Abstract
Introduction
While the evolutionary adaptation of enzymes to their own substrates is a well assessed and rationalized field, how molecules have been originally selected in order to initiate and assemble convenient metabolic pathways is a fascinating, but still debated argument.
Objectives
Aim of the present study is to give a rationale for the preferential selection of specific molecules to generate metabolic pathways.
Methods
The comparison of structural features of molecules, through an inductive methodological approach, offer a reading key to cautiously propose a determining factor for their metabolic recruitment.
Results
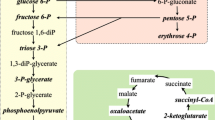
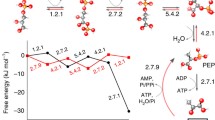
Starting with some commonplaces occurring in the structural representation of relevant carbohydrates, such as glucose, fructose and ribose, arguments are presented in associating stable structural determinants of these molecules and their peculiar occurrence in metabolic pathways.
Conclusions
Among other possible factors, the reliability of the structural asset of a molecule may be relevant or its selection among structurally and, a priori, functionally similar molecules.



Similar content being viewed by others
References
Agranoff, B. W., & Brady, R. O. (1956). Purification and properties of calf liver ribokinase. Journal of Biological Chemistry, 219, 221–229.
Angyal, S. J., & Bethell, G. S. (1976). Conformational analysis in carbohydrate chemistry III. The 13C NMR spectra of the hexuloses. Australian Journal of Chemistry, 29, 1249–1265.
Angyal, S. J., & Pickles, V. A. (1972). Equilibria between pyranoses and furanoses. Australian Journal of Chemistry, 25, 1695–1710.
Balestri, F., Cappiello, M., Meschini, R., Rotondo, R., Abate, M., Del Corso, A., & Mura, U. (2015). Modulation of aldose reductase activity by aldose hemiacetals. Biochimica et Biophysica Acta, 1850, 2329–2339.
Bar-Even, A., Flamholz, A., Noor, E., & Milo, R. (2012). Rethinking glycolysis: on the biochemical logic of metabolic pathways. Nature Chemical Biology, 8, 509–517.
Brownlee, M. (2005). The pathobiology of diabetic complications: a unifying mechanism. Diabetes, 54, 1615–1625.
Bunn, H. F., & Higgins, P. J. (1981). Reaction of monosaccharides with proteins: possible evolutionary significance. Science, 213, 222–224.
Campos, V. C., & Tappy, L. (2016). Physiological handling of dietary fructose-containing sugars: implications for health. Internationl Journal of Obesity, 40, S6–S11.
Copley, S. D. (2012). Toward a systems biology perspective on enzyme evolution. Journal of Biological Chemistry, 287, 3–10.
Del Corso, A., Cappiello, M., & Mura, U. (2008). From a dull enzyme to something else: facts and perspectives regarding aldose reductase. Current Medicinal Chemistry, 15, 1452–1461.
Dworkin, J. P., & Miller, S. L. (2000). A kinetic estimate of the free aldehyde content of aldoses. Carbohydrate Research, 329, 359–363.
Fersht, A. (1999). Structure and mechanism in protein science: Guide to enzyme catalysis and protein folding. New York: Freeman WH and Company.
Fields, P. A., Dong, Y., Meng, X., & Somero, G. N. (2015). Adaptations of protein structure and function to temperature: There is more than one way to ‘skin a cat’. Journal of Experimental Biology, 218, 1801–1811.
Firn, R. D., & Jones, C. G. A. (2009). Darwinian view of metabolism: Molecular properties determine fitness. Journal of Experimental Botany, 60, 719–726.
Frey, P. A. (1996). The Leloir pathway: A mechanistic imperative for three enzymes to change the stereochemical configuration of a single carbon in galactose. FASEB Journal, 10, 461–470.
Goldberg, R. N., & Tewari, Y. (1989). Thermodynamic and transport properties of carbohydrates and their monophosphates: The pentoses and hexoses. Journal of Physical and Chemical Reference Data, 18, 809–880.
Hall, D. R., Bond, C. S., Leonard, G. A., Watt, C. I., Berry, A., & Hunter, W. N. (2002). Structure of tagatose-1,6-bisphosphate aldolase insight into chiral discrimination, mechanism, and specificity of class II aldolases. Journal of Biological Chemistry, 277, 22018–22024.
Havel, P. J. (2005). Dietary fructose: Implications for dysregulation of energy homeostasis and lipid/carbohydrate metabolism. Nutrition Reviews, 63, 133–157.
Hawley, J. A., Maughan, R. J., & Hargreaves, M. (2015). Exercise metabolism: Historical perspective. Cell Metabolism, 22, 12–17.
Holmes, E. (2015). The metabolism of living tissues. Cambridge: Cambridge University Press.
Ichikawa, M., Scott, D. A., Losfeld, M. E., & Freeze, H. H. (2014). The metabolic origins of mannose in glycoproteins. Journal of Biological Chemistry, 289, 6751–6761.
Mulichak, A. M., Wilson, J. E., Padmanabhan, K., & Garavito, R. M. (1998). The structure of mammalian hexokinase-1. Nature Structural Biology, 5, 555–560.
Noor, E., Bar-Even, A., Flamholz, A., Reznik, E., Liebermeister, W., & Milo, R. (2014). Pathway thermodynamics highlights kinetic obstacles in central metabolism. PLOS Computational Biology, 10(2), e1003483. https://doi.org/10.1371/journal.pcbi.1003483.
Price, N. C., & Stevens, L. (1999). Fundamentals of enzymology: The cell and molecular biology of catalytic proteins. New York: Oxford University Press.
Sajitz-Hermstein, M., & Nikoloski, Z. (2013). Structural control of metabolic flux. PLOS Computational Biology, 9(12), e1003368. https://doi.org/10.1371/journal.pcbi.1003368.
Sharma, V., Ichikawa, M., Hudson, H., & Freeze, H. H. (2014). Mannose metabolism: More than meets the eye. Biochemical and Biophysical Research Communications, 453, 220–228.
Slein, M. W., Cori, G. T., & Cori, C. F. (1950). A comparative study of hexokinase from yeast and animal tissues. Journal of Biological Chemistry, 186, 763–780.
Steinmann, B., Santer, R. (2012). Disorders of fructose metabolism. In J. M. Saudubray, G. van der Berghe, J. H. Walter (Eds.), Inborn metabolic diseases: Diagnosis and treatment (5th edn.) (pp. 157–165). New York: Springer.
Stincone, A., Prigione, A., Cramer, T., Wamelink, M. M. C., Campbell, K., Cheung, E., et al. (2015). The return of metabolism: Biochemistry and physiology of the pentose phosphate pathway. Biological Reviews, 90, 927–963.
Zgiby, S. M., Thomson, G. J., Qamar, S., & Berry, A. (2000). Exploring substrate binding and discrimination in fructose 1, 6-bisphosphate and tagatose 1,6-bisphosphate aldolases. European Journal of Biochemistry, 267, 1858–1868.
Acknowledgements
This paper is dedicated to our biochemistry professor Pier Luigi Ipata for his continued passion for biochemical research. Thanks are also due to our colleagues at Pisa University, Prof. Mario Pellegrino and Prof. Aldo Paolicchi for their helpful discussions.
Funding
This work was supported by the University of Pisa.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
All authors declare that they have no conflict of interest.
Human and animal rights
This article does not contain any studies with human participants or animals performed by any of the authors.
Rights and permissions
About this article
Cite this article
Del-Corso, A., Cappiello, M., Moschini, R. et al. How the chemical features of molecules may have addressed the settlement of metabolic steps. Metabolomics 14, 2 (2018). https://doi.org/10.1007/s11306-017-1300-1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11306-017-1300-1