Abstract
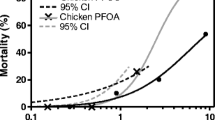
Birds have enough conditions to be used as bioindicators for the presence of environmental contaminants. Notably, chlorpyrifos (CPF) remains extensively employed in Argentina, particularly in fruit plantations and livestock settings This study aimed to assess the potential impacts of CPF on common quail (Coturnix coturnix) embryos through external eggshell application during key embryonic stages (days 1, 4, and 14). Concentrations equivalent to those used in fruit applications, 5× and 10 × (38.4, 192, and 384 µg/egg), were employed. A 48% commercial formulation of CPF (Pirfos - Icona®) was utilized. An increase in embryonic deaths, as well as a statistical association between the degree of liver or kidney lesions and CPF concentrations was observed. The results suggest that CPF can induce embryotoxic effects with a single exposure to quail embryos and compromise the viability of the organisms. The study suggests a potential exposure risk for embryos through contact with the insecticide on the eggshell’s exterior.


Similar content being viewed by others
Data availability
The histological images presented in this article, as well as complementary and underlying data, are supported by the doctoral thesis manuscript available in the digital repository of the National University of Litoral Library, Argentina. URI: https://hdl.handle.net/11185/7100.
References
Ahmad MZ, Khan A, Javed MT, Hussain I (2015) Impact of chlorpyrifos on health biomarkers of broiler chicks. Pest Biochem Physiol 12:50–58. https://doi.org/10.1016/j.pestbp.2014.12.024
Al-Badrany YMA, Mohammad FK (2007) Effects of acute and repeated oral exposure to the organophosphate insecticide chlorpyrifos on open-field activity in chicks. Toxicol Lett 174(1–3):110–116. https://doi.org/10.1016/j.toxlet.2007.09.001
Anwar K and Shakoori AR (2010) Cypermethrin toxicity in the liver of developing chick embryo. Pak J Zool 42(6):725–733
Arrieta-Mendoza D, Perez-Arevalo ML, Luengo A, Hernández JP, Lista-Alves D (2007) Alteraciones histológicas hepáticas e incremento de proteínas séricas en pollos de engorde alimentados con dietas suplementadas con Saccharomyces cerevisiae. Inv Clín 48:341–443
Bhaskar N, Shahani L, Bhatnagar P (2014) Biochemical and histological alterations induced by a formulation of dicofol in the embryonic liver of Gallus domesticus. Toxicol Environ Chem 96:1394–1401. https://doi.org/10.1080/02772248.2014.950267
Braun EJ (1998) Comparative renal function in reptiles, birds, and mammals. Semin avian exot pet med 7(2):62–71. https://doi.org/10.1016/S1055-937X(98)80044-3
Çakıcı Ö (2015) Histopathologic changes in liver and kidney tissues induced by carbaryl in Bufo tesvariabilis (Anura: Bufonidae). Exp Toxicol Pathol 67:237–243. https://doi.org/10.1016/j.etp.2014.12.003
Elsayed MAE, Mohamed NES, Hatab MH, Elaroussi MA (2019) Oxidative stress of in-ovo ochratoxin a administered during chick embryonic development. Braz J Poultry Sci 21:1–10. https://doi.org/10.1590/1806-9061-2017-0637
Ferré DM, Ludueña R, Romano R, Gorla NBM (2020) Evaluation of the genotoxic potential of cypermethrin, chlorpyrifos and their subsequent mixture on cultured bovine lymphocytes. Chemosphere 243:125341. https://doi.org/10.1016/j.chemosphere.2019.125341
García-Fernández AJ, Espín S, Gómez-Ramírez P, Martínez-López E, Navas I (2020) Wildlife sentinels for human and environmental health hazards in ecotoxicological risk assessment. Ecotoxicol QSARs 77–94. https://doi.org/10.1007/978-1-0716-0150-1_4
Huss D, Poynter G, Lansford R (2008) Japanese quail (Coturnix japonica) as a laboratory animal model. Lab Anim 37:513–519. https://doi.org/10.1038/laban1108-513
Kaneko JJ, Harvey JW, Bruss ML (2008) Clinical biochemistry of domestic animals. Academic Press, Cambridge
Mahmood I, Imadi SR, Shazadi K, Gul A, Hakeem KR (2016) Effects of pesticides on environment. In Hakeem KR, Akhtar MS, Abdullah SNA (eds) Plant soil microbes. Springer International Publishing, Cham, pp 253–269. https://doi.org/10.1007/978-3-319-27455-3_13
Martin PA (1990) Effects of carbofuran, chlorpyrifos and deltamethrin on hatchability, deformity, chick size and incubation time of Japanese quail (Coturnix japonica) eggs. Environ Toxicol Chem 9(4):529–534. https://doi.org/10.1002/etc.5620090417
Martul EV (2013) Orientación diagnóstica de las enfermedades renales glomerulares. Rev Esp Patol 46(1):3–13. https://doi.org/10.1016/j.patol.2012.09.003
Moraes TGV, Romao JM, Teixeira RSC, Cardoso WM (2018) Effects of egg position in artificial incubation of Japanese quail eggs (Coturnix japonica). Anim Rep 5:50–54
Raimondo S, Mineau P, Barron MG (2007) Estimation of chemical toxicity to wildlife species using interspecies correlation models. Environ Sci Technol 41:5888–5894. https://doi.org/10.1021/es070359o
Sahu CR, Ghatak S (2002) Effects of dimecron on developing chick embryo: malformations and other histopathological changes. Anat Histol Embryol 31:15–20. https://doi.org/10.1046/j.1439-0264.2002.00355.x
Saraswati TR, Tana S (2015) Development of Japanese quail (Coturnix coturnix japonica) embryo. Int J Sci Eng 8:38–41. https://doi.org/10.12777/ijse.8.1
Slotkin TA, Seidler FJ, Ryde IT, Yanai J (2008) Developmental neurotoxic effects of chlorpyrifos on acetylcholine and serotonin pathways in an avian model. Neurotoxicol Teratol 30:433–439. https://doi.org/10.1016/j.ntt.2008.02.005
Stončius D, Lazutka JR (2003) Spontaneous and benzo[a]pyrene-induced micronuclei in the embryos of the black-headed gull (Larus ridibundus L.). Mutat Res Genet Toxicol Environ Mutagen 538:31–39. https://doi.org/10.1016/S1383-5718(03)00092-5
Acknowledgements
The authors would like to thank the Universidad Juan Agustín Maza for institutional support and to Tech. Sandra Letelier for her invaluable assistance in sample processing.
Funding
Authors has received research support from Juan Agustín Maza University.
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Conceptualization: NBG. Methodology: AMQ, AP. Formal analysis and investigation: AMQ, AP and NG. Funding acquisition: NBG. The first draft of the manuscript was written by NBG and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval
The methodology used was approved by the Institutional Committee for the Care and Use of Laboratory, Research and Teaching Animals (CICUALE), Juan Agustín Maza University (Protocol No. 119/Year 2016). DGXI European Commission recommendations regarding the protection of animals used for experimental and scientific purposes were followed (EU, 2010).
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Quero, A.A.M., Pedrosa, A. & Gorla, N.B.M. Liver and kidney damage in quail embryos hatched from eggs sprayed once with chlorpyrifos during artificial incubation. Vet Res Commun 48, 1285–1291 (2024). https://doi.org/10.1007/s11259-024-10298-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11259-024-10298-z