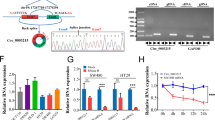
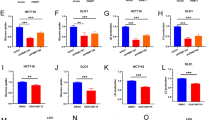
Abstract
Cancer cells remodel their metabolic network to adapt to variable nutrient availability. Pentose phosphate pathway (PPP) plays protective and biosynthetic roles by oxidizing glucose to generate reducing power and ribose. How cancer cells modulate PPP activity in response to glucose supply remains unclear. Here we show that ribose-5-phosphate isomerase A (RPIA), an enzyme in PPP, directly interacts with co-activator associated arginine methyltransferase 1 (CARM1) and is methylated at arginine 42 (R42). R42 methylation up-regulates the catalytic activity of RPIA. Furthermore, glucose deprivation strengthens the binding of CARM1 with RPIA to induce R42 hypermethylation. Insufficient glucose supply links to RPIA hypermethylation at R42, which increases oxidative PPP flux. RPIA methylation supports ROS clearance by enhancing NADPH production and fuels nucleic acid synthesis by increasing ribose supply. Importantly, RPIA methylation at R42 significantly potentiates colorectal cancer cell survival under glucose starvation. Collectively, RPIA methylation connects glucose availability to nucleotide synthesis and redox homeostasis.
Similar content being viewed by others
References
Bedford, M.T., and Clarke, S.G. (2009). Protein arginine methylation in mammals: Who, what, and why. Mol Cell 33, 1–13.
Brooks, S.S., Anderson, S., Bhise, V., and Botti, C. (2018). Further delineation of ribose-5-phosphate isomerase deficiency: Report of a third case. J Child Neurol 33, 784–787.
Cairns, R.A., Harris, I.S., and Mak, T.W. (2011). Regulation of cancer cell metabolism. Nat Rev Cancer 11, 85–95.
Capriles, P.V.S.Z., Baptista, L.P.R., Guedes, I.A., Guimarães, A.C.R., Custódio, F.L., Alves-Ferreira, M., and Dardenne, L.E. (2015). Structural modeling and docking studies of ribose 5-phosphate isomerase from Leishmania major and Homo sapiens: A comparative analysis for Leishmaniasis treatment. J Mol Graph Model 55, 134–147.
Chou, Y.T., Jiang, J.K., Yang, M.H., Lu, J.W., Lin, H.K., Wang, H.D., Yuh, C.H., and Christofk, H. (2018). Identification of a noncanonical function for ribose-5-phosphate isomerase A promotes colorectal cancer formation by stabilizing and activating β-catenin via a novel C-terminal domain. PLoS Biol 16, e2003714.
Chou, Y.T., Chen, L.Y., Tsai, S.L., Tu, H.C., Lu, J.W., Ciou, S.C., Wang, H. D., and Yuh, C.H. (2019). Ribose-5-phosphate isomerase A overexpression promotes liver cancer development in transgenic zebrafish via activation of ERK and β-catenin pathways. Carcinogenesis 40, 461–473.
Ciou, S.C., Chou, Y.T., Liu, Y.L., Nieh, Y.C., Lu, J.W., Huang, S.F., Chou, Y.T., Cheng, L.H., Lo, J.F., Chen, M.J., et al. (2015). Ribose-5-phosphate isomerase A regulates hepatocarcinogenesis via PP2A and ERK signaling. Int J Cancer 137, 104–115.
DeBerardinis, R.J., Lum, J.J., Hatzivassiliou, G., and Thompson, C.B. (2008). The biology of cancer: Metabolic reprogramming fuels cell growth and proliferation. Cell Metab 7, 11–20.
Graham, N.A., Tahmasian, M., Kohli, B., Komisopoulou, E., Zhu, M., Vivanco, I., Teitell, M.A., Wu, H., Ribas, A., Lo, R.S., et al. (2012). Glucose deprivation activates a metabolic and signaling amplification loop leading to cell death. Mol Syst Biol 8, 589.
Gu, H., Ren, J.M., Jia, X., Levy, T., Rikova, K., Yang, V., Lee, K.A., Stokes, M.P., and Silva, J.C. (2016). Quantitative profiling of post-translational modifications by immunoaffinity enrichment and LC-MS/MS in cancer serum without immunodepletion. Mol Cell Proteomics 15, 692–702.
Guo, A., Gu, H., Zhou, J., Mulhern, D., Wang, Y., Lee, K.A., Yang, V., Aguiar, M., Kornhauser, J., Jia, X., et al. (2014). Immunoaffinity enrichment and mass spectrometry analysis of protein methylation. Mol Cell Proteomics 13, 372–387.
Hanahan, D., and Weinberg, R.A. (2011). Hallmarks of cancer: The next generation. Cell 144, 646–674.
Hitosugi, T., Zhou, L., Elf, S., Fan, J., Kang, H.B., Seo, J.H., Shan, C., Dai, Q., Zhang, L., Xie, J., et al. (2012). Phosphoglycerate mutase 1 coordinates glycolysis and biosynthesis to promote tumor growth. Cancer Cell 22, 585–600.
Hong, H., Kao, C., Jeng, M.H., Eble, J.N., Koch, M.O., Gardner, T.A., Zhang, S., Li, L., Pan, C.X., Hu, Z., et al. (2004). Aberrant expression of CARM1, a transcriptional coactivator of androgen receptor, in the development of prostate carcinoma and androgen-independent status. Cancer 101, 83–89.
Huck, J.H.J., Verhoeven, N.M., Struys, E.A., Salomons, G.S., Jakobs, C., and van der Knaap, M.S. (2004). Ribose-5-phosphate isomerase deficiency: New inborn error in the pentose phosphate pathway associated with a slowly progressive leukoencephalopathy. Am J Human Genets 74, 745–751.
Jiang, P., Du, W., and Wu, M. (2014). Regulation of the pentose phosphate pathway in cancer. Protein Cell 5, 592–602.
Kim, D., Lee, J., Cheng, D., Li, J., Carter, C., Richie, E., and Bedford, M.T. (2010). Enzymatic activity is required for the in vivo functions of CARM1. J Biol Chem 285, 1147–1152.
Kim, N.H., Cha, Y.H., Lee, J., Lee, S.H., Yang, J.H., Yun, J.S., Cho, E.S., Zhang, X., Nam, M., Kim, N., et al. (2017). Snail reprograms glucose metabolism by repressing phosphofructokinase PFKP allowing cancer cell survival under metabolic stress. Nat Commun 8, 14374.
Kim, Y.R., Lee, B.K., Park, R.Y., Nguyen, N.T.X., Bae, J.A., Kwon, D.D., and Jung, C. (2010). Differential CARM1 expression in prostate and colorectal cancers. BMC Cancer 10, 197.
Kuehne, A., Emmert, H., Soehle, J., Winnefeld, M., Fischer, F., Wenck, H., Gallinat, S., Terstegen, L., Lucius, R., Hildebrand, J., et al. (2015). Acute activation of oxidative pentose phosphate pathway as first-line response to oxidative stress in human skin cells. Mol Cell 59, 359–371.
Li, T.Y., Sun, Y., Liang, Y., Liu, Q., Shi, Y., Zhang, C.S., Zhang, C., Song, L., Zhang, P., Zhang, X., et al. (2016). ULK1/2 constitute a bifurcate node controlling glucose metabolic fluxes in addition to autophagy. Mol Cell 62, 359–370.
Lin, R., Elf, S., Shan, C., Kang, H.B., Ji, Q., Zhou, L., Hitosugi, T., Zhang, L., Zhang, S., Seo, J.H., et al. (2015). 6-Phosphogluconate dehydrogenase links oxidative PPP, lipogenesis and tumour growth by inhibiting LKB1-AMPK signalling. Nat Cell Biol 17, 1484–1496.
Liu, F., Ma, F., Wang, Y., Hao, L., Zeng, H., Jia, C., Wang, Y., Liu, P., Ong, I.M., Li, B., et al. (2017). PKM2 methylation by CARM1 activates aerobic glycolysis to promote tumorigenesis. Nat Cell Biol 19, 1358–1370.
Mitsuishi, Y., Taguchi, K., Kawatani, Y., Shibata, T., Nukiwa, T., Aburatani, H., Yamamoto, M., and Motohashi, H. (2012). Nrf2 redirects glucose and glutamine into anabolic pathways in metabolic reprogramming. Cancer Cell 22, 66–79.
Morales, Y., Cáceres, T., May, K., and Hevel, J.M. (2016). Biochemistry and regulation of the protein arginine methyltransferases (PRMTs). Archives Biochem Biophys 590, 138–152.
Naik, N., Shah, A., Wamelink, M.M.C., van der Knaap, M.S., and Hingwala, D. (2017). Rare case of ribose 5 phosphate isomerase deficiency with slowly progressive leukoencephalopathy. Neurology 89, 1195–1196.
Patra, K.C., and Hay, N. (2014). The pentose phosphate pathway and cancer. Trends Biochem Sci 39, 347–354.
Peng, C., and Wong, C.C. (2017). The story of protein arginine methylation: Characterization, regulation, and function. Expert Rev Proteomics 14, 157–170.
Qiu, Z., Guo, W., Wang, Q., Chen, Z., Huang, S., Zhao, F., Yao, M., Zhao, Y., and He, X. (2015). MicroRNA-124 reduces the pentose phosphate pathway and proliferation by targeting PRPS1 and RPIA mRNAs in human colorectal cancer cells. Gastroenterology 149, 1587–1598.e11.
Santana-Codina, N., Roeth, A.A., Zhang, Y., Yang, A., Mashadova, O., Asara, J.M., Wang, X., Bronson, R.T., Lyssiotis, C.A., Ying, H., et al. (2018). Oncogenic KRAS supports pancreatic cancer through regulation of nucleotide synthesis. Nat Commun 9, 4945.
Shishkova, E., Zeng, H., Liu, F., Kwiecien, N.W., Hebert, A.S., Coon, J.J., and Xu, W. (2017). Global mapping of CARM1 substrates defines enzyme specificity and substrate recognition. Nat Commun 8, 15571.
Spencer, N., and Hopkinson, D.A. (1980). Biochemical genetics of the pentose phosphate cycle: Human ribose 5-phosphate isomerase (RPI) and ribulose 5-phosphate 3-epimerase (RPE). Ann Human Genet 43, 335–342.
Szwarc, M.M., Kommagani, R., Putluri, V., Dubrulle, J., Stossi, F., Mancini, M.A., Coarfa, C., Lanz, R.B., Putluri, N., DeMayo, F.J., et al. (2018). Steroid receptor coactivator-2 controls the pentose phosphate pathway through RPIA in human endometrial cancer cells. Sci Rep 8, 13134.
Wang, L., Zhao, Z., Meyer, M.B., Saha, S., Yu, M., Guo, A., Wisinski, K. B., Huang, W., Cai, W., Pike, J.W., et al. (2014). CARM1 methylates chromatin remodeling factor BAF155 to enhance tumor progression and metastasis. Cancer Cell 25, 21–36.
Wang, Y.P., Zhou, W., Wang, J., Huang, X., Zuo, Y., Wang, T.S., Gao, X., Xu, Y.Y., Zou, S.W., Liu, Y.B., et al. (2016). Arginine methylation of MDH1 by CARM1 inhibits glutamine metabolism and suppresses pancreatic cancer. Mol Cell 64, 673–687.
Warburg, O. (1956). On the origin of cancer cells. Science 123, 309–314.
Yamamoto, T., Takano, N., Ishiwata, K., Ohmura, M., Nagahata, Y., Matsuura, T., Kamata, A., Sakamoto, K., Nakanishi, T., Kubo, A., et al. (2014). Reduced methylation of PFKFB3 in cancer cells shunts glucose towards the pentose phosphate pathway. Nat Commun 5, 3480.
Yang, Y., and Bedford, M.T. (2013). Protein arginine methyltransferases and cancer. Nat Rev Cancer 13, 37–50.
Yi, W., Clark, P.M., Mason, D.E., Keenan, M.C., Hill, C., Goddard, W.A., Peters, E.C., Driggers, E.M., and Hsieh-Wilson, L.C. (2012). Phosphofructokinase 1 glycosylation regulates cell growth and metabolism. Science 337, 975–980.
Ying, H., Kimmelman, A.C., Lyssiotis, C.A., Hua, S., Chu, G.C., Fletcher-Sananikone, E., Locasale, J.W., Son, J., Zhang, H., Coloff, J.L., et al. (2012). Oncogenic Kras maintains pancreatic tumors through regulation of anabolic glucose metabolism. Cell 149, 656–670.
Zhong, X.Y., Yuan, X. M., Xu, Y. Y., Yin, M., Yan, W. W., Zou, S. W., Wei, L. M., Lu, H.J., Wang, Y.P., and Lei, Q.Y. (2018). CARM1 methylates GAPDH to regulate glucose metabolism and is suppressed in liver cancer. Cell Rep 24, 3207–3223.
Acknowledgements
This work was supported by the Ministry of Science and Technology (2019YFA0801703), the National Natural Science Foundation of China (81790250, 81790253 and 91959202), and the Innovation Program of Shanghai Municipal Education Commission (N173606). We thank the members of the Cancer Metabolism Laboratory for discussions and support throughout this study. We thank Dr. Yanzhong Yang (City of Hope) for sharing plasmids encoding PRMTs.
Author information
Authors and Affiliations
Corresponding author
Additional information
Compliance and ethics
The procedures related to animal subjects of our study were approved by Ethic Committee of the Institutes of Biomedical Sciences (IBS), Fudan University, China. The author(s) declare that they have no conflict of interest.
Electronic supplementary material
11427_2019_1562_MOESM1_ESM.docx
Supplementary information for Arginine Methylation of Ribose-5-phosphate Isomerase A Senses Glucose to Promote Human Colorectal Cancer Cell Survival
Rights and permissions
About this article
Cite this article
Guo, J., Zhang, Q., Su, Y. et al. Arginine methylation of ribose-5-phosphate isomerase A senses glucose to promote human colorectal cancer cell survival. Sci. China Life Sci. 63, 1394–1405 (2020). https://doi.org/10.1007/s11427-019-1562-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11427-019-1562-y