Abstract
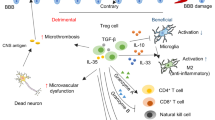
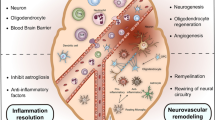
Regulatory T cells (Tregs) inhibit the activation of the immune response which could down-regulate the systemic and focal activation observed during ischemic stroke. In fact, in animal models, Tregs infiltrate the infarcted brain and reduce the pro-inflammatory cytokine production and infarct volume, mainly in late stages of ischemia. Recently, an expansion and greater suppressive capacity of circulating Tregs after treatment with statins was observed, in addition to their cardio- and neuroprotective actions demonstrated previously. Thus, to determine whether Treg modulation mediated by statins can also be beneficial during stroke, cerebral ischemia was artificially induced in Wistar rats by transient middle cerebral artery occlusion (tMCAO) during 60 minutes with subsequent reperfusion for 7 days. Six hours after surgery, some animals were treated with atorvastatin (ATV, 10 mg/kg) or carboxymethylcellulose as vehicle at the same concentration every other day during 7 days. Some animals were sham operated as control group of surgery. Interestingly, ATV treatment prevented the development of infarct volume, reduced the neurological deficits, and the circulating and cervical lymph node CD25+FoxP3+ Treg population. Moreover, there was a reduction of glial cell activation, which correlated with decreased circulating Tregs. Remarkably, treatment with ATV induced an increase in the frequency of CD4+CD25+ T cells, in particular of those expressing CTLA-4, in brain samples. Together, these results suggest that ATV can modulate Tregs in peripheral tissue and favor their accumulation in the brain, where they can exert neuroprotective actions maybe by the reduction of glial cell activation.






Similar content being viewed by others
References
Ajmo CT, Vernon DO, Collier L, Hall AA, Garbuzova-Davis S, Willing A, Pennypacker KR (2008) The spleen contributes to stroke-induced neurodegeneration. J Neurosci Res 86:2227–2234
Anderson SL, Duke-Novakovski T, Singh B (2014) The immune response to anesthesia: part 1. Vet Anaesth Analg 41:113–126
Barakat R, Redzic Z (2015) The role of activated microglia and resident macrophages in the neurovascular unit during cerebral ischemia: is the jury still out? Med Princ Pract 25:3–14
Callaway JK, Wood C, Jenkins TA, Royse AG, Royse CF (2016) Isoflurane in the presence or absence of surgery increases hippocampal cytokines associated with memory deficits and responses to brain injury in rats. Behav Brain Res 303:44–52
Céspedes-Rubio A, Jurado FW, Cardona-Gómez GP (2010) p120 catenin/αN-catenin are molecular targets in the neuroprotection and neuronal plasticity mediated by atorvastatin after focal cerebral ischemia. J Neurosci Res 88:3621–3634
Chan A, Yan J, Csurhes P, Greer J, McCombe P (2015) Circulating brain derived neurotrophic factor (BDNF) and frequency of BDNF positive T cells in peripheral blood in human ischemic stroke: effect on outcome. J Neuroimmunol 286:42–47
Chen J, Zhang ZG, Li Y, Wang Y, Wang L, Jiang H, Zhang C, Lu M, Katakowski M, Feldkamp CS, Chopp M (2003) Statins induce angiogenesis, neurogenesis, and synaptogenesis after stroke. Ann Neurol 53:743–751
Corbett D, Nurse S (1998) The problem of assessing effective neuroprotection in experimental cerebral ischemia. Prog Neurobiol 54:531–548
Cui X, Chopp M, Zacharek A, Roberts C, Lu M, Savant-Bhonsale S, Chen J (2009) Chemokine, vascular and therapeutic effects of combination simvastatin and BMSC treatment of stroke. Neurobiol Dis 36:35–41
Culley DJ, Cotran EK, Karlsson E, Palanisamy A, Boyd JD, Xie Z, Crosby G (2013) Isoflurane affects the cytoskeleton but not survival, proliferation, or synaptogenic properties of rat astrocytes in vitro. Br J Anaesth 110(Suppl 1):i19–i28
Dirnagl U, Klehmet J, Braun JS, Harms H, Meisel C, Ziemssen T, Prass K, Meisel A (2007) Stroke-induced immunodepression: experimental evidence and clinical relevance. Stroke; J Cereb Circ 38:770–773
García-Bonilla L, Campos M, Giralt D, Salat D, Chacón P, Hernández-Guillamon M, Rosell A, Montaner J (2012) Evidence for the efficacy of statins in animal stroke models: a meta-analysis. J Neurochem 122:233–243
Gelderblom M, Leypoldt F, Steinbach K, Behrens D, Choe CU, Siler DA, Arumugam TV, Orthey E, Gerloff C, Tolosa E, Magnus T (2009) Temporal and spatial dynamics of cerebral immune cell accumulation in stroke. Stroke; J Cereb Circ 40:1849–1857
Gelderblom M, Weymar A, Bernreuther C, Velden J, Arunachalam P, Steinbach K, Orthey E, Arumugam TV, Leypoldt F, Simova O, Thom V, Friese MA, Prinz I, Hölscher C, Glatzel M, Korn T, Gerloff C, Tolosa E, Magnus T (2012) Neutralization of the IL-17 axis diminishes neutrophil invasion and protects from ischemic stroke. Blood 120:3793–3802
Graca L, Cobbold SP, Waldmann H (2002) Identification of regulatory T cells in tolerated allografts. J Exp Med 195:1641–1646
Greenwood J, Steinman L, Zamvil SS (2006) Statin therapy and autoimmune disease: from protein prenylation to immunomodulation. Nat Rev Immunol 6:358–370
Gu L, Xiong X, Wei D, Gao X, Krams S, Zhao H (2013) T cells contribute to stroke-induced lymphopenia in rats. PLoS One 8:e59602
Gutierrez-Vargas JA, Muñoz-Manco JI, Garcia-Segura LM, Cardona-Gómez GP (2014) GluN2B N-methyl-D-aspartic acid receptor subunit mediates atorvastatin-induced neuroprotection after focal cerebral ischemia. J Neurosci Res 92:1529–1548
Gutiérrez-Vargas JA, Moreno H, Cardona-Gómez GP (2016) Targeting CDK5 post-stroke provides long-term neuroprotection and rescues synaptic plasticity. J Cereb Blood Flow Metab. doi:10.1177/0271678X16662476
Hewett SJ, Jackman NA, Claycomb RJ (2012) Interleukin-1β in central nervous system injury and repair. Eur J Neurodegener Dis 1:195–211
Hu Y, Zheng Y, Wu Y, Ni B, Shi S (2014) Imbalance between IL-17 A-producing cells and regulatory T cells during ischemic stroke. Mediat Inflamm 2014:813045
Huehn J, Siegmund K, Lehmann JC, Siewert C, Haubold U, Feuerer M, Debes GF, Lauber J, Frey O, Przybylski GK, Niesner U, de la Rosa M, Schmidt CA, Bräuer R, Buer J, Scheffold A, Hamann A (2004) Developmental stage, phenotype, and migration distinguish naive- and effector/memory-like CD4+ regulatory T cells. J Exp Med 199:303–313
Kawai H, Deguchi S, Deguchi K, Yamashita T, Ohta Y, Omote Y, Kurata T, Ikeda Y, Matsuura T, Abe K (2011) Protection against ischemic stroke damage by synergistic treatment with amlodipine plus atorvastatin in Zucker metabolic rat. Brain Res 1382:308–314
Ke D, Fang J, Fan L, Chen Z, Chen L (2013) Regulatory T cells contribute to rosuvastatin-induced cardioprotection against ischemia-reperfusion injury. Coron Artery Dis 24:334–341
Kim E, Yang J, Beltran CD, Cho S (2014) Role of spleen-derived monocytes/macrophages in acute ischemic brain injury. J Cereb Blood Flow Metab 34:1411–1419
Kingsley CI, Karim M, Bushell AR, Wood KJ (2002) CD25 + CD4+ regulatory T cells prevent graft rejection: CTLA-4- and IL-10-dependent immunoregulation of alloresponses. J Immunol 168:1080–1086
Kinsey GR, Sharma R, Huang L, Li L, Vergis AL, Ye H, Ju ST, Okusa MD (2009) Regulatory T cells suppress innate immunity in kidney ischemia-reperfusion injury. J Am Soc Nephrol 20:1744–1753
Kleinschnitz C, Kraft P, Dreykluft A, Hagedorn I, Göbel K, Schuhmann MK, Langhauser F, Helluy X, Schwarz T, Bittner S, Mayer CT, Brede M, Varallyay C, Pham M, Bendszus M, Jakob P, Magnus T, Meuth SG, Iwakura Y, Zernecke A, Sparwasser T, Nieswandt B, Stoll G, Wiendl H (2013) Regulatory T cells are strong promoters of acute ischemic stroke in mice by inducing dysfunction of the cerebral microvasculature. Blood 121:679–691
Klingenberg R, Gerdes N, Badeau RM, Gisterå A, Strodthoff D, Ketelhuth DF, Lundberg AM, Rudling M, Nilsson SK, Olivecrona G, Zoller S, Lohmann C, Lüscher TF, Jauhiainen M, Sparwasser T, Hansson GK (2013) Depletion of FOXP3+ regulatory T cells promotes hypercholesterolemia and atherosclerosis. J Clin Invest 123:1323–1334
Lai LW, Yong KC, Lien YH (2012) Pharmacologic recruitment of regulatory T cells as a therapy for ischemic acute kidney injury. Kidney Int 81:983–992
Li P, Gan Y, Sun BL, Zhang F, Lu B, Gao Y, Liang W, Thomson AW, Chen J, Hu X (2013) Adoptive regulatory T-cell therapy protects against cerebral ischemia. Ann Neurol 74:458–471
Li P, Mao L, Liu X, Gan Y, Zheng J, Thomson AW, Gao Y, Chen J, Hu X (2014) Essential role of program death 1-ligand 1 in regulatory T-cell-afforded protection against blood-brain barrier damage after stroke. Stroke; J Cereb Circ 45:857–864
Liesz A, Suri-Payer E, Veltkamp C, Doerr H, Sommer C, Rivest S, Giese T, Veltkamp R (2009) Regulatory T cells are key cerebroprotective immunomodulators in acute experimental stroke. Nat Med 15:192–199
Ling EM, Smith T, Nguyen XD, Pridgeon C, Dallman M, Arbery J, Carr VA, Robinson DS (2004) Relation of CD4 + CD25+ regulatory T-cell suppression of allergen-driven T-cell activation to atopic status and expression of allergic disease. Lancet 363:608–615
Mausner-Fainberg K, Luboshits G, Mor A, Maysel-Auslender S, Rubinstein A, Keren G, George J (2008) The effect of HMG-CoA reductase inhibitors on naturally occurring CD4 + CD25+ T cells. Atherosclerosis 197:829–839
Mira E, Leon B, Barber DF, Jimenez-Baranda S, Goya I, Almonacid L, Marquez G, Zaballos A, Martinez AC, Stein JV, Ardavin C, Manes S (2008) Statins induce regulatory T cell recruitment via a CCL1 dependent pathway. J Immunol 181:3524–3534
Mor A, Planer D, Luboshits G, Afek A, Metzger S, Chajek-Shaul T, Keren G, George J (2007) Role of naturally occurring CD4+ CD25+ regulatory T cells in experimental atherosclerosis. Arterioscler Thromb Vasc Biol 27:893–900
Mottet C, Uhlig HH, Powrie F (2003) Cutting edge: cure of colitis by CD4 + CD25+ regulatory T cells. J Immunol 170:3939–3943
Offner H, Subramanian S, Parker SM, Wang C, Afentoulis ME, Lewis A, Vandenbark AA, Hurn PD (2006) Splenic atrophy in experimental stroke is accompanied by increased regulatory T cells and circulating macrophages. J Immunol 176:6523–6531
Pfaffl MW (2001) A new mathematical model for relative quantification in real-time RT-PCR. Nucleic Acids Res 29:e45
Popp A, Jaenisch N, Witte OW, Frahm C (2009) Identification of ischemic regions in a rat model of stroke. PLoS One 4:e4764
Posada-Duque RA, Velasquez-Carvajal D, Eckert GP, Cardona-Gomez GP (2013) Atorvastatin requires geranylgeranyl transferase-I and Rac1 activation to exert neuronal protection and induce plasticity. Neurochem Int 62:433–445
Potey C, Ouk T, Petrault O, Petrault M, Berezowski V, Salleron J, Bordet R, Gautier S (2015) Early treatment with atorvastatin exerts parenchymal and vascular protective effects in experimental cerebral ischaemia. Br J Pharmacol 172:5188–5198
Qiu R, Cai A, Dong Y, Zhou Y, Yu D, Huang Y, Zheng D, Rao S, Feng Y, Mai W (2012) SDF-1α upregulation by atorvastatin in rats with acute myocardial infarction via nitric oxide production confers anti-inflammatory and anti-apoptotic effects. J Biomed Sci 19:99
Reynolds AD, Banerjee R, Liu J, Gendelman HE, Mosley RL (2007) Neuroprotective activities of CD4 + CD25+ regulatory T cells in an animal model of Parkinson's disease. J Leukoc Biol 82:1083–1094
Rodríguez-Perea AL, Montoya CJ, Olek S, Chougnet CA, Velilla PA (2015) Statins increase the frequency of circulating CD4+ FOXP3+ regulatory T cells in healthy individuals. J Immunol Res 2015:762506
Saito T, Nito C, Ueda M, Inaba T, Kamiya F, Muraga K, Katsura K, Katayama Y (2014) Continuous oral administration of atorvastatin ameliorates brain damage after transient focal ischemia in rats. Life Sci 94:106–114
Sakaguchi S, Sakaguchi N, Asano M, Itoh M, Toda M (1995) Immunologic self-tolerance maintained by activated T cells expressing IL-2 receptor alpha-chains (CD25). Breakdown of a single mechanism of self-tolerance causes various autoimmune diseases. J Immunol 155:1151–1164
Seifert HA, Hall AA, Chapman CB, Collier LA, Willing AE, Pennypacker KR (2012) A transient decrease in spleen size following stroke corresponds to splenocyte release into systemic circulation. J NeuroImmune Pharmacol 7:1017–1024
Shim R, Wong CH (2016) Ischemia, immunosuppression and infection--tackling the predicaments of post-stroke complications. Int J Mol Sci 17
Skinner R, Georgiou R, Thornton P, Rothwell N (2009) Psychoneuroimmunology of stroke. Immunol Allergy Clin N Am 29:359–379
Stubbe T, Ebner F, Richter D, Engel O, Randolf Engel O, Klehmet J, Royl G, Meisel A, Nitsch R, Meisel C, Brandt C (2013) Regulatory T cells accumulate and proliferate in the ischemic hemisphere for up to 30 days after MCAO. J Cereb Blood Flow Metab 33:37–47
Tai X, Van Laethem F, Pobezinsky L, Guinter T, Sharrow SO, Adams A, Granger L, Kruhlak M, Lindsten T, Thompson CB, Feigenbaum L, Singer A (2012) Basis of CTLA-4 function in regulatory and conventional CD4(+) T cells. Blood 119:5155–5163
Tang TT, Song Y, Ding YJ, Liao YH, Yu X, Du R, Xiao H, Yuan J, Zhou ZH, Liao MY, Yao R, Jevallee H, Shi GP, Cheng X (2011) Atorvastatin upregulates regulatory T cells and reduces clinical disease activity in patients with rheumatoid arthritis. J Lipid Res 52:1023–1032
Urra X, Cervera A, Villamor N, Planas AM, Chamorro A (2009) Harms and benefits of lymphocyte subpopulations in patients with acute stroke. Neuroscience 158:1174–1183
Veenhof AA, Vlug MS, van der Pas MH, Sietses C, van der Peet DL, de Lange-de Klerk ES, Bonjer HJ, Bemelman WA, Cuesta MA (2012) Surgical stress response and postoperative immune function after laparoscopy or open surgery with fast track or standard perioperative care: a randomized trial. Ann Surg 255:216–221
Villamil-Ortiz JG, Cardona-Gomez GP (2015) Comparative analysis of autophagy and tauopathy related markers in cerebral ischemia and Alzheimer's disease animal models. Front Aging Neurosci 7:84
Wang J, Xie L, Yang C, Ren C, Zhou K, Wang B, Zhang Z, Wang Y, Jin K, Yang GY (2015) Activated regulatory T cell regulates neural stem cell proliferation in the subventricular zone of normal and ischemic mouse brain through interleukin 10. Front Cell Neurosci 9:361
Xie L, Choudhury GR, Winters A, Yang SH, Jin K (2015) Cerebral regulatory T cells restrain microglia/macrophage-mediated inflammatory responses via IL-10. Eur J Immunol 45:180–191
Yan J, Read SJ, Henderson RD, Hull R, O'Sullivan JD, McCombe PA, Greer JM (2012) Frequency and function of regulatory T cells after ischaemic stroke in humans. J Neuroimmunol 243:89–94
Yang S, Liu J, Zhang X, Tian J, Zuo Z, Yue X (2016) Anesthetic isoflurane attenuates activated microglial cytokine-induced VSC4.1 motoneuronal apoptosis. Am J Transl Res 8:1437–1446
Yasuno F, Taguchi A, Yamamoto A, Kajimoto K, Kazui H, Kudo T, Kikuchi-Taura A, Sekiyama A, Kishimoto T, Iida H, Nagatsuka K (2014) Microstructural abnormality in white matter, regulatory T lymphocytes, and depressive symptoms after stroke. Psychogeriatrics 14:213–221
Yilmaz G, Arumugam TV, Stokes KY, Granger DN (2006) Role of T lymphocytes and interferon-gamma in ischemic stroke. Circulation 113:2105–2112
Zhou W, Liesz A, Bauer H, Sommer C, Lahrmann B, Valous N, Grabe N, Veltkamp R (2013) Postischemic brain infiltration of leukocyte subpopulations differs among murine permanent and transient focal cerebral ischemia models. Brain Pathol 23:34–44
Acknowledgments
The authors wish to thank Anne-Lise Haenni for her constructive comments. Ana Lucia Rodriguez is recipient of a doctoral scholarship from COLCIENCIAS.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interests
The authors declare that there is no conflict of interests regarding the publication of this paper.
Additional information
An erratum to this article is available at http://dx.doi.org/10.1007/s11481-016-9714-5.
Electronic Supplementary Material
Supplemental Figure 1
mRNA expression of IL-1β (a), IFN-γ (b), IL-6 (c), TNF-α (d), MPO (e), MMP-9 (f), NOX-2 (g) and TjP-1 (h) from tissue obtained of penumbra zone in sham-operated rats, tMCAO and tMCAO ATV. Graphs represent median and IQR; n = 1–5 rats/group. (TIFF 110 kb) (GIF 110 kb)
Supplemental Figure 2
The percentage of CD25+FoxP3+ Treg cells was determined by flow cytometry in peripheral blood (a) spleen (b) and cervical lymph nodes (c) after 60 min of tMCAO at day 7 and 15 postreperfusion. The sham-operated rats and tMCAO rats were treated with vehicle (carboxymethylcellulose) administered at the same dose and frequency as the treatment. Another group of tMCAO rats was treated with ATV (10 mg/Kg). First doses of ATV or vehicle began 6 h after surgical procedure; the rats sacrificed at day 3 received each day a dose of ATV or vehicle, whereas animals sacrificed at day 15 received doses every other day. Graphs represent median and IQR. Mann–Whitney test shows no significant differences between the groups studied. n = 2–5 rats/group. (GIF 67 kb)
Supplemental Figure 3
The percentage of CD3+CD4+FoxP3−CTLA-4+ cells was determined by flow cytometry in peripheral blood, spleen and lymphoid nodules in sham operated rats, tMCAO-Veh and tMCAO-ATV at day 7 post reperfusion. Graphs represent median and IQR. The Mann–Whitney test showed no significant differences between the groups studied. n = 8–9 rats/group. (GIF 24 kb)
Supplemental Figure 4
mRNA expression of FOXP3 (a), CTLA-4 (b), IDO (c), IL-10 (d) and TGF-β (e) from splenic CD4+ T cells in sham operated rats, tMCAO-Veh and tMCAO-ATV at day 7 post reperfusion. Graphs represent median and IQR. The Mann–Whitney test showed no significant differences between the groups studied. n = 4–5 rats/group. (GIF 60 kb)
Supplemental Figure 5
mRNA expression of IL-1β (a), IFN-γ (b), IL-6 (c), TNF-α (d) and IL-17 (e) from splenic CD4+ T cells in sham operated rats, tMCAO-Veh and tMCAO-ATV at day 7 post reperfusion. Graphs represent median and IQR. The Mann–Whitney test showed no significant differences between the groups studied. n = 4–5 rats/group. (GIF 65 kb)
Supplemental Figure 6
Representative histograms of CFSE labelling of CD3+CD4+ and CD3+CD4− cells showing proliferation of both cell types. Mononuclear cells from spleen of sham operated rats, tMCAO-Veh and tMCAO-ATV at day 7 post reperfusion were thawed and stained with Carboxyfluorescein succinimidyl ester (CFSE) (1.25 μM, Molecular Probes/Invitrogen, Eugene, OR, USA) and seeded in 96-well culture plates and stimulation or not with concanavalin A (1.5 μg/ml, Sigma) at 37 °C and 5 % CO2 for 72 h. Proliferation was measured in both T cell types. Data were collected on a FACSDiVa (BD Biosciences) flow cytometer and analyzed using FlowJo software (Tree Star, San Carlos, CA, USA). Proliferation index (PI) is shown in each histogram. The Mann–Whitney test showed no significant differences between the groups studied. n = 2–4 rats/group. (GIF 112 kb)
Supplemental Table 1
(GIF 153 kb)
Rights and permissions
About this article
Cite this article
Rodríguez-Perea, A.L., Gutierrez-Vargas, J., Cardona-Gómez, G.P. et al. Atorvastatin Modulates Regulatory T Cells and Attenuates Cerebral Damage in a Model of Transient Middle Cerebral Artery Occlusion in Rats. J Neuroimmune Pharmacol 12, 152–162 (2017). https://doi.org/10.1007/s11481-016-9706-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11481-016-9706-5