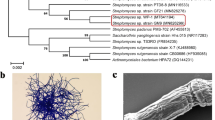
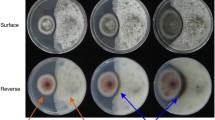
Abstract
Fungal co-culture is a strategy to induce the production of secondary metabolites by activating cryptic genes. We discovered the production of a new compound, talarodone A (1), along with five known compounds 2–6 in co-culture of Talaromyces pinophilus and Paraphaeosphaeria sp. isolated from soil collected in Miyazaki Prefecture, Japan. Among them, the productions of penicidones C (2) and D (3) were enhanced 27- and sixfold, respectively, by the co-culture. The structure of 3 should be represented as a γ-pyridol form with the reported chemical shifts, but not as a γ-pyridone form, based on DFT calculation.
Graphic abstract





Similar content being viewed by others
References
Newman DJ, Cragg GM (2016) Natural products as sources of new drugs from 1981 to 2014. J Nat Prod 79:629–661
Newman DJ (2016) Predominately uncultured microbes as sources of bioactive agents. Front Microbiol 7:1832
Cimermancic P, Medema MH, Claesen J, Kurita K, Wieland Brown LC, Mavrommatis K, Pati A, Godfrey PA, Koehrsen M, Clardy J, Birren BW, Takano E, Sali A, Linington RG, Fischbach MA (2014) Insights into secondary metabolism from a global analysis of prokaryotic biosynthetic gene clusters. Cell 158:412–421
Katz M, Hover BM, Brady SF (2016) Culture-independent discovery of natural products from soil metagenomes. J Ind Microbiol Biotechnol 43:129–141
Scherlach K, Hertweck C (2009) Triggering cryptic natural product biosynthesis in microorganisms. Org Biomol Chem 7:1753–1760
Moon K, Xu F, Seyedsayamdost MR (2019) Cebulantin, a cryptic lanthipeptide antibiotic uncovered using bioactivity-coupled HiTES. Angew Chem Int Ed 58:5973–5977
Li HT, Zhou H, Duan RT, Li HY, Tang LH, Yang XQ, Yang YB, Ding ZT (2019) Inducing secondary metabolite production by co-culture of the endophytic fungus Phoma sp. and the symbiotic fungus Armillaria sp. J Nat Prod 82:1009–1013
Bertrand S, Schumpp O, Bohni N, Monod M, Gindro K, Wolfender JL (2013) De novo production of metabolites by fungal co-culture of Trichophyton rubrum and Bionectria ochroleuca. J Nat Prod 76:1157–1165
Sadahiro Y, Kato H, Williams RM, Tsukamoto S (2020) Irpexine, a new isoindolinone alkaloid produced by co-culture of endophytic fungi, Irpex lacteus and Phaeosphaeria oryzae. submitted
Ge HM, Shen Y, Zhu CH, Tan SH, Ding H, Song YC, Tan RX (2008) Penicidones A-C, three cytotoxic alkaloidal metabolites of an endophytic Penicillium sp. Phytochemistry 69:571–576
Liu Y, Yang Q, Xia G, Huang H, Li H, Ma L, Lu Y, He L, Xia X, She Z (2015) Polyketides with α-glucosidase inhibitory activity from a mangrove endophytic fungus, Penicillium sp. HN29-3B1. J Nat Prod 78:1816–1822
Buommino E, Tirino V, De Filippis A, Silvestri F, Nicoletti R, Ciavatta ML, Pirozzi G, Tufano MA (2011) 3-O-Methylfunicone, from Penicillium pinophilum, is a selective inhibitor of breast cancer stem cells. Cell Prolif 44:401–409
Chen MJ, Fu YW, Zhou QY (2014) Penifupyrone, a new cytotoxic funicone derivative from the endophytic fungus Penicillium sp. HSZ-43. Nat Prod Res 28:1544–1548
Myobatake Y, Takeuchi T, Kuramochi K, Kuriyama I, Ishido T, Hirano K, Sugawara F, Yoshida H, Mizushina Y (2012) Pinophilins A and B, inhibitors of mammalian A-, B-, and Y-family DNA polymerases and human cancer cell proliferation. J Nat Prod 75:135–141
Torii M, Kato H, Hitora Y, Angkouw ED, Mangindaan REP, de Voogd NJ, Tsukamoto S (2017) Lamellodysidines A and B, sesquiterpenes isolated from the marine sponge Lamellodysidea herbacea. J Nat Prod 80:2536–2541
Acknowledgements
This work was supported by JSPS KAKENHI Grants 17H03994 (S.T.), 18K06719 (H.K.), and 18K14933 (Y. H.), from the Noda Institute for Scientific Research (S.T.), and Useful and Unique Natural Products for Drug Discovery and Development (UpRod), Program for Building Regional Innovation Ecosystems at Kumamoto University, Japan. We are grateful to Dr. K. Miyazaki and Dr. A. Kinoshita of Kyushu Research Center, Forestry and Forest Products Research Institute, Japan, for identification of the fungal isolates.
Author information
Authors and Affiliations
Corresponding authors
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Murakami, S., Hayashi, N., Inomata, T. et al. Induction of secondary metabolite production by fungal co-culture of Talaromyces pinophilus and Paraphaeosphaeria sp.. J Nat Med 74, 545–549 (2020). https://doi.org/10.1007/s11418-020-01400-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11418-020-01400-1