Abstract
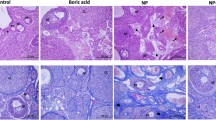
Lead (Pb) is an environmental pollutant. Selenium (Se) has alleviative effect on Pb poisoning. However, mitigative effect of Se on Pb-induced apoptosis has not been unclear via endoplasmic reticulum (ER) pathway in chicken testes. The aim of this study was to investigate mitigative effect of Se on apoptosis induced by Pb poisoning via ER pathway in chicken testes. Sixty male chickens (7-day-old) were randomly divided into the control group offered drinking water (DW) and basic diet (BD) (0.49 mg/kg Se), the Se group offered DW and BD containing Na2SeO3 (SeBD) (1.00 mg/kg Se), the Pb group offered DW containing (CH3OO)2Pb (PbDW) (350.00 mg/L Pb) and BD, and the Pb + Se group offered PbDW and SeBD; and were fed for 90 days. The following contents were performed as follows: histology; antioxidant indexes (reduced glutathione (GSH), malondialdehyde (MDA), glutathione peroxidase (GPx), glutathione S-transferase (GST), and superoxide dismutase (SOD)); mRNA expressions of ER-related genes (glucose-related protein 78 (GRP78), protein kinase-like ER kinase (PERK), eukaryotic initiation factor 2α (eIF2α), activating transcription factor 4 (ATF4), and enhancer-binding protein homologous protein (CHOP)); and apoptosis-related genes (cysteine-aspartic protease (caspase)-3 and caspase-12) in chicken testes. The results indicated that Pb poisoning caused histological changes; increased MDA content; decreased the content of GSH and the activities of GPx, GST, and SOD; and upregulated mRNA expressions of the above five ER-related genes and two apoptosis-related genes in the chicken testes. Se alleviated Pb-induced oxidative stress, ER stress, and apoptosis via CHOP/caspase-3 signal pathway in the chicken testes.





Similar content being viewed by others
References
Butkauskas D, Sruoga A (2004) Effect of lead and chromium on reproductive success of Japanese quail. Environ Toxicol 19:412–415. https://doi.org/10.1002/tox.20021
Corsetti G, Romano C, Stacchiotti A, Pasini E, Dioguardi FS (2017) Endoplasmic reticulum stress and apoptosis triggered by sub-chronic lead exposure in mice spleen: a histopathological study. Biol Trace Elem Res 178:86–97. https://doi.org/10.1007/s12011-016-0912-z
Flora SJ, Singh S, Tandon SK (1983) Role of selenium in protection against lead intoxication. Acta Pharmacol Toxicol (Copenh) 53(1):28–32
Gasparik J, Venglarcik J, Slamecka J, Kropil R, Smehyl P, Kopecky J (2012) Distribution of lead in selected organs and its effect on reproduction parameters of pheasants (Phasianus colchicus) after an experimental per oral administration. J Environ Sci Health A Toxicol Hazard Subst Environ Eng 47:1267–1271. https://doi.org/10.1080/10934529.2012.672127
Hanada S, Harada M, Kumemura H, Bishr O, Mary M, Koga H, Kawaguchi T, Taniguchi E, Yoshida T, Hisamoto T, Yanagimoto C, Maeyama M, Ueno T, Sata M (2007) Oxidative stress induces the endoplasmic reticulum stress and facilitates inclusion formation in cultured cells. J Hepatol 47:93–102. https://doi.org/10.1016/j.jhep.2007.01.039
Jiao XY, Yang KX, An Y, Teng XJ, Teng XH (2017) Alleviation of lead-induced oxidative stress and immune damage by selenium in chicken bursa of Fabricius. Environ Sci Pollut Res Int 24:7555–7564. https://doi.org/10.1007/s11356-016-8329-y
Jin X, Xu Z, Zhao X, Chen MH, Xu SW (2017) The antagonistic effect of selenium on lead-induced apoptosis via mitochondrial dynamics pathway in the chicken kidney. Chemosphere 180:259–266. https://doi.org/10.1016/j.chemosphere.2017.03.130
Klaassen CD, Watkins JB (2013) Casarett and Doull’s toxicology: the basic science of poisons. McGraw-Hill 35:477–25256. https://doi.org/10.1042/bst0090255a
Liu CM, Ma JQ, Liu SS, Zheng GH, Feng ZJ, Sun JM (2014) Proanthocyanidins improves lead-induced cognitive impairments by blocking endoplasmic reticulum stress and nuclear factor-κB-mediated inflammatory pathways in rats. Food Chem Toxicol 72:295–302. https://doi.org/10.1016/j.fct.2014.07.033
Liu CM, Zheng GH, Ming QL, Sun JM, Cheng C (2013) Protective effect of quercetin on lead-induced oxidative stress and endoplasmic reticulum stress in rat liver via the IRE1/JNK and PI3K/Akt pathway. Free Radic Res 47:192–201. https://doi.org/10.3109/10715762.2012.760198
Liu CM, Ma JQ, Sun JM, Feng ZJ, Cheng C, Yang W, Jiang H (2017a) Association of changes in ER stress-mediated signaling pathway with lead-induced insulin resistance and apoptosis in rats and their prevention by A-type dimeric epigallocatechin-3-gallate. Food Chem Toxicol 110:325–332. https://doi.org/10.1016/j.fct.2017.10.040
Liu Y, Pan X, Li S, Yu Y, Chen J, Yin J, Li G (2017b) Endoplasmic reticulum stress restrains hepatocyte growth factor expression in hepatic stellate cells and rat acute liver failure model. Chem Biol Interact 277:43–54. https://doi.org/10.1016/j.cbi.2017.08.015
Mabrouk A, Cheikh HB (2015) Thymoquinone supplementation reverses lead-induced oxidative stress in adult rat testes. Gen Physiol Biophys 34:65–72. https://doi.org/10.4149/gpb_2014022
McKelvey SM, Horgan KA, Murphy RA (2015) Chemical form of selenium differentially influences DNA repair pathways following exposure to lead nitrate. J Trace Elem Med Biol 29:151–169
Özkan-Yilmaz F, Özlüer-Hunt A, Gündüz SG, Berköz M, Yalin S (2014) Effects of dietary selenium of organic form against lead toxicity on the antioxidant system in Cyprinus carpio. Fish Physiol Biochem 40:355–363. https://doi.org/10.1007/s10695-013-9848-9
Pawlas N, Dobrakowski M, Kasperczyk A, Kozłowska A, Mikołajczyk A, Kasperczyk S (2016) The level of selenium and oxidative stress in workers chronically exposed to lead. Biol Trace Elem Res 170:1–8. https://doi.org/10.1007/s12011-015-0435-z
Rahman S, Sultana S (2006) Chemopreventive activity of glycyrrhizin on lead acetate mediated hepatic oxidative stress and its hyperproliferative activity in Wistar rats. Chem Biol Interact 160:61–69. https://doi.org/10.1016/j.cbi.2005.12.003
Shen W, Chen J, Yin J, Wang SL (2016) Selenium protects reproductive system and foetus development in a rat model of gestational lead exposure. EurRev Med Pharmacol Sci 20:773–780
Vengris VE, Mare CJ (1974) Lead poisoning in chickens and the effect of lead on interferon and antibody production. Can J Comp Med 38:328–335
Wang L, Wang H, Hu MZ, Cao J, Chen DW, Liu ZP (2009) Oxidative stress and apoptotic changes in primary cultures of rat proximal tubular cells exposed to lead. Arch Toxicol 83:417–427. https://doi.org/10.1007/s00204-009-0425-z
Wang M, Fu H, Xiao Y, Ai B, Wei Q, Wang S, Liu T, Ye L, Hu Q (2013) Effects of low-level organic selenium on lead-induced alterations in neural cell adhesion molecules. Brain Res 1530:76–81
Wang MG, Li WH, Wang XY, Yang DB, Wang ZY, Wang L (2017a) CaMKII is involved in subcellular Ca2+ redistribution-induced endoplasmic reticulum stress leading to apoptosis in primary cultures of rat proximal tubular cells exposed to lead. Oncotarget 8:91162–91173. https://doi.org/10.18632/oncotarget.20035. eCollection 2017 Oct 31.
Wang X, An Y, Jiao W, Zhang Z, Han H, Gu X, Teng X (2018) Selenium protects against lead-induced apoptosis via endoplasmic reticulum stress in chicken kidneys. Biol Trace Elem Res 182:354–363. https://doi.org/10.1007/s12011-017-1097-9
Wang Y, Wang K, Huang H, Gu X, Teng X (2017b) Alleviative effect of selenium on inflammatory damage caused by lead via inhibiting inflammatory factors and heat shock proteins in chicken testes. Environ Sci Pollut Res Int 24:13405–13413. https://doi.org/10.1007/s11356-017-8785-z
Xu T, Gao X, Liu G (2016) The antagonistic effect of selenium on lead toxicity is related to the ion profile in chicken liver. Biol Trace Elem Res 169:365–373. https://doi.org/10.1007/s12011-015-0422-4
Yao HD, Wu Q, Zhang ZW, Li S, Wang XL, Lei XG, Xu SW (2013a) Selenoprotein W serves as an antioxidant in chicken myoblasts. Biochim Biophys Acta 1830:3112–3120. https://doi.org/10.1016/j.bbagen.2013.01.007
Yao HD, Wu Q, Zhang ZW, Zhang JL, Li S, Huang JQ, Ren FZ, Xu SW, Wang XL, Lei XG (2013b) Gene expression of endoplasmic reticulum resident selenoproteins correlates with apoptosis in various muscles of Se-deficient chicks. J Nutr 143:613–619. https://doi.org/10.3945/jn.112.172395
Yao L, Du Q, Yao H, Chen X, Zhang Z, Xu S (2015) Roles of oxidative stress and endoplasmic reticulum stress in selenium deficiency-induced apoptosis in chicken liver. Biometals 28:255–265. https://doi.org/10.1007/s10534-014-9819-3
Yi S, Shi W, Wang H, Ma C, Zhang X, Wang S, Cong B, Li Y (2017) Endoplasmic reticulum stress PERK-ATF4-CHOP pathway is associated with hypothalamic neuronal injury in different durations of stress in rats. Front Neurosci 11:152. https://doi.org/10.3389/fnins.2017.00152. eCollection 2017
Zhong B, Wang X, Mao H, Wan Y, Liu Y, Zhang T, Hu C (2017) A mechanism underlies fish GRP78 protection against Pb2+ toxicity. Fish Shellfish Immunol 66:185–188. https://doi.org/10.1016/j.fsi.2017.03.056
Funding
The work was supported by the Heilongjiang Province on Natural Fund Project of China (no. C201420).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
All procedures used in this experiment were approved by the Northeast Agricultural University’s Institutional Animal Care and Use Committee under the approved protocol number SRM-06.
Conflict of interest
The authors declare that they have no conflicts of interest.
Additional information
Responsible editor: Philippe Garrigues
All authors have read the manuscript and have agreed to submit it in its current form for consideration for publication in the journal.
Rights and permissions
About this article
Cite this article
Huang, H., An, Y., Jiao, W. et al. CHOP/caspase-3 signal pathway involves in mitigative effect of selenium on lead-induced apoptosis via endoplasmic reticulum pathway in chicken testes. Environ Sci Pollut Res 25, 18838–18845 (2018). https://doi.org/10.1007/s11356-018-1950-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-018-1950-1