Abstract
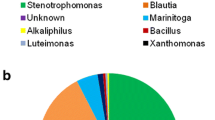
One hundred and fifty soil samples collected from different crop fields in southern Ontario, Canada were screened to obtain microorganisms capable of transforming deoxynivalenol (DON) to de-epoxy DON (dE-DON). Microbial DON to dE-DON transformation (i.e. de-epoxydation) was monitored by using liquid chromatography-ultraviolet-mass spectrometry (LC-UV–MS). The effects of growth substrates, temperature, pH, incubation time and aerobic versus anaerobic conditions on the ability of the microbes to de-epoxydize DON were evaluated. A mixed microbial culture from one composite soil sample showed 100% DON to dE-DON biotransformation in mineral salts broth (MSB) after 144 h of incubation. Treatments of the culture with selective antibiotics followed an elevated temperature (50°C) for 1.5 h considerably reduced the microbial diversity. Partial 16S-rRNA gene sequence analysis of the bacteria in the enriched culture indicated the presence of at least six bacterial genera, namely Serratia, Clostridium, Citrobacter, Enterococcus, Stenotrophomonas and Streptomyces. The enriched culture completely de-epoxydized DON after 60 h of incubation. Bacterial de-epoxydation of DON occurred at pH 6.0–7.5, and a wide array of temperatures (12–40°C). The culture showed rapid de-epoxydation activity under aerobic conditions compared to anaerobic conditions. This is the first report on microbial DON to dE-DON transformation under aerobic conditions and moderate temperatures. The culture could be used to detoxify DON contaminated feed and might be a potential source for gene(s) for DON de-epoxydation.




Similar content being viewed by others
References
Bae HK, Pestka JJ (2008) Deoxynivalenol induces p38 interaction with the ribosome in monocytes and macrophages. Toxicol Sci 105:59–66
Daniel R (2005) The metagenomics of soil. Nat Rev Microbiol 3:470–478
Davis KER, Joseph SJ, Janssen PH (2005) Effects of growth medium, inoculum size, and incubation time on culturability and isolation of soil bacteria. Appl Environ Microbiol 71:826–834
Eriksen GS, Pettersson H, Lundh T (2004) Comparative cytotoxicity of deoxynivalenol, nivalenol, their acetylated derivatives and de-epoxy metabolites. Food Chem Toxicol 42:619–624
Fierer N, Jackson RB (2006) The diversity and biogeography of soil bacterial community. Proc Nat Acad Sci 103:626–631
Foroud NA, Eudes F (2009) Trichothecenes in cereal grains. Int J Mol Sci 10:147–173
Fuchs E, Binder EM, Heidler D, Krska R (2002) Structural characterization of metabolites after the microbial degradation of type A trichothecenes by the bacterial strain BBSH 797. Food Addit Contam 19:379–386
Geng L, Xin W, Haung DW, Feng G (2006) A universal cloning vector using vaccinia topoisomerase I. Mol Biotech 33:23–28
Goodstadt L, Ponting CP (2004) Vitamin K epoxide reductase: homology, active site and catalytic mechanism. Trends Biochem Sci 29:289–292
Guan S, He J, Young JC, Zhu H, Li X, Ji C, Zhou T (2009) Transformation of trichothecene mycotoxins by microorganisms from fish digesta. Aquaculture 290:290–295
He J (2007) Microbial strategies for management of Fusarium graminearum and transformation of deoxynivalenol. Master of Science Thesis. University of Guelph, Guelph, Ontario, Canada
He P, Young LG, Forsberg C (1992) Microbial transformation of deoxynivalenol (vomitoxin). Appl Environ Microbiol 58:3857–3863
Kaplan CW, Kitts CL (2004) Bacterial succession in a petroleum land treatment unit. Appl Environ Microbiol 70:1777–1786
Karlovsky P (1999) Biological detoxification of fungal toxins and its use in plant breeding, feed, and food production. Nat Toxins 7:1–23
King RR, McQueen RE, Levesque D, Greenhalgh R (1984) Transformation of deoxynivalenol (vomitoxin) by rumen microorganisms. J Agric Food Chem 32:1181–1183
Kollarczik B, Gareis M, Hanelt M (1994) In vitro transformation of the Fusarium mycotoxins deoxynivalenol and zearalenone by the normal gut microflora of pigs. Nat Toxins 2:105–110
Lane DJ (1991) 16S/23S rRNA sequencing. In: Stackebrandt E, Goodfellow M (eds) Nucleic acid techniques in bacterial systematics. Wiley, New York, pp 115–175
Matsumoto E, Kawanaka Y, Yun SJ, Oyaizu H (2008) Isolation of dieldrin- and endrin-degrading bacteria using 1, 2-epoxycyclohexane as a structural analog of both compounds. Appl Microbiol Biotech 80:1095–1103
Pestka JJ, Smolinski AT (2005) Deoxynivalenol: Toxicology and potential effects on humans. J Toxicol Environ Health 8:39–69
Rocha O, Ansari K, Doohan FM (2005) Effects of trichothecene mycotoxins on eukaryotic cells: A review. Food Addit Contam 22:369–378
Starkey DE, Ward TJ, Aoki T, Gale LR, Kistler HC, Geiser DM, Suga H, Tóth B, Varga J, O’Donnell K (2007) Global molecular surveillance reveals novel Fusarium head blight species and trichothecene toxin diversity. Fung Genet Biol 44:1191–1204
Swanson SP, Rood JHD, Behrens JC, Sanders PE (1987) Preparation and characterization of the deepoxy trichothecenes: deepoxy HT-2, deepoxy T-2 triol, deepoxy T-2 tetraol, deepoxy 15-monoacetoxyscirpenol, and deepoxy scirpentriol. Appl Environ Microbiol 53:2821–2826
Swanson SP, Helaszek C, Buck WB, Rood JHD, Haschek WM (1988) The role of intestinal microflora in the metabolism of trichothecene mycotoxins. Food Chem Toxicol 26:823–829
Tian F, Yu Y, Chen B, Li H, Yao Y-F, Guo X-K (2009) Bacterial, archaeal and eukaryotic diversity in Arctic sediment as revealed by 16S rRNA and 18S rRNA gene clone libraries analysis. Polar Biol 32:93–103
Ueno Y (1985) The toxicicology of mycotoxins. Crit Rev Toxicol 14:99–132
Völkl A, Vogler B, Schollenberger M, Karlovsky P (2004) Microbial detoxification of mycotoxin deoxynivalenol. J Basic Microbiol 44:147–156
Young JC, Zhou T, Yu H, Zhu H, Gong J (2007) Degradation of trichothecene mycotoxins by chicken intestinal microbes. Food Chem Toxicol 45:136–143
Yu Z, Morrison M (2004) Comparisions of different hypervariable regions of rrs genes for use in fingerprinting of microbial communities by PCR-denaturing gradient gel electrophoresis. Appl Environ Microbiol 70:4800–4806
Zhao JS, Manno D, Thiboutot S, Ampleman G, Hawari J (2007) Shewanella canadensis sp. nov. and Shewanella atlantica sp. nov., manganese dioxide- and hexahydro-1, 3, 5-trinitro-1, 3, 5-triazine-reducing, psychrophilic marine bacteria. Inter J System Evolu Microbiol 57:2155–2162
Zhou T, He J, Gong J (2008) Microbial transformation of trichothecene mycotoxins. World Mycot J 1:23–30
Acknowledgments
The authors gratefully acknowledged the Natural Sciences and Engineering Research Council of Canada (NSERC) for awarding a scholarship to Rafiqul Islam for PhD program. The research was supported by the Agriculture and Agri-Food Canada/Binational Agricultural Research and Development Fund (AAFC/BARD) project. We are thankful for the contributions of Jianwei He, Xiu-Zhen Li and Honghui Zhu.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Islam, R., Zhou, T., Christopher Young, J. et al. Aerobic and anaerobic de-epoxydation of mycotoxin deoxynivalenol by bacteria originating from agricultural soil. World J Microbiol Biotechnol 28, 7–13 (2012). https://doi.org/10.1007/s11274-011-0785-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11274-011-0785-4