Abstract
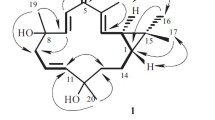
Two new alkyl coumarins, isophellodenol C (1) and candinol A (2); four new spirobifuranocoumarins, candibirins B–E (3-6); and two trifuranocoumarins, canditririns A and B (7 and 8), were isolated from the roots of Heracleum candicans WALL. Their structures were established using chemical and spectral means.









Similar content being viewed by others
References
Zhonghuabencao Editorial Board (1999). Zhonghuabencao, Vol. 5. Shanghai Scientific and Technological Press, Shanghai (in Chinese) p. 958
Nath Y, Nazir BN, Handa KL (1961) Bergapten from the fruits of Heracleum candicans. Indian J Pharm 23:303–304
Sharma YH, Zaman A, Kidwai AR (1964) Examination of Heracleum candicans. I. Isolation and structure of a new furocoumarin, heraclenin. Tetrahedron 20:87–90
Sharma YN, Sharma RC, Zaman Z, Kidwai AR (1964) Chemical examination of Heracleum candicans. II. Isolation and structure of a new furocoumarin, heraclenol. Naturwissenschaften 51:537
Sharma YN, Zaman A, Kidwai AR, Bates RB, Thalacker VP (1966) Coumarin constituents of Heracleum candicans III. Tetrahedron 22:3221–3225
Bandopadhyay M, Seshadri TR (1970) Components of Heracleum candicans. I. Indian J Chem 8:855–856
Bandopadhyay M, Seshadri TR (1970) Components of Heracleum candicans II. Indian J Chem 8:1146–1147
Bandopadhyay M, Malik SB, Seshadri TR (1971) Minor components of Heracleum candicans. I. Indian J Chem 9:731–732
Bandopadhyay M, Malik SB, Seshadri TR (1971) Candicanin, a novel bicoumarinyl derivative from the roots of Heracleum candicans. Tetrahedron Lett 45:4221–4222
Bandopadhyay M, Malik SB, Seshadri TR (1973) Coumarins from the roots and seeds of Heracleum candicans. Indian J Chem 11:410–412
Bandopadhyay M, Malik SB, Seshadri TR (1973) Candicopimaric acid Diterpene acid from the roots and seeds of Heracleum candicans. Indian J Chem 11:1097–1098
Sun H, Lin Z, Niu F (1984) Study of traditional Chinese drugs of Umbelliferae. VII. Chemical constituents of the roots of Heracleum candicans wall. (1). Yunnan Zhiwu Yanjiu 6:99–102
Lin Z, Gao L, Chen Y, Rao G, Pu F, Sun H (1993) Coumarins of Heracleum candicans. 15:313–314
Bal-Tenbe S, Joshi DD, Lakdawala AD (1996) Isolation of the anti-inflammatory principle of Heracleum candicans. Indian J Chem 35B:518–519
Doi M, Nakamori T, Shibano M, Taniguchi M, Wang N-H, Baba K (2004) Candibirin A, a furanocoumarin dimer isolated from Heracleum candicans WALL. Acta Crystallogr C 60:o833–o835
Wu T-S, Hsu M-Y, Kuo P-C, Sreenivasulu B, Damu AG, Su C-R, Li C-Y, Chang H-C (2003) Constituents from the Leaves of Phellodendron amurense var wilsonii and their Bioactivity. J Nat Prod 66:1207–1211
Dale JA, Mosher HS (1973) Nuclear magnetic resonance enantiomer regents. Configurational correlations via nuclear magnetic resonance chemical shifts of diastereomeric mandelate, O-methylmandelate, and α-methoxy-α-trifluoromethyl–phenylacetate (MTPA) esters. J Am Chem Soc 95:542–519
Ohtani I, Kusumi T, Kashman Y, Kakisawa H (1991) High-field FT NMR application of Mosher’s method The absolute configurations of marine terpenoids. J Am Chem Soc 113:4092–4096
Ohtani I, Kusumi T, Kashman Y, Kakisawa H (1991) A new aspect of the high-field NMR application of Mosher’s method The absolute configuration of marin triterpene sipholenol A. J Org Chem 56:1296–1298
Zhou P, Takaishi Y, Duan H, Chen B, Honda G, Itoh M, Takeda Y, Kodzhimatov OK, Lee K-H (2000) Coumarins and bicoumarin from Ferula sumbul: anti-HIV activity and inhibition of cytokine release. Phytochemistry 53:689–697
Acknowledgments
The authors are grateful to Mr. K. Minoura for NMR spectral recordings and to Mrs. M. Fujitake for mass spectra recordings at Osaka University of Pharmaceutical Sciences. This work was partially supported by NSFC (project 30270097), China.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Nakamori, T., Taniguchi, M., Shibano, M. et al. Chemical studies on the root of Heracleum candicans WALL.. J Nat Med 62, 403–412 (2008). https://doi.org/10.1007/s11418-008-0266-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11418-008-0266-8