohyn C, Koç U, Wu J, Veean S, Fleischmann D, Leiner T, Willemink MJ (2021) An international survey on AI in radiology in 1041 radiologists and radiology residents part 2: expectations, hurdles to implementation, and education. Eur Radiol 31(11):8797–8806. https://doi.org/10.1007/s00330-021-07782-4
https://www.signifyresearch.net/medical-imaging/ai-medical-imaging-top-2-billion-2023/
Langlotz CP (2019) Will artificial intelligence replace radiologists? Radiol Artif Intell 1(3):e190058. https://doi.org/10.1148/ryai.2019190058.
Chiwome L, Okojie OM, Rahman AKMJ, Javed F, Hamid P (2020) Artificial intelligence: is it armageddon for breast radiologists? Cureus 12(6):e8923. https://doi.org/10.7759/cureus.8923.
van Leeuwen KG, Schalekamp S, Rutten MJCM, van Ginneken B, de Rooij M (2021) Artificial intelligence in radiology: 100 commercially available products and their scientific evidence. Eur Radiol 31(6):3797–3804. https://doi.org/10.1007/s00330-021-07892-z
Fryback DG, Thornbury JR (1991) The efficacy of diagnostic imaging. Med Decis Making 11(2):88–94. https://doi.org/10.1177/0272989X9101100203
Ding IJ, Zheng NW (2022) CNN deep learning with wavelet image fusion of CCD RGB-IR and Depth-grayscale sensor data for hand gesture intention recognition. Sensors 22(3):803. https://doi.org/10.3390/s22030803.
Roy D, Panda P, Roy K (2020) Tree-CNN: a hierarchical deep convolutional neural network for incremental learning. Neural Netw 121:148–160. https://doi.org/10.1016/j.neunet.2019.09.010
Elizar E, Zulkifley MA, Muharar R, Zaman MHM, Mustaza SM (2022) A review on multiscale-deep-learning applications. Sensors 22(19):7384. https://doi.org/10.3390/s22197384
Gholami H, Mohammadifar A (2022) Novel deep learning hybrid models (CNN-GRU and DLDL-RF) for the susceptibility classification of dust sources in the middle east: a global source. Sci Rep 12(1):19342. https://doi.org/10.1038/s41598-022-24036-5
Singh SP, Wang L, Gupta S, Goli H, Padmanabhan P, Gulyás B (2020) 3D deep learning on medical images: a review. Sensors 20(18):5097. https://doi.org/10.3390/s20185097
Hamabe A, Ishii M, Kamoda R, Sasuga S, Okuya K, Okita K, Akizuki E, Sato Y, Miura R, Onodera K, Hatakenaka M, Takemasa I (2022) Artificial intelligence-based technology for semi-automated segmentation of rectal cancer using high-resolution MRI. PLoS ONE 17(6):e0269931. https://doi.org/10.1371/journal.pone.0269931
Lewandrowski KU, Muraleedharan N, Eddy SA, Sobti V, Reece BD, Ramírez León JF, Shah S (2020) Artificial intelligence comparison of the radiologist report with endoscopic predictors of successful transforaminal decompression for painful conditions of the lumber spine: application of deep learning algorithm interpretation of routine lumbar magnetic resonance imaging scan. Int J Spine Surg 14(s3):S75–S85. https://doi.org/10.14444/7130
Grob A, Loibl M, Jamaludin A, Winklhofer S, Fairbank JCT, Fekete T, Porchet F, Mannion AF (2022) External validation of the deep learning system “SpineNet” for grading radiological features of degeneration on MRIs of the lumbar spine. Eur Spine J 31(8):2137–2148. https://doi.org/10.1007/s00586-022-07311-x
Granata V, Fusco R, De Muzio F, Cutolo C, Setola SV, Dell’Aversana F, Ottaiano A, Nasti G, Grassi R, Pilone V, Miele V, Brunese MC, Tatangelo F, Izzo F, Petrillo A (2022) EOB-MR based radiomics analysis to assess clinical outcomes following liver resection in colorectal liver metastases. Cancers 14(5):1239. https://doi.org/10.3390/cancers14051239
Granata V, Ianniello S, Fusco R, Urraro F, Pupo D, Magliocchetti S, Albarello F, Campioni P, Cristofaro M, Di Stefano F, Fusco N, Petrone A, Schininà V, Villanacci A, Grassi F, Grassi R, Grassi R (2021) Quantitative analysis of residual COVID-19 lung CT features: consistency among two commercial software. J Pers Med 11(11):1103. https://doi.org/10.3390/jpm11111103
Bernardo P, Budetta M, Aliberti F, Carpentieri ML, De Brasi D, Sorrentino L, Russo C, D’amico A, Cinalli G, Santoro C, Coppola A (2021) Temporal lobe malformations, focal epilepsy, and FGFR3 mutations: a non-causal association? Neurol Sci 42(5):2063–2067. https://doi.org/10.1007/s10072-020-04923-3
Rubino A, Bernardo P, Russo C, Tucci C, D’Amato L, Piccolo V, Andreone V, Striano P, Zito Marinosci G, Varone A (2023) Immediate and progressive neurological damage after electrical injury: a pediatric case report. Brain Dev 45(1):87–91. https://doi.org/10.1016/j.braindev.2022.08.007
D’Amico A, Perillo T, Russo C, Ugga L, Melis D, Santoro C, Piluso G, Cinalli G (2021) Enhancing cyst-like lesions of the white matter in tuberous sclerosis complex: a novel neuroradiological finding. Neuroradiology 63(6):971–974. https://doi.org/10.1007/s00234-021-02647-5
Acknowledgements
The authors are grateful to Alessandra Trocino, librarian at the National Cancer Institute of Naples, Italy.
Funding
The authors have not disclosed any funding.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors have no conflicts of interest to be disclosed. The authors confirm that the article is not under consideration for publication elsewhere. Each author has participated sufficiently to take public responsibility for the content of the manuscript.
Ethical approval
The study was performed in accordance with an up-to-date Declaration of Helsinki version and International Conference on Harmonization of Good Clinical Practice Guidelines.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Granata, V., Fusco, R., Coluccino, S. et al. Preliminary data on artificial intelligence tool in magnetic resonance imaging assessment of degenerative pathologies of lumbar spine. Radiol med 129, 623–630 (2024). https://doi.org/10.1007/s11547-024-01791-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11547-024-01791-1