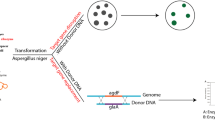
Abstract
The endophytic fungus Berkleasmium sp. Dzf12 that was isolated from Dioscorea zingiberensis, is a proficient producer of palmarumycins, which are intriguing polyketides of the spirobisnaphthalene class. These compounds displayed a wide range of bioactivities, including antibacterial, antifungal, and cytotoxic activities. However, conventional genetic manipulation of Berkleasmium sp. Dzf12 is difficult and inefficient, partially due to the slow-growing, non-sporulating, and highly pigmented behavior of this fungus. Herein, we developed a CRISPR/Cas9 system suitable for gene editing in Berkleasmium sp. Dzf12. The protoplast preparation was optimized, and the expression of Cas9 in Berkleasmium sp. Dzf12 was validated. To assess the gene disruption efficiency, a putative 1, 3, 6, 8-tetrahydroxynaphthalene synthase encoding gene, bdpks, involved in 1,8-dihydroxynaphthalene (DHN)-melanin biosynthesis, was selected as the target for gene disruption. Various endogenous sgRNA promoters were tested, and different strategies to express sgRNA were compared, resulting in the construction of an optimal system using the U6 snRNA-1 promoter as the sgRNA promoter. Successful disruption of bdpks led to a complete abolishment of the production of spirobisnaphthalenes and melanin. This work establishes a useful gene targeting disruption system for exploration of gene functions in Berkleasmium sp. Dzf12, and also provides an example for developing an efficient CRISPR/Cas9 system to the fungi that are difficult to manipulate using conventional genetic tools.









Similar content being viewed by others
Data availability
Data is provided within the manuscript or supplementary information files.
References
Akamatsu HO, Chilvers MI, Stewart JE et al (2010) Identification and function of a polyketide synthase gene responsible for 1,8-dihydroxynaphthalene-melanin pigment biosynthesis in Ascochyta rabiei. Curr Genet 56:349–360
Arazoe T, Miyoshi K, Yamato T et al (2015) Tailor-made CRISPR/Cas system for highly efficient targeted gene replacement in the rice blast fungus. Biotechnol Bioeng 112:2543–2549
Bode HB, Wegner B, Zeeck A (2000) Biosynthesis of cladospirone bisepoxide, a member of the spirobisnaphthalene family. J Antibiot 53:153–157
Cai X, Shan T, Li P et al (2009) Spirobisnaphthalenes from the endophytic fungus Dzf12 of Dioscorea zingiberensis and their antimicrobial activities. Nat Prod Commun 4:1469–1472
Cai Y-S, Guo Y-W, Krohn K (2010) Structure, bioactivities, biosynthetic relationships and chemical synthesis of the spirodioxynaphthalenes. Nat Prod Rep 27:1840–1870
Chen GZ, Chu J (2019) Characterization of two polyketide synthases involved in sorbicillinoid biosynthesis by Acremonium chrysogenum using the CRISPR/Cas9 system. Appl Biochem Biotechnol 188:1134–1144
Cong L, Ran FA, Cox D et al (2013) Multiplex genome engineering using CRISPR/Cas systems. Science 339:819–823
Doudna JA, Charpentier E (2014) Genome editing. The new frontier of genome engineering with CRISPR-Cas9. Science 346:1258096
Fujii I, Yasuoka Y, Tsai H-F et al (2004) Hydrolytic polyketide shortening by Ayg1p, a novel enzyme involved in fungal melanin biosynthesis. J Biol Chem 279:44613–44620
Gakuubi MM, Ching KC, Munusamy M et al (2022) CRISPR/Cas9 RNP-assisted validation of palmarumycin biosynthetic gene cluster in Lophiotrema sp. F6932. Front Microbiol 13:1012115
Gardiner DM, Kazan K (2018) Selection is required for efficient Cas9-mediated genome editing in Fusarium graminearum. Fungal Biol 122:131–137
Huang P-W, Yang Q, Zhu Y-L et al (2020) The construction of CRISPR-Cas9 system for endophytic Phomopsis liquidambaris and its PmkkA-deficient mutant revealing the effect on rice. Fungal Genet Biol 136:103301
Jiang W, Bikard D, Cox D et al (2013) RNA-guided editing of bacterial genomes using CRISPR-Cas systems. Nat Biotechnol 31:233–239
Katayama T, Tanaka Y, Okabe T et al (2016) Development of a genome editing technique using the CRISPR/Cas9 system in the industrial filamentous fungus Aspergillus oryzae. Biotechnol Lett 38:637–642
Krohn K, Michel A, Floerke U et al (1994) Palmarumycins C1-C16 from Coniothyrium sp.: isolation, structure elucidation, and biological activity. Liebigs Ann Chem : 1099–1108
Li Y, Li P, Mou Y et al (2012) Enhancement of diepoxin ζ production in liquid culture of endophytic fungus Berkleasmium sp. Dzf12 by polysaccharides from its host plant Dioscorea zingiberensis. World J Microbiol Biotechnol 28:1407–1413
Liang Y, Han Y, Wang C et al (2018) Targeted deletion of the USTA and UvSLT2 genes efficiently in Ustilaginoidea virens with the CRISPR-Cas9 system. Front Plant Sci 9:699
Liu R, Chen L, Jiang YP et al (2015) Efficient genome editing in filamentous fungus Trichoderma reesei using the CRISPR/Cas9 system. Cell Discov 1:15007
Liu X, Zhao Y, Wang W et al (2017) Recent progress of natural product spirobisnaphthalenes. Chin J Org Chem 37:2883–2894
Luo N, Li ZY, Ling J et al (2023) Establishment of a CRISPR/Cas9-mediated efficient knockout system of Trichoderma hamatum T21 and pigment synthesis PKS gene knockout. J Fungi 9:595
Matsu-Ura T, Baek M, Kwon J et al (2015) Efficient gene editing in Neurospora crassa with CRISPR technology. Fungal Biol Biotechnol 2:4–4
Mou Y, Meng J, Fu X et al (2013) Antimicrobial and antioxidant activities and effect of 1-hexadecene addition on palmarumycin C2 and C3 yields in liquid culture of endophytic fungus Berkleasmium sp. Dzf12 Molecules 18:15587–15599
Ran FA, Hsu PD, Wright J et al (2013) Genome engineering using the CRISPR-Cas9 system. Nat Protoc 8:2281–2308
Schuster M, Schweizer G, Reissmann S et al (2016) Genome editing in Ustilago maydis using the CRISPR-Cas system. Fungal Genet Biol 89:3–9
Shan Q, Wang Y, Li J et al (2013) Targeted genome modification of crop plants using a CRISPR-Cas system. Nat Biotechnol 31:686–688
Shan T, Tian J, Wang X et al (2014) Bioactive spirobisnaphthalenes from the endophytic fungus Berkleasmium sp. J Nat Prod 77:2151–2160
Shi TQ, Gao J, Wang WJ et al (2019) CRISPR/Cas9-based genome editing in the filamentous fungus Fusarium fujikuroi and its application in strain engineering for gibberellic acid production. ACS Synth Biol 8:445–454
Song L, Ouedraogo JP, Kolbusz M et al (2018) Efficient genome editing using tRNA promoter-driven CRISPR/Cas9 gRNA in Aspergillus niger. PLoS ONE 13:e0202868
Thompson JE, Fahnestock S, Farrall L et al (2000) The second naphthol reductase of fungal melanin biosynthesis in Magnaporthe grisea: tetrahydroxynaphthalene reductase. J Biol Chem 275:34867–34872
Tian J, Liu XC, Liu ZL et al (2016) Larvicidal spirobisnaphthalenes from the endophytic fungus Berkleasmium sp. against Aedes albopictus. Pest Manage Sci 72:961–965
Tong Y, Weber T, Lee SY (2019) CRISPR/Cas-based genome engineering in natural product discovery. Nat Prod Rep 36:1262–1280
Vagstad Anna L, Hill Eric A, Labonte Jason W et al (2012) Characterization of a fungal thioesterase having claisen cyclase and deacetylase activities in melanin biosynthesis. Chem Biol 19:1525–1534
Wang Q, Cobine PA, Coleman JJ (2018) Efficient genome editing in Fusarium oxysporum based on CRISPR/Cas9 ribonucleoprotein complexes. Fungal Genet Biol 117:21–29
Weber J, Valiante V, Nodvig CS et al (2017) Functional reconstitution of a fungal natural product gene cluster by advanced genome editing. ACS Synth Biol 6:62–68
Weyda I, Yang L, Vang J et al (2017) A comparison of Agrobacterium-mediated transformation and protoplast-mediated transformation with CRISPR-Cas9 and bipartite gene targeting substrates, as effective gene targeting tools for Aspergillus carbonarius. J Microbiol Methods 135:26–34
Wheeler MH, Abramczyk D, Puckhaber LS et al (2008) New biosynthetic step in the melanin pathway of Wangiella (Exophiala) dermatitidis: evidence for 2-acetyl-1,3,6,8-tetrahydroxynaphthalene as a novel precursor. Eukaryot Cell 7:1699–1711
Xie SS, Shen B, Zhang CB et al (2014) sgRNAcas9: a software package for designing CRISPR sgRNA and evaluating potential off-target cleavage sites. PLoS ONE 9:e100448
Xu L, Zhou L, Zhao J et al (2008) Fungal endophytes from Dioscorea zingiberensis rhizomes and their antibacterial activity. Lett Appl Microbiol 46:68–72
Xu XR, Huang RY, Yin WB (2021) An optimized and efficient CRISPR/Cas9 system for the endophytic fungus Pestalotiopsis fici. J Fungi 7:809
Yang J, Zhao X, Sun J et al (2010) A novel protein Com1 is required for normal conidium morphology and full virulence in Magnaporthe oryzae. Mol Plant-Microbe Interact 23:112–123
Yu JH, Hamari Z, Han KH et al (2004) Double-joint PCR: a PCR-based molecular tool for gene manipulations in filamentous fungi. Fungal Genet Biol 41:973–981
Zhang C, Meng X, Wei X et al (2016) Highly efficient CRISPR mutagenesis by microhomology-mediated end joining in Aspergillus fumigatus. Fungal Genet Biol 86:47–57
Zhao J, Li Y, Shan T et al (2011) Enhancement of diepoxin ζ production with in situ resin adsorption in mycelial liquid culture of the endophytic fungus Berkleasmium sp. Dzf12 from Dioscorea zingiberensis. World J Microbiol Biotechnol 27:2753–2758
Zheng YM, Lin FL, Gao H et al (2017) Development of a versatile and conventional technique for gene disruption in filamentous fungi based on CRISPR-Cas9 technology. Sci Rep 7
Zheng X, Zheng P, Zhang K et al (2019) 5S rRNA promoter for guide RNA expression enabled highly efficient CRISPR/Cas9 genome editing in Aspergillus niger. ACS Synth Biol 8:1568–1574
Zhong L, Zhou Y, Gao S et al (2011) Endophytic fungi from the hybrid ‘Neva’ of Populus deltoides Marsh × Populus nigra L. and their antimicrobial activity. Afr J Microbiol Res 5:3924–3929
Zhou L, Zhao J, Shan T et al (2010) Spirobisnaphthalenes from fungi and their biological activities. Mini-Rev Med Chem 10:977–989
Funding
This work was supported by the National Natural Science Foundation of China (No. 31872623, 31071710), and National Key Research and Development Program of China (2023YFD1401400).
Author information
Authors and Affiliations
Contributions
Conceptualization, L.Z. and D.L.; methodology, S.Z., D.X., D.L.; software, S.Z., D.X.; formal analysis, S.Z., Z.S., D.L.; investigation, S.Z., R.Y., M.Z., Z.Z., Z.S and Y.M.; data curation, S.Z.; writing, S.Z., D.L.; supervision, L.Z. and D.L. All authors have read and agreed to the published version of the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Zhao, S., Yin, R., Zhang, M. et al. Efficient gene editing in the slow-growing, non-sporulating, melanized, endophytic fungus Berkleasmium sp. Dzf12 using a CRISPR/Cas9 system. World J Microbiol Biotechnol 40, 176 (2024). https://doi.org/10.1007/s11274-024-03988-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11274-024-03988-y