Abstract
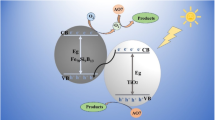
Roxarsone (3-nitro-4-hydroxyphenylarsonic acid) is an extensively used organoarsenic feed additive. The effective removal of arsenic from roxarsone degradation before discharging is of great importance for controlling artificial arsenic pollution in aquatic environment. In this study, a bifunctional TiO2/ferrihydrite (TiO2/FeOOH) hybrid was synthesized by a hydrothermal method for the simultaneously photocatalytic degradation of roxarsone and adsorption removal of released arsenic. The analysis of the prepared TiO2/FeOOH by field-emission scanning electron microscope (FE-SEM), transmission electron microscopy (TEM), Raman spectra, X-ray diffraction (XRD), diffuse reflectance spectroscopy (DRS), and X-ray photoelectron spectroscopy (XPS) confirmed the successful formation of the hybrid of crystalline TiO2 and no crystalline FeOOH. TiO2/FeOOH hybrid had better adsorption capacity for As(V) than roxarsone. Compared to TiO2, the TiO2/FeOOH hybrid exhibited much superior UV-driven photocatalytic activities for roxarsone degradation. After 12 h irradiation, more than 96% of roxarsone was degraded by 1:1 TiO2/FeOOH hybrid, and the released As(V) was simultaneously removed from the solution. The residual As(V) concentration was lower than 0.02 mg L−1. The reusability test indicated that TiO2/FeOOH hybrid had excellent stability and reliability. The possible mechanism of roxarsone degradation and released inorganic arsenics removal by this hybrid was also proposed. These results clearly indicated that the TiO2/FeOOH hybrid could be used for the removal of roxarsone and its degradation product.








Similar content being viewed by others
References
Adak A, Mangalgiri KP, Lee J, Blaney L (2015) UV irradiation and UV-H(2)O(2) advanced oxidation of the roxarsone and nitarsone organoarsenicals. Water Res 70:74–85. https://doi.org/10.1016/j.watres.2014.11.025
Bednar AJ, Garbarino JR, Ferrer I, Rutherford DW, Wershaw RL, Ranville JF, Wildeman TR (2003) Photodegradation of roxarsone in poultry litter leachates Sci Total Environ 302:237–245. https://doi.org/10.1016/s0048-9697(02)00322-4
Bhandari N, Reeder RJ, Strongin DR (2011) Photoinduced oxidation of arsenite to arsenate on ferrihydrite Environ Sci Technol 45:2783–2789. https://doi.org/10.1021/es103793y
Cortinas I, Field JA, Kopplin M, Garbarino JR, Gandolfi AJ, Sierra-Alvarez R (2006) Anaerobic biotransformation of roxarsone and related N-substituted phenylarsonic acids Environ Sci Technol 40:2951–2957. https://doi.org/10.1021/es051981o
Czaplicka M, Bratek Ł, Jaworek K, Bonarski J, Pawlak S (2014) Photo-oxidation of p-arsanilic acid in acidic solutions: kinetics and the identification of by-products and reaction pathways. Chem Eng J 243:364–371. https://doi.org/10.1016/j.cej.2014.01.016
Czaplicka M, Jaworek K, Bak M (2015) Study of photodegradation and photooxidation of p-arsanilic acid in water solutions at pH = 7: kinetics and by-products. Environ Sci Pollut Res Int 22:16927–16935. https://doi.org/10.1007/s11356-015-4890-z
Das S, Hendry MJ (2011) Application of Raman spectroscopy to identify iron minerals commonly found in mine wastes. Chem Geol 290:101–108. https://doi.org/10.1016/j.chemgeo.2011.09.001
Dixit S, Hering JG (2003) Comparison of arsenic(V) and arsenic(III) sorption onto iron oxide minerals: implications for arsenic mobility. Environ Sci Technol 37:4182–4189. https://doi.org/10.1021/es030309t
Francisco MSP, Mastelaro VR, Nascente PAP, Florentino AO (2001) Activity and characterization by XPS, HR-TEM, Raman spectroscopy, and BET surface area of CuO/CeO2-TiO2 catalysts. J Phys Chem B 105:10515–10522. https://doi.org/10.1021/jp0109675
Frau F, Addari D, Atzei D, Biddau R, Cidu R, Rossi A (2010) Influence of major anions on As(V) adsorption by synthetic 2-line ferrihydrite. Kinetic investigation and XPS study of the competitive Effect of bicarbonate Water Air Soil Pollut 205:25-41. https://doi.org/10.1007/s11270-009-0054-4
Friedmann D, Mendive C, Bahnemann D (2010) TiO2 for water treatment: parameters affecting the kinetics and mechanisms of photocatalysis. Appl Catal B-Environ 99:398–406. https://doi.org/10.1016/j.apcatb.2010.05.014
He P, Wu GX, Tang R, Ji PL, Yuan SJ, Wang W, Hu ZH (2018) Influence of arsanilic acid, Cu2+, PO43- and their interaction on anaerobic digestion of pig manure Frontiers of Environmental Science & Engineering 12. https://doi.org/10.1007/s11783-017-1004-9
Hou Y, Li X-Y, Zhao Q-D, Quan X, Chen G-H (2010) Electrochemical method for synthesis of a ZnFe2O4/TiO2 composite nanotube array modified electrode with enhanced photoelectrochemical activity, Advanced Functional Materials. 20:2165–2174. https://doi.org/10.1002/adfm.200902390
Huang C, Wang Y, Gong M, Wang W, Mu Y, Hu Z-H (2020) α-MnO2/Palygorskite composite as an effective catalyst for heterogeneous activation of peroxymonosulfate (PMS) for the degradation of Rhodamine B. Sep Purif Technol 230. https://doi.org/10.1016/j.seppur.2019.115877
Ji PL, Tang R, He P, Yuan SJ, Wang W, Hu ZH (2017) Characterization of arsenic species in the anaerobic granular sludge treating roxarsone-contaminated wastewater. Chem Eng J 327:162–168. https://doi.org/10.1016/j.cej.2017.06.087
Jin JY, Zhang SJ, Wu BD, Chen ZH, Zhang GY, Tratnyek PG (2019) Enhanced photooxidation of hydroquinone by acetylacetone, a novel photosensitizer and electron shuttle Environ Sci Technol 53:11232-11239. https://doi.org/10.1021/acs.est.9b02751
Li MH, Zhang SJ, Peng Y, Lv L, Pan BC (2015) Enhanced visible light responsive photocatalytic activity of TiO2-based nanocrystallites: impact of doping sequence. RSC Adv 5:7363–7369. https://doi.org/10.1039/c4ra10604k
Li R et al (2019) Construction of heterostructured CuFe2O4/g-C3N4 nanocomposite as an efficient visible light photocatalyst with peroxydisulfate for the organic oxidation. Appl Catal B Environ 244:974–982. https://doi.org/10.1016/j.apcatb.2018.12.043
Liu G, Wang LZ, Yang HG, Cheng HM, Lu GQ (2010) Titania-based photocatalysts-crystal growth, doping and heterostructuring. J Mater Chem 20:831–843. https://doi.org/10.1039/b909930a
Liu XP, Zhang WF, Hu YN, Cheng HF (2013) Extraction and detection of organoarsenic feed additives and common arsenic species in environmental matrices by HPLC-ICP-MS. Microchem J 108:38–45. https://doi.org/10.1016/j.microc.2012.12.005
Liu Y, Hu P, Zheng J, Wu M, Jiang B (2016) Utilization of spent aluminum for p-arsanilic acid degradation and arsenic immobilization mediated by Fe(II) under aerobic condition. Chem Eng J 297:45–54. https://doi.org/10.1016/j.cej.2016.03.092
Lu DL, Ji F, Wang W, Yuan SJ, Hu ZH, Chen TH (2014) Adsorption and photocatalytic decomposition of roxarsone by TiO2 and its mechanism. Environ Sci Pollut Res 21:8025–8035. https://doi.org/10.1007/s11356-014-2729-7
Lv YC et al (2018) Removal of p-arsanilic acid by an amino-functionalized indium-based metal-organic framework: adsorption behavior and synergetic mechanism. Chem Eng J 339:359–368. https://doi.org/10.1016/j.cej.2018.01.139
Malato S, Fernandez-Ibanez P, Maldonado MI, Blanco J, Gernjak W (2009) Decontamination and disinfection of water by solar photocatalysis: recent overview and trends Catal Today 147:1–59. https://doi.org/10.1016/j.cattod.2009.06.018
Muller K, Ciminelli VST, Dantas MSS, Willscher S (2010) A comparative study of As(III) and As(V) in aqueous solutions and adsorbed on iron oxy-hydroxides by Raman spectroscopy. Water Res 44:5660–5672. https://doi.org/10.1016/j.watres.2010.05.053
Nanda B, Pradhan AC, Parida KM (2016) A comparative study on adsorption and photocatalytic dye degradation under visible light irradiation by mesoporous MnO 2 modified MCM-41 nanocomposite. Microporous Mesoporous Mater 226:229–242. https://doi.org/10.1016/j.micromeso.2015.12.027
Peng HL et al (2013) High performance Fe- and N- doped carbon catalyst with graphene structure for oxygen reduction. Sci Rep 3. https://doi.org/10.1038/srep01765
Rezig W, Hadjel M (2014) Photocatalytic degradation of Vat Green 03 textile dye, using the ferrihydrite-modified diatomite with TiO2 /UV process. Orient J Chem 30:993–1007. https://doi.org/10.13005/ojc/300310
Rincon AG, Pulgarin C (2004) Effect of pH, inorganic ions, organic matter and H2O2 on E-coli K12 photocatalytic inactivation by TiO2 - implications in solar water disinfection. Appl Catal B-Environ 51:283–302. https://doi.org/10.1016/j.apcatb.2004.03.007
Rout K, Mohapatra M, Layek S, Dash A, Verma HC, Anand S (2014) The influence of precursors on phase evolution of nano iron oxides/oxyhydroxides: optical and magnetic properties New J Chem 38:3492–3506. https://doi.org/10.1039/c4nj00526k
Shi L, Wang W, Yuan SJ, Hu ZH (2014) Electrochemical stimulation of microbial roxarsone degradation under anaerobic conditions. Environ Sci Technol 48:7951–7958. https://doi.org/10.1021/es501398j
Silbergeld EK, Nachman K (2008) The environmental and public health risks associated with arsenical use in animal feeds. In: Carpenter DO (ed) Environmental challenges in the Pacific Basin, Annals of the New York Academy of Sciences, vol 1140. Wiley-Blackwell, Malden, pp 346–357. https://doi.org/10.1196/annals.1454.049
Stolz JF et al (2007) Biotransformation of 3-nitro-4-hydroxybenzene arsonic acid (roxarsone) and release of inorganic arsenic by Clostridium species. Environ Sci Technol 41:818–823. https://doi.org/10.1021/es061802i
Tang R, Chen H, Yuan SJ, Zhan XM, Wang W, Hu ZH (2017) Arsenic accumulation and volatilization in a 260-day cultured upflow anaerobic sludge blanket (UASB) reactor. Chem Eng J 311:277–283. https://doi.org/10.1016/j.cej.2016.11.097
Vaiano V, Iervolino G, Sannino D, Rizzo L, Sarno G, Farina A (2014) Enhanced photocatalytic oxidation of arsenite to arsenate in water solutions by a new catalyst based on MoOx supported on TiO2. Appl Catal B-Environ 160:247–253. https://doi.org/10.1016/j.apcatb.2014.05.034
Wang FM, Chen ZL, Sun YX, Gao YL, Yu JX (2006) Investigation on the pollution of organoarsenical additives to animal feed in the surroundings and farmland near hog farms. Acta Ecol Sin 26:154–162
Wang XP, Tang YX, Chen Z, Lim T (2012) Highly stable heterostructured Ag-AgBr/TiO 2 composite: a bifunctional visible-light active photocatalyst for destruction of ibuprofen and bacteria. J Mater Chem 22:23149–23158. https://doi.org/10.1039/C2JM35503E
Wang YJ, Ji F, Wang W, Yuan SJ, Hu ZH (2016) Removal of roxarsone from aqueous solution by Fe/La-modified montmorillonite. Desalin Water Treat 57:20520–20533. https://doi.org/10.1080/19443994.2015.1108875
Xie X, Hu Y, Cheng H (2016) Rapid degradation of p-arsanilic acid with simultaneous arsenic removal from aqueous solution using Fenton process. Water Res 89:59–67. https://doi.org/10.1016/j.watres.2015.11.037
Yu MD, Jia JH, Liu XY, Cui JX, Xi BD, He XS, Mao XH (2019) p-Arsanilic acid degradation and arsenic immobilization by a disilicate-assisted iron/aluminum electrolysis process. Chem Eng J 368:428–437. https://doi.org/10.1016/j.cej.2019.02.159
Funding
This research was supported by the National Natural Science Foundation of China (Grant Nos. 51538012, 51578205, and 51728801).
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible editor: Suresh Pillai
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Fu, W., Lu, DL., Yao, H. et al. Simultaneous roxarsone photocatalytic degradation and arsenic adsorption removal by TiO2/FeOOH hybrid. Environ Sci Pollut Res 27, 18434–18442 (2020). https://doi.org/10.1007/s11356-020-08310-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-020-08310-5