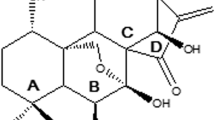
Abstract
Three new sesquiterpenes, valerianaterpenes I–III, and eight known compounds have been isolated from the methanol extract of the rhizomes and roots of Valeriana fauriei. The chemical structures of the three new sesquiterpenes were elucidated based on chemical and spectroscopic evidence. The absolute stereochemistry of valerianaterpene I was determined using X-ray crystallography. The cell death-inducing activity of isolated compounds alone or combination with Adriamycin (ADR) was observed by time-lapse cell imaging. Although the isolated compounds did not affect the number of mitotic entry cells and dead cells alone, kessyl glycol, kessyl glycol diacetate, and iso-teucladiol significantly increased the number of dead cells on ADR treated human cervical cancer cells. One of the mechanisms of cell death-inducing activity for the kessyl glycol acetate was suggested to be the inhibition of heat-shock protein 105 (Hsp105) expression level. This paper first deals with the naturally occurring compounds as Hsp105 inhibitor.






Similar content being viewed by others
References
Wang P-C, Ran X-H, Luo H-R, Hu J-M, Chen R, Ma Q-Y, Dai H-F, Liu Y-Q, Xie M-J, Zhou J, Zhao Y-X (2011) Volvalerelactones A and B, two new sesquiterpenoid lactones with an unprecedented skeleton from Valeriana officinalis var. latifolia. Org Lett 13:3036–3039
Piccinelli A-L, Arana S, Caceres A, di Villa Bianca Rd, Sorrentino R, Rastrelli L, (2004) New lignans from the roots of Valeriana prionophylla with antioxidative and vasorelaxant activities. J Nat Prod 67:1135–1140
Lee D-H, Park S-H, Huh Y-H, Kim M-J, Seo H-D, Ha T-Y, Ahn J, Jang Y-J, Jung C-H (2020) Iridoids of Valeriana fauriei contribute to alleviating hepatic steatosis in obese mice by lipophagy. Biomed Pharmacother 125:109950
Liu X-G, Gao P-Y, Wang G-S, Song S-J, Li L-Z, Li X, Yao X-S, Zhang Z-X (2012) In vivo antidepressant activity of sesquiterpenes from the roots of Valeriana fauriei Briq. Fitoterapia 83:599–603
Wang P-C, Ran X-H, Luo H-R, Ma Q-Y, Zhou J, Hu J-M, Zhao Y-X (2016) Volvalerine A, an unprecedented N-containing sesquiterpenoid dimer derivative from Valeriana officinalis var. latifolia. Fitoterapia 109:174–178
Oshima Y, Matsuoka S, Ohizumi Y (1995) Antidepressant principles of Valeriana fauriei roots. Chem Pharm Bull 43:169–170
Guo Y, Xu J, Li Y, Watanabe R, Oshima Y, Yamakuni T, Ohizumi Y (2006) Iridoids and sesquiterpenoids with NGF-potentiating activity from the rhizomes and roots of Valeriana fauriei. Chem Pharm Bull 54:123–125
Liu X-G, Zhang W-C, Gao P-Y, Wang G-S, Li L-Z, Song S-J, Zhang X, Yao X-S, Liu K, Zhang Z-X (2013) Two new sesquiterpenes from the roots of Valeriana fauriei Briq. Helv Chim Acta 96:651–655
Nishiya K, Kimura T, Takeya K, Itokawa H (1992) Sesquiterpenoids and iridoid glycosides from Valeriana faueriei. Phytochemistry 31:3511–3514
Matsumoto T, Kitagawa T, Imahori D, Matsuzaki A, Saito Y, Ohta T, Yoshida T, Nakayama Y, Ashihara E, Watanabe T (2021) Linderapyrone: A Wnt signal inhibitor isolated from Lindera umbellata. Bioorg Med Chem Lett 45:128161
Matsumoto T, Kitagawa T, Teo S, Anai Y, Ikeda R, Imahori D, Ahmad HSB, Watanabe T (2018) Structures and antimutagenic effects of onoceranoid-type triterpenoids from the leaves of Lansium domesticum. J Nat Prod 81:2187–2194
Matsumoto T, Imahori D, Saito Y, Zhang W, Ohta T, Yoshida T, Nakayama Y, Ashihara E, Watanabe T (2020) Cytotoxic activities of sesquiterpenoids from the aerial parts of Petasites japonicus against cancer stem cells. J Nat Med 74:689–701
Matsumoto T, Imahori D, Achiwa K, Saito Y, Ohta T, Yoshida T, Kojima N, Yamashita M, Nakayama Y, Watanabe T (2020) Chemical structures and cytotoxic activities of the constituents isolated from Hibiscus tiliaceus. Fitoterapia 142:104524
Damiani RM, Moura DJ, Viau CM, Caceres RA, Henriques JAP, Saffi J (2016) Pathways of cardiac toxicity: comparison between chemotherapeutic drugs doxorubicin and mitoxantrone. Arch Toxicol 90:2063–2076
Cheung KG, Cole LK, Xiang B, Chen K, Ma X, Myal Y, Hatch GM, Tong Q, Dolinsky VW (2015) Sirtuin-3 (SIRT3) protein attenuates doxorubicin-induced oxidative stress and improves mitochondrial respiration in H9c2 cardiomyocytes. J Biol Chem 290:10981–10993
Yamane T, Saito Y, Teshima H, Hagino M, Kakihana A, Sato S, Shimada M, Kato Y, Kuga T, Yamagishi N, Nakayama Y (2019) Hsp105α suppresses Adriamycin-induced cell death via nuclear localization signal-dependent nuclear accumulation. J Cell Biochem 120:17951–17962
Lee S-O, Choi S-Z, Choi S-U, Kim G-H, Kim Y-C, Lee K-R (2006) Cytotoxic terpene hydroperoxides from the aerial parts of Aster spathulifofius. Arc Pharm Res 29:845–848
Fokialakis N, Magiatis P, Mitaku S (2002) Essential oil constituents of Valeriana italica and Valeriana tuberosa. Stereochemical and conformational study of 15-acetoxyvaleranone. Z Naturforsch C 57:791–796
Dowling M-S, Vanderwal C-D (2010) Ring-closing metathesis of allylsilanes as a flexible strategy toward cyclic terpenes. short syntheses of teucladiol, isoteucladiol, poitediol, and dactylol and an attempted synthesis of caryophyllene. J Org Chem 75:6908–6922
Buchi G, White D-M (1957) The ring fission of cyclopropanes: the constitution of maaliol. J Am Chem Soc 79:750–751
Ito S, Kodama M, Nozoe T (1967) Structure and absolute configuration of α-kessyl alcohol and kessyl glycol. Tetrahedron lett 23:553–563
Takamura K, Kawaguchi M, Nabata H (1975) The preparation and pharmacological screening of kessoglycol derivative. Yakugaku Zasshi 95:1198–1204
Takamura K, Nabata H, Kawaguchi M (1975) The pharmaceutical action on the kessoglycol 8-monoacetate. Yakugaku Zasshi 95:1205–1209
Williams M-J, Deak H-L, Snapper M-L (2007) Intramolecular cyclobutadiene cycloaddition/cyclopropanation/thermal fragmentation: An effective strategy for the asymmetric synthesis of pleocarpenene and pleocarpenone. J Am Chem Soc 129:486–487
Silva M, Wiesenfeld A, Sammes P-G, Tyler T-W (1977) New sesquiterpenes from Pleocarpus revolutus. Phytochemistry 16:379–385
Aguilar-Guadarrama A-B, Rios M-Y (2004) Three new sesquiterpenes from Croton arboreous. J Nat Prod 67:914–917
Kitagawa T, Matsumoto T, Imahori D, Kobayashi M, Okayama M, Ohta T, Yoshida T, Watanabe T (2021) Limonoids isolated from the Fortunella crassifolia and the Citrus junos with their cell death inducing activity on Adriamycin treated cancer cell. J Nat Med, in press
Kai M, Nakatsura T, Egami H, Senju S, Nishimura Y, Ogawa M (2003) Heat shock protein 105 is overexpressed in a variety of human tumors. Oncol Rep 10:1777–1782
Yang S, Ren X, Liang Y, Yan Y, Zhou Y, Hu J, Wang Z, Song F, Wang F, Liao W, Liao W, Ding Y, Liang L (2020) KNK437 restricts the growth and metastasis of colorectal cancer via targeting DNAJA1/CDC45 axis. Oncogene 39:249–261
Saito Y, Yamagishi N, Hatayama T (2008) Nuclear localization mechanism of Hsp105β and its possible function in mammalian cells. J Biochem 145:185–191
Griffin T-M, Valdez T-V, Mestril R (2003) Radicicol activates heat shock protein expression and cardioprotection in neonatal rat cardiomyocytes. Amer J Physiol Heart Circ Phy 287:1081–1088
Acknowledgements
This work was supported by JSPS KAKENHI Grant Numbers 20H03397 and 20J14567.
Author information
Authors and Affiliations
Corresponding authors
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Matsumoto, T., Kitagawa, T., Imahori, D. et al. Cell death-inducing activities via Hsp inhibition of the sesquiterpenes isolated from Valeriana fauriei. J Nat Med 75, 942–948 (2021). https://doi.org/10.1007/s11418-021-01543-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11418-021-01543-9