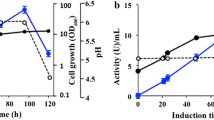
Abstract
N-acetyl-d-glucosamine (GlcNAc) is an important amino-monosaccharide with great potential for biotechnological applications. It has traditionally been produced by the chemical hydrolysis of chitin, despite certain industrial and environmental drawbacks, including acidic wastes, low yields and high costs. Therefore, enzymatic production has gained attention as a promising environmentally-friendly alternative to the chemical processes. In this study we demonstrate the GlcNAc bioproduction from colloidal α-chitin using an enzyme cocktail containing endochitinases and exochitinases (chitobiosidases and N-acetyl-glucosaminidases). The enzyme cocktail was extracted after fermentation in a bioreactor by Aeromonas caviae CHZ306, a chitinolytic marine bacterium with great potential for chitinase production. Hydrolysis parameters were studied in terms of temperature, pH, enzyme and substrate concentration, and reaction time, achieving over 90% GlcNAc yield within 6 h. The use of colloidal α-chitin as substrate showed a substantial improvement of GlcNAc yields, when compared with β-chitin and α-chitin polymorphs. Such result is directly related to a significant decrease in crystallinity and viscosity from natural α-chitin, providing the chitinase with greater accessibility to the depolymerized chains. This study provides valuable information on the GlcNAc bioproduction from chitin using an enzymatic approach, addressing the key points for its production, including the enzyme cocktail composition and the substrate structures.
Graphic abstract





Similar content being viewed by others
References
Binod P, Pusztahelyi T, Nagy V et al (2005) Production and purification of extracellular chitinases from Penicillium aculeatum NRRL 2129 under solid-state fermentation. Enzyme Microb Technol 36:880–887. https://doi.org/10.1016/j.enzmictec.2004.12.031
Bissett DL (2006) Glucosamine: an ingredient with skin and other benefits. J Cosmet Dermatol 5:309–315. https://doi.org/10.1111/j.1473-2165.2006.00277.x
Bissett DL, Farmer T, McPhail S et al (2007) Genomic expression changes induced by topical N-acetyl glucosamine in skin equivalent cultures in vitro. J Cosmet Dermatol 6:232–238. https://doi.org/10.1111/j.1473-2165.2007.00339.x
Cardozo FA, Gonzalez JM, Feitosa VA et al (2017) Bioconversion of α-chitin into N-acetyl-glucosamine using chitinases produced by marine-derived Aeromonas caviae isolates. World J Microbiol Biotechnol 33:201. https://doi.org/10.1007/s11274-017-2373-8
Chen RH, Hsu CN, Chung MY et al (2008) Effect of different concentrations of collagen, ceramides, N-acetyl glucosamine, or their mixture on enhancing the proliferation of keratinocytes, fibroblasts and the secretion of colagen and/or the expression of mRNA of type I collagen. J Food Drug Anal 16:66–74
Chen J-K, Shen C-R, Liu C-L (2010) N-acetylglucosamine: production and applications. Mar Drugs 8:2493–2516. https://doi.org/10.3390/md8092493
Chernin LS, Winson MK, Thompson JM et al (1998) Chitinolytic activity in Chromobacterium violaceum: substrate analysis and regulation by quorum sensing. J Bacteriol 180:4435–4441
Dalirfardouei R, Karimi G, Jamialahmadi K (2016) Molecular mechanisms and biomedical applications of glucosamine as a potential multifunctional therapeutic agent. Life Sci 152:21–29. https://doi.org/10.1016/j.lfs.2016.03.028
Facchinatto WM, Fiamingo A, dos Santos DM, Campana-Filho SP (2019) Characterization and physical-chemistry of methoxypoly(ethylene glycol)-g-chitosan. Int J Biol Macromol 124:828–837. https://doi.org/10.1016/J.IJBIOMAC.2018.11.246
Fiamingo A, de Delezuk JAM, Trombotto S et al (2016) Extensively deacetylated high molecular weight chitosan from the multistep ultrasound-assisted deacetylation of beta-chitin. Ultrason Sonochem 32:79–85. https://doi.org/10.1016/j.ultsonch.2016.02.021
Frändberg E, Schnürer J (1994) Evaluation of a chromogenic chito-oligosaccharide analogue, p-nitrophenyl-β-D-N, N’’%3c/i%3e-diacetylchitobiose, for the measurement of the chitinolytic activity of bacteria. J Appl Bacteriol 76:259–263. https://doi.org/10.1111/j.1365-2672.1994.tb01625.x
Gardner KH, Blackwell J (1974) The structure of native cellulose. Biopolymers 13:1975–2001. https://doi.org/10.1002/bip.1974.360131005
Halder SK, Jana A, Paul T et al (2016) Purification and biochemical characterization of chitinase of Aeromonas hydrophila SBK1 biosynthesized using crustacean shell. Biocatal Agric Biotechnol 5:211–218. https://doi.org/10.1016/j.bcab.2015.11.003
Heux L, Brugnerotto J, Desbrières J et al (2000) Solid State NMR for determination of degree of acetylation of chitin and chitosan. Biomacromol 1:746–751. https://doi.org/10.1021/bm000070y
Hiraga K, Shou L, Kitazawa M et al (1997) Isolation and characterization of chitinase from a flake-chitin degrading marine bacterium, Aeromonas hydrophila H-2330. Biosci Biotechnol Biochem 61:174–176. https://doi.org/10.1271/bbb.61.174
Hirano S, Nagao N (1988) An improved method for the preparation of colloidal chitin by using methanesulfonic acid. Agric Biol Chem 52:2111–2112. https://doi.org/10.1080/00021369.1988.10868977
Kim TI, Lim DH, Baek KS et al (2018) Production of chitinase from Escherichia fergusonii, chitosanase from Chryseobacterium indologenes, Comamonas koreensis and its application in N-acetylglucosamine production. Int J Biol Macromol 112:1115–1121. https://doi.org/10.1016/J.IJBIOMAC.2018.02.056
Jamialahmadi K, Behravan J, Najafi MF et al (2011) Enzymatic production of N-acetyl-d-glucosamine from chitin using crude enzyme preparation of aeromonas sp. PTCC1691. Biotechnology 10:292–297
Jeuniaux C, Voss-Foucart MF (1991) Chitin biomass and production in the marine environment. Biochem Syst Ecol 19:347–356. https://doi.org/10.1016/0305-1978(91)90051-Z
Johnson RL, Schmidt-Rohr K (2014) Quantitative solid-state 13C NMR with signal enhancement by multiple cross polarization. J Magn Reson 239:44–49. https://doi.org/10.1016/j.jmr.2013.11.009
Jung W-J, Park R-D (2014) Bioproduction of chitooligosaccharides: present and perspectives. Mar Drugs 12:5328–5356. https://doi.org/10.3390/md12115328
Jung W-J, Souleimanov A, Park R-D, Smith DL (2007) Enzymatic production of N-acetyl chitooligosaccharides by crude enzyme derived from Paenibacillus illioisensis KJA-424. Carbohydr Polym 67:256–259. https://doi.org/10.1016/j.carbpol.2006.05.005
Karube I, Morita T (1993) Enzymatic decomposition method of chitin-containing materials. US Patent no. 5262310
Kaya M, Mujtaba M, Ehrlich H et al (2017) On chemistry of γ-chitin. Carbohydr Polym 176:177–186. https://doi.org/10.1016/j.carbpol.2017.08.076
Klaikherd A, Siripastr Jayanta ML, Boonjawat J et al (2004) Depolymerization of β-chitin to mono- and disaccharides by the serum fraction from the para rubber tree, Hevea brasiliensis. Carbohydr Res 339:2799–2804. https://doi.org/10.1016/j.carres.2004.09.011
Kuk JH, Jung WJ, Jo GH et al (2005) Production of N-acetyl-β-d-glucosamine from chitin by Aeromonas sp. GJ-18 crude enzyme. Appl Microbiol Biotechnol 68:384–389. https://doi.org/10.1007/s00253-004-1877-y
Kuk JH, Jung WJ, Jo GH et al (2006) Production of N,N′-diacetylchitobiose from chitin using temperature-sensitive chitinolytic enzyme preparations of Aeromonas sp. GJ-18. World J Microbiol Biotechnol 22:135–139. https://doi.org/10.1007/s11274-005-9009-0
Kurita K, Kaji Y, Mori T, Nishiyama Y (2000) Enzymatic degradation of β-chitin: susceptibility and the influence of deacetylation. Carbohydr Polym 42:19–21. https://doi.org/10.1016/S0144-8617(99)00127-7
Lan X, Ozaqua N, Nishiwaki N et al (2004) Purification, cloning, and sequence analysis of β-N-acetylglucosaminidase from the chitinolytic bacterium Aeromonas hydrophila strain SUWA-9. Biosci Biotechnol Biochem 68:1082–1090. https://doi.org/10.1271/bbb.68.1082
Lavall RL, Assis OBG, Campana-Filho SP (2007) Beta-chitin from the pens of Loligo sp.: extraction and characterization. Bioresour Technol 98:2465–2472
Liu L, Liu Y, Shin H et al (2013) Microbial production of glucosamine and N-acetylglucosamine: advances and perspectives. Appl Microbiol Biotechnol 97:6149–6158. https://doi.org/10.1007/s00253-013-4995-6
Minke R, Blackwell J (1978) The structure of α-chitin. J Mol Biol 120:167–181. https://doi.org/10.1016/0022-2836(78)90063-3
Nawani NN, Kapadnis BP (2001) One-step purification of chitinase from Serratia marcescens NK1, a soil isolate. J Appl Microbiol 90:803–808. https://doi.org/10.1046/j.1365-2672.2001.01308.x
Nguyen-Thi N, Doucet N (2016) Combining chitinase C and N-acetylhexosaminidase from Streptomyces coelicolor A3(2) provides an efficient way to synthesize N-acetylglucosamine from crystalline chitin. J Biotechnol 220:25–32. https://doi.org/10.1016/j.jbiotec.2015.12.038
Osorio-Madrazo A, David L, Trombotto S et al (2010) Kinetics study of the solid-state acid hydrolysis of chitosan: evolution of the crystallinity and macromolecular structure. Biomacromol 11:1376–1386. https://doi.org/10.1021/bm1001685
Ottey MH, Vårum KM, Smidsrød O (1996) Compositional heterogeneity of heterogeneously deacetylated chitosans. Carbohydr Polym 29:17–24. https://doi.org/10.1016/0144-8617(95)00154-9
Park SH, Lee J-H, Lee HK (2000) Purification and characterization of chitinase from a marine bacterium, Vibrio sp. 98CJ11027. J Microbiol 38:224–229
Pedrali A, Bleve M, Capra P et al (2015) Determination of N-acetylglucosamine in cosmetic formulations and skin test samples by hydrophilic interaction liquid chromatography and UV detection. J Pharm Biomed Anal 107:125–130. https://doi.org/10.1016/j.jpba.2014.12.014
Percot A, Viton C, Domard A (2003) Optimization of chitin extraction from shrimp shells. Biomacromol 4:12–18. https://doi.org/10.1021/bm025602k
Pichyangkura R, Kudan S, Kuttiyawong K et al (2002) Quantitative production of 2-acetamido-2-deoxy-d-glucose from crystalline chitin by bacterial chitinase. Carbohydr Res 337:557–559. https://doi.org/10.1016/S0008-6215(02)00007-1
Rinaudo M (2006) Chitin and chitosan: properties and applications. Prog Polym Sci 31:603–632. https://doi.org/10.1016/J.PROGPOLYMSCI.2006.06.001
Roseman S, Li X, Comb D (2010) Conversion of chitin into N-acetylglucosamine, glucosamine and bioethanol. Patent no. WO/2010/123784
Saito Y, Putaux J-L, Okano T et al (1997) Structural aspects of the swelling of chitin in HCl and its conversion into R Chitin. Macromolecules 30:3867–3873
Saito Y, Okano T, Gaill F et al (2000) Structural data on the intra-crystalline swelling of β-chitin. Int J Biol Macromol 28:81–88. https://doi.org/10.1016/S0141-8130(00)00147-1
Sashiwa H, Fujishima S, Yamano N et al (2002) Production of N-acetyl-d-glucosamine from α-chitin by crude enzymes from Aeromonas hydrophila H-2330. Carbohydr Res. https://doi.org/10.1016/S0008-6215(02)00034-4
Shen C-R, Chen Y-S, Yang C-J et al (2010) Colloid chitin azure is a dispersible, low-cost substrate for chitinase measurements in a sensitive, fast, reproducible assay. J Biomol Screen 15:213–217. https://doi.org/10.1177/1087057109355057
Silva DS, Almeida A, Prezotti FG et al (2017) Self-aggregates of 3,6-O,O′-dimyristoylchitosan derivative are effective in enhancing the solubility and intestinal permeability of camptothecin. Carbohydr Polym 177:178–186. https://doi.org/10.1016/j.carbpol.2017.08.114
Souza CP, Burbano-Rosero EM, Almeida BC et al (2009) Culture medium for isolating chitinolytic bacteria from seawater and plankton. World J Microbiol Biotechnol 25:2079–2082. https://doi.org/10.1007/s11274-009-0098-z
Souza CP, Almeida BC, Colwell RR, Rivera ING (2011) The importance of chitin in the marine environment. Mar Biotechnol 13:823–830. https://doi.org/10.1007/s10126-011-9388-1
Stoykov YM, Pavlov AI, Krastanov AI (2015) Chitinase biotechnology: production, purification, and application. Eng Life Sci 15:30–38. https://doi.org/10.1002/elsc.201400173
Sukwattanasinitt M, Zhu H, Sashiwa H, Aiba S-I (2002) Utilization of commercial non-chitinase enzymes from fungi for preparation of 2-acetamido-2-deoxy-d-glucose from β-chitin. Carbohydr Res 337:133–137. https://doi.org/10.1016/S0008-6215(01)00298-1
Terbojevich M, Carraro C, Cosani A, Marsano E (1988) Solution studies of the chitin-lithium chloride-N, N-di-methylacetamide system. Carbohydr Res 180:73–86. https://doi.org/10.1016/0008-6215(88)80065-X
Villa-Lerma G, González-Márquez H, Gimeno M et al (2013) Ultrasonication and steam-explosion as chitin pretreatments for chitin oligosaccharide production by chitinases of Lecanicillium lecanii. Bioresour Technol 146:794–798. https://doi.org/10.1016/J.BIORTECH.2013.08.003
Wang S-Y, Moyne A-L, Thottappilly G et al (2001) Purification and characterization of a Bacillus cereus exochitinase. Enzyme Microb Technol 28:492–498
Wang S-L, Shih I-L, Liang T-W, Wang C-H (2002) Purification and characterization of two antifungal chitinases extracellularly produced by Bacillus amyloliquefaciens V656 in a shrimp and crab shell powder medium. J Agric Food Chem 50:2241–2248. https://doi.org/10.1021/jf010885d
Wang D, Li A, Han H et al (2018) A potent chitinase from Bacillus subtilis for the efficient bioconversion of chitin-containing wastes. Int J Biol Macromol 116:863–868. https://doi.org/10.1016/J.IJBIOMAC.2018.05.122
Yabuki M, Mizushina K, Amatatsu T et al (1986) Purification and characterization of chitinase and chitobiase produced by Aeromonas hydrophila subsp. anaerogenes A52. J Gen Appl Microbiol 32:25–38
Yan N, Chen X (2015) Sustainability: don’t waste seafood waste. Nature 524:155–157. https://doi.org/10.1038/524155a
Zeng J-B, He Y-S, Li S-L, Wang Y-Z (2012) Chitin Whiskers: an overview. Biomacromol 13:1–11. https://doi.org/10.1021/bm201564a
Zhang Y, Zhou Z, Liu Y et al (2014) High-yield production of a chitinase from Aeromonas veronii B565 as a potential feed supplement for warm-water aquaculture. Appl Microbiol Biotechnol 98:1651–1662. https://doi.org/10.1007/s00253-013-5023-6
Zhu W, Wang D, Liu T, Yang Q (2016) Production of N-Acetyl-d-glucosamine from mycelial waste by a combination of bacterial chitinases and an insect N-acetyl-d-glucosaminidase. J Agric Food Chem 64:6738–6744. https://doi.org/10.1021/acs.jafc.6b03713
Acknowledgements
The authors would like to thank the financial support provided from Sao Paulo Research Foundation (FAPESP), Grants 2012/16824–0, 2013/18773–6 and 2016/20970–2
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
11274_2019_2694_MOESM1_ESM.pdf
Electronic supplementary material 1 (PDF 158 kb) HPLC analysis from GlcNAc bioproduction using an enzyme cocktail produced by A. caviae CHZ306. (a) HPLC profile of N-acetylglucosamine and glucosamine standards; (b) HPLC profile of a representative sample of GlcNAc bioproduction; (c) HPLC profile from blank sample
Rights and permissions
About this article
Cite this article
Cardozo, F.A., Facchinatto, W.M., Colnago, L.A. et al. Bioproduction of N-acetyl-glucosamine from colloidal α-chitin using an enzyme cocktail produced by Aeromonas caviae CHZ306. World J Microbiol Biotechnol 35, 114 (2019). https://doi.org/10.1007/s11274-019-2694-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11274-019-2694-x