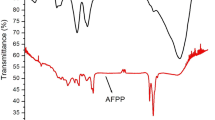
Abstract
Since ancient time, plants and there parts have been used widely against dreadful pathogens due to ability of killing microbes. Waste from fruits and vegetables are pulling in more interest in exploration due to their therapeutic properties such as anti-pathogenic activity. In the present study antimicrobial and cytotoxicity properties of herbal combination prepared from peels of Allium cepa, Cucumis sativus, Citrus reticulata, and Mangifera indica were investigated. The herbal combination was tested for broad spectrum antimicrobial activity against Streptococcus mutans, Bacillus licheniformis, Lactobacillus plantarum, Escherichia coli, Pseudomonas aeruginosa, and Citrobacter freundii. The presence of phytochemical markers such as phenolics and flavanoids were also investigated. The results revealed that the herbal combination exhibited antimicrobial activity against S. mutans, B. licheniformis, L. plantarum, E. coli and moderate against P. aeruginosa and C. freundii. The presence of phenolics (798 ± 1.52 µg/g) and flavanoids (355.3 ± 2.081 µg/g) was also detected. Also, the herbal combination contains flavanoids such as quercetin and rutin was confirmed with the help of column chromatography, high-performance liquid chromatography (HPLC), fourier-transform infrared spectroscopy (FTIR) and nuclear magnetic resonance spectroscopy (NMR) analysis. The herbal combination helps in the proliferation of L929 cells without affecting their viability was confirmed by MTT assay. The results of the present research suggests the possibility to use herbal combination as source for plausible antibacterial agent which could be isolated and used as a lead candidate for the development of antibacterial drug that help to limit or stop infectious illnesses caused by different pathogenic microbes.





Similar content being viewed by others
Data availability
The datasets generated during the current study is included in this article.
References
Abraham EP, Chain E, Fletcher CM, Gardner AD, Heatley NG, Jennings MA, Florey HW (1941) Further observations on penicillin. Lancet 238:177–189. https://doi.org/10.1016/S0140-6736(00)72122-2
Blunder M, Orthaber A, Bauer R, Kunert O (2017) Efficient identification of flavones, flavanones and their glycosides in routine analysis via off-line combination of sensitive NMR and HPLC experiments. Food Chem 218:600–609. https://doi.org/10.1016/j.foodchem.2022.132531
Braz VS, Melchior K, Moreira CG (2020) Escherichia coli as a multifaceted pathogenic and versatile bacterium. Front Cell Infect Microbiol 10:1–9. https://doi.org/10.3389/fcimb.2020.548492
Chhetry AK, Dhakal S, Chaudhary L, Karki K, Khadka RB, Chaudhary GP, Bastola T, Poudel A, Aryal P, Pandey J (2022) Study of antibacterial activity of root bark, leaves, and pericarp extracts of Diploknema butyracea and evaluation of prospective antioxidant activity. J Trop Med 2022:1–12. https://doi.org/10.1155/2022/6814901
Chourasiya A, Upadhayay A, Shukla RN (2012) Isolation of quercetin from the leaves of Azadirachta indica and antidiabetic study of crude extracts. J Pharm Biomed Sci 25:179–181
CLSI, (2016). Performance standards for Antimicrobial Susceptibility Testing, 18th Informational Supplement. M100-S18. Wayne, PA: Clinical and Laboratory Standards Institute.
Coman V, Teleky BE, Mitrea L, Martau GA, Szabo K, Calinoiu LF, Vodnar DC (2020) Bioactive potential of fruit and vegetable wastes. Adv Food Nutr Res 91:157–225
Dahmani S, Chabir R, Errachidi F, Berrada W, Lansari H, Benidir M, Ghadraoui LE, Bour A (2020) Evaluation of in vivo wound healing activity of moroccan Citrus reticulata peel extract. Clin Phytoscience 6:1–9. https://doi.org/10.1186/s40816-020-00222-8
Dai J, Mumper RJ (2010) Plant phenolics: extraction, analysis and their antioxidant and anticancer properties. Molecules 15:7313–7352
Das DC, Sinha NK, Chattopadhyay JC, Das M (2013) Antibacterial activity and photochemical analysis of cardanthera difformis druce leaf extracts from West Bengal, India. Int J Phytomedicine 5:446–451
Devika R, Koilpillai J (2015) Column chromatography separation of bioactive compounds from Tagetes Erecta Linn. Int J Pharm Sci Res 6:762–766. https://doi.org/10.13040/IJPSR.0975-8232.6(2).762-66
Gini TG, Jothi GJ (2018) Column chromatography and HPLC analysis of phenolic compounds in the fractions of Salvinia molesta mitchell. Egypt J Basic Appl Sci 5:197–203
Greenwood D (1989) Antibiotic Sensitivity Testing. Antimicrobial Chemotherapy. Oxford University Press, New York, pp 91–100
Harty DW, Oakey HJ, Patrikakis M, Hume EB, Knox KW (1994) Pathogenic potential of lactobacilli. Int J Food Microbiol 24:179–189. https://doi.org/10.1016/0168-1605(94)90117-1
Haydushka IA, Markova N, Kirina V, Atanassov M (2012) Recurrent sepsis due to Bacillus Licheniformis. J Glob Infect Dis 4:82–83. https://doi.org/10.4103/0974-777X.93768
Huang CY, Kuo CH, Wu CH, Kuan AW, Guo HR, Lin YH, Wang PK (2018) Free radical-scavenging, anti-inflammatory, and antibacterial activities of water and ethanol extracts prepared from compressional-puffing pretreated mango (Mangifera indica L.) peels. J Food Qual. https://doi.org/10.1155/2018/1025387
Jiao M, Wu ZL, Liu Y, Liu W, Li R (2016) Surfactant-assisted separation of ginkgo flavanoids from Ginkgo biloba leaves using leaching and foam fractionation. Asia-Pac J Chem Eng 11:664–672
John S, Priyadarshini S, Monica SJ, Sivaraj C, Arumugam P (2018) In Vitro antioxidant and antimicrobial properties of Cucumis Sativus L. peel extracts. Int Res J Pharm 9:56–60. https://doi.org/10.7897/2230-8407.0918
Kumar H, Bhardwaj K, Sharma R, Nepovimova E, Kuca K, Dhanjal DS, Verma R, Bhardwaj P, Sharma S, Kumar D (2020) Fruit and vegetable peels: utilization of high value horticulture waste in novel industrial applications. Molecules 25:1–21. https://doi.org/10.3390/molecules25122812
Kumar K, Srivastav S, Sharanagat VS (2021) Ultrasound assisted extraction (UAE) of bioactive compounds from fruit and vegetable processing by-products: a review. Ultrason Sonochem 70:1350–4177. https://doi.org/10.1016/j.ultsonch.2020.105325
Lim YY, Lim TT, Tee JJ (2007) Antioxidant properties of several tropical fruits: a comparative study. Food Chem 103:1003–1008. https://doi.org/10.1016/j.foodchem.2006.08.038
Liu LH, Wang NY, Wu AYJ, Lin CC, Lee CM, Liu CP (2018) Citrobacter freundii bacteremia: risk factors of mortality and prevalence of resistance genes. J Microbiol Immunol Infect 51:565–572. https://doi.org/10.1016/j.jmii.2016.08.016
Moradali MF, Ghods S, Rehm BHA (2017) Pseudomonas aeruginosa lifestyle: a paradigm for adaptation, survival, and persistence. Front Cell Infect Microbiol 7:1–29. https://doi.org/10.3389/fcimb.2017.00039
Nakano K, Nomura R, Ooshima T (2008) Streptococcus mutans and cardiovascular diseases. Jap Dent Sci Rev 44:29–37. https://doi.org/10.1016/j.jdsr.2007.09.001
Obi FO, Ugwuishiwu BO, Nwakaire JN (2016) Agricultural waste concept, generation, utilization and management. Niger J Technol 35:957–964
Petchi RR, Vijaya C, Parasuraman S (2013) Anti-arthritic activity of ethanolic extract of Tridax procumbens (Linn.) in sprague dawley rats. Pharmacogn Res 5:113–117
Phukan K, Devi R, Chowdhury D (2021) Green synthesis of gold nano-bioconjugates from onion peel extract and evaluation of their antioxidant, anti-inflammatory, and cytotoxic studies. ACS Omega 6:17811–17823. https://doi.org/10.1021/acsomega.1c00861
Safdar MN, Kausar T, Jabbar S, Mumtaz A, Ahad K, Saddozai AA (2017) Exreaction and quatification of polyphenols from kinnow (Citrus Reticulate L.) peels using ultrasound and maceration. J Food Drug Anal 25:488–500. https://doi.org/10.1016/j.jfda.2016.07.010
Sagar NA, Pareek S, Gonzalez AG (2020) Quantification of flavanoids, total phenols and antioxidant properties of onion skin: a comparative study of fifteen Indian cultivares. J Food Sci Technol 57:2423–2432. https://doi.org/10.1007/s13197-020-04277-w
Saini A, Panesar PS, Bera MB (2019) Comparative study on the extraction and quantification of Polyphenols from citrus peels using maceration and ultrasonic technique. Curr Res in Nutr Food Sci 7:678–685. https://doi.org/10.12944/CRNFSJ.7.3.08
Selvaraj K, Chowdhury R, Bhattacharjee C (2013) Isolation and structural elucidation of flavanoid from aquatic fern azolla microphylla and evaluation of free radical scavenging activity. Int J Pharm Sci 5:743–749
Singh K, Kumar T, Prince VK, Sharma S, Rani J (2019) A review on conversion of food wastes and by-products into value added products. Int J Chem Stud 7:2068–2073
Singleton VL, Rossi JA (1965) Colorimetry of total phenolics with phosphomolybdic-phosphotungstic acid reagents. Am J Enol Vitic 16:144–158
Someya S, Yoshiki Y, Okubo K (2002) Antioxidant compounds from bananas (Musa cavendish). Food Chem 79:351–354. https://doi.org/10.1016/S0308-8146(02)00186-3
Stefanescu BE, Calinoiu LF, Ranga F, Fetea F, Mocan A, Vodnar DC, Crisan G (2020) Chemical composition and biological activities of the nord-west Romanian wild bilberry (Vaccinium myrtillus L.) and Lingonberry (Vaccinium vitis-idaea L.) leaves. Antioxidants 9:1–21. https://doi.org/10.3390/antiox9060495
Szabo K, Dulf FV, Diaconeasa Z, Vodnar DC (2019) Antimicrobial and antioxidant properties of tomato processing byproducts and their correlation with the biochemical composition. LWT 116:108558
Thiyagarajan D, Sambandam B, Ayyaswamy A, Raman P (2016) Extraction and isolation of flavanoids from the leaves of Trigonella Foenum-Graecum and their antioxidant activity. Int J Pharm and Pharm Sci 8:120–212
Usman JG, Sodipo OA, Sandabe UK (2014) In vitro antimicrobial activity of Cucumis metuliferus E. Mey. Ex. Naudin fruit extracts against Salmonella gallinarum. Int J Phytomedcine 6:268–274
Vajrabhaya L, Korsuwannawong S (2018) Cytotoxicity evaluation of a Thai herb using tetrazolium (MTT) and sulforhodamine B (SRB) assays. J Anal Sci Technol 9:1–6. https://doi.org/10.1186/s40543-018-0146-0
Zahoor M, Shafiq S, Ullah H, Sadiq A, Ullah F (2018) Isolation of quercetin and mandelic acid from Aesculue indica fruit and their biological activities. BMC Biochem 19:1–14
Zhishen J, Mengcheng T, Jianming W (1999) The determination of flavanoid content in mulberry and their scavenging effect on superoxide radicals. Food Chem 64:555–559. https://doi.org/10.1016/S0308-8146(98)00102-2
Acknowledgements
The authors are thankful to Prof. Ina Aditya Shastri Vice-Chancellor, Banasthali Vidyapith and Prof. Dipjyoti Chakraborty, Head of the Department, Bioscience and Biotechnology, Banasthali Vidyapith for their encouragement and providing scientific platform to carry out present work. We are also thankful to DBT supported Bioinformatics Center, for providing computational assistance and Dr Swati Paliwal, Associate professor, Department of Bioscience and Biotechnology, Banasthali Vidyapith, for providing guidance during cell culture work.
Funding
The authors declare that no funds, grants, or other support were received during the preparation of this manuscript.
Author information
Authors and Affiliations
Contributions
JM and NS conceived the present idea of writing the research and designed the content. JM and NS encouraged to investigate and supervised the findings of this work. AS developed the theory and performed the experiments. AS contributed to the analysis of the results and writing of the manuscript.
Corresponding authors
Ethics declarations
Conflict of interest
The authors confirm that they have no conflicts of interest with respect to the work described in this manuscript.
Ethical approval
Ethical approval is not applicable. This article does not contain any studies with human participants or animals performed by any of the authors.
Consent to publish
The authors have consent for publication.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Anjani, Mathur, J. & Srivastava, N. Value added bioactive compounds from fruits & vegetables waste for assessing their antimicrobial activity. World J Microbiol Biotechnol 39, 71 (2023). https://doi.org/10.1007/s11274-022-03488-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11274-022-03488-x