Abstract
Alternation of day and night is the oldest cycle on Earth, which is increasingly disturbed by the accelerating rate of urbanization and technological development. Despite the ubiquity of light pollution in cities, many aspects of its influence on urban ecosystems are still poorly understood. Here we studied the effect of artificial light at night (ALAN) on the biomass of arboreal caterpillar populations, which are a major component of the diet of many insectivorous animals. We predicted that increasing ALAN intensity is associated with reduced caterpillar biomass, because ALAN may increase predation risk for both caterpillars and adult lepidopterans (i.e. moths), and can also hinder the moths’ reproductive rate. We estimated caterpillar biomass from frass samples (n = 3061) collected from 36 focal trees in two cities in Hungary during four consecutive years. To quantify ALAN we measured light intensity during night at each focal tree (range of illumination: 0.69–3.18 lx). We found that caterpillar biomass of individual trees was repeatable over the four years. This temporal consistency in prey biomass production may be important for birds because it can help predict territory quality, especially in cities where caterpillar abundance is generally low. Our results did not support the negative effect of ALAN on urban caterpillar populations, because ALAN intensity was not related to caterpillar biomass, and this lack of effect was consistent between study sites and tree species. We suggest that the effect of ALAN on urban caterpillar biomass is either weak and thus can be masked by other, local environmental factors, or light pollution may have antagonistic effects acting during different stages of the lepidopteran life cycle. Another explanation could be that even the lower levels of our sites’ public lighting are strong enough to cause serious detrimental effects for caterpillars, resulting in their uniformly low biomass.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The day-night cycle is one of the oldest natural cycles on Earth. Most living organisms are influenced by light or its absence, hence the natural light-dark cycles are vital for them. Light pollution by artificial light at night (ALAN) alters this cycle. Although light pollution is a longstanding concern and the evidence for its wide-ranging negative effects on the biota is continuously mounting, the amount of ALAN is still increasing globally with an average of ca. 6% per year (0 to 20% depending on location, Hölker et al. 2010; Gaston et al. 2013; Davies and Smyth 2018), mostly as a consequence of urbanization. The effects of ALAN can be observed at every level of biological organization: it affects cells (e.g. by disrupting circadian rhythm of cell division), individuals (e.g. by changing behaviour during orientation, reproduction or communication) and even communities (by affecting competition, predation), and it is also listed as a major threat to biodiversity (Woelfle et al. 2004; Rich and Longcore 2006; Hölker et al. 2010; Brown 2014; Knop et al. 2017).
ALAN can have particularly strong effects on nocturnal organisms that are attracted to light sources, such as adult lepidopterans (Schacht and Witt 1986; Simon and Bradstreet 1991; Eisenbeis and Hänel 2009), potentially disturbing all aspects of their life cycle. For example, light pollution negatively affects moth reproduction via multiple mechanisms: it can inhibit the release of female sex pheromones (Sower et al. 1970; Van Geffen et al. 2015), and it is also likely to disrupt mate finding behaviour when males favour approaching light sources over the pheromone signals of females (Delisle et al. 1998). Furthermore, ALAN can also suppress oviposition (Nemec 1969), inhibit pupal diapause and reduce the size and age at the pupation of caterpillars (Van Geffen et al. 2014), ultimately resulting in their decreased fitness. The predation rate of moths (by bats, birds and predatory arthropods) also can be higher near light sources, for at least two reasons. First, because high density of prey attracts higher number of predators (Simon and Bradstreet 1991; Rydell 1992), and second, because ALAN hinders moths’ defensive behaviour against bats by impeding the moths’ ultrasound detection and emittance, leading to increased susceptibility to bat predation (Acharya and Fenton 1999). Light sources in cities artificially increase the length of natural photoperiod which in turn has been demonstrated to greatly increase larval growth rates – however, fast-growing larvae were also significantly more prone to predation, revealing a trade-off between growth rate and predation risk (Gotthard 2000). Additionally, birds residing at more illuminated territories have prolonged daily activity periods (Tarlow et al. 2003; Dominoni et al. 2013, 2014) which might lead to increased foraging times, thus elevated predation rates on moths and caterpillars. Combined together, these ALAN-induced processes can greatly contribute to the local and regional decline of lepidopteran populations. For example, in Germany during a single summer the number of moths killed by light sources was estimated to reach the scale of 1011 (Bauer 1993).
Despite the obvious negative effects documented in some populations (see above), other studies argue that ALAN does not necessarily have a strong influence on lepidopterans. For example, a study comparing natural (ALAN free) and urban populations of the small ermine moth (Yponomeuta cagnagella) found that urban populations living under long term exposure to ALAN are less attracted to light sources, perhaps as a consequence of an adaptive response (Altermatt and Ebert 2016). Similarly, the experiment of Van Grunsven et al. (2014) demonstrated that, when released from the close proximity of an artificial light source, almost 75% of moth individuals (including different species) were not attracted by the experimental light source. The spectral composition of the emitted light is also very important, as lamps emitting shorter wavelengths (around 380 nm, i.e. blue and violet) attract significantly more moths (species and individuals alike) than light sources with longer wavelengths (617 nm, i.e. red; van Langevelde et al. 2011). The severity of ALAN’s impacts on nocturnal moth communities varies considerably between studies, as it depends on factors like species composition of local communities (because susceptibility to light pollution differs between moth taxa; van Langevelde et al. 2011; Van Grunsven et al. 2014) and local characteristics of the natural and artificial light circumstances (Eisenbeis and Hänel 2009).
These studies clearly illustrate the complex and diverse effects of ALAN on lepidopterans; thus, to get a better understanding on its actual impacts, it is necessary to conduct more studies in different environments, on more species and communities, involving different stages of the lepidopteran life cycle. As the significant majority of ALAN comes from urban areas, it is important to explore the impacts of light pollution on lepidopterans in our built-up environment. Furthermore, although several studies tested the effects of ALAN on adult moths, relatively few studies focused on their larvae, the caterpillars, and these produced contrasting results. Welbers et al. (2017) studied the effect of ALAN in a deciduous forest, with experimentally altering light conditions by street lamps emitting different colours of lights. This study found significantly higher peak caterpillar abundances in trees with green and white light than with red light or without light. Van Geffen et al. (2014), by contrast, found a negative effect of green and white light on the development of caterpillars: male caterpillars had lower body mass and pupated earlier than under red light or dark treatments. A third study found that the length of the illumination can also have influence on the abundance of caterpillars, because of its effect on the level of predation by predatory insects Gotthard (2000).
Caterpillars are important food items for several insectivorous bird species occupying urban habitats (e.g. great tit Parus major, blue tit Cyanistes caeruleus, Naef-Daenzer and Keller 1999) and for predatory arthropods (Ferrante et al. 2014), while at their imago life stage they are preferred preys of urban-dwelling bat species (e.g. common noctule Nyctalus noctule or common pipistrelle Pipistrellus pipistrellus; Rydell 2006), and also play an important role as pollinators (Macgregor et al. 2015). Therefore, in this study our aim is to investigate the effect of artificial light at night on tree-dwelling caterpillar populations in urban areas. Due to the various negative effects that ALAN can exert on both adult moths and caterpillars (see above) we predicted decreased caterpillar biomass on trees that are exposed to higher intensity of ALAN. To explore the relationship between caterpillar biomass and light pollution, we collected data on caterpillar biomass from several tree species throughout four consecutive years (2014–2017), from two cities with differing urban environments.
Materials and methods
Study sites and selected tree species
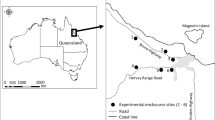
We conducted our study in two cities in Hungary, Balatonfüred (46°57′30”N, 17°53′34″E) and Veszprém (47°05′17”N, 17°54′29″E). Both cities are characterized by typical urban vegetation, i.e. maintained green areas such as public parks, street tree lines, and suburban gardens. However, the two urban sites differ in their intensity of urban development: the study site in Balatonfüred is an urban park with continuous and relatively dense vegetation, and typically has detached, single-storeyed houses, whereas the study site in Veszprém consists of more roads, impervious surfaces and apartment blocks, and scattered patches of less dense vegetation (Electronic Supplementary Material (ESM), Fig. S1). Following the study setup of Seress et al. (2018) we identified the three most common tree species in each study site (by field survey conducted in 2013) and selected six individual trees per species as focal individuals (n = 18 trees in Balatonfüred and n = 18 in Veszprém; see ESM, Fig. S1). We used these focal trees for caterpillar biomass monitoring and light intensity measurements (see below). In Balatonfüred, the selected tree species were small-leaved lime (Tilia cordata), sessile oak (Quercus petraea) and Norway maple (Acer platanoides), whereas in Veszprém we chose silver linden (Tilia tomentosa), horse-chestnut (Aesculus hippocastanum) and Norway maple. The rationale behind this sampling strategy was to estimate caterpillar abundance as experienced by foraging birds (Seress et al. 2018). Furthermore, the sampling of the most common tree species at each study site increased our chance to get a general picture of urban caterpillar abundance and its response to ALAN intensity for each site, compared to a sampling regime that would focus on a single common tree species at both sites (e.g. the Norway maple). Note that, the trees in our two urban locations are characterized by significantly lower caterpillar abundance than forest trees, which is consistent with the findings of several other studies (see Seress et al. 2018 for a detailed discussion of caterpillar biomass in urban areas).
Caterpillar biomass
To quantify arboreal caterpillar biomass, we used the commonly applied frassfall method (Tinbergen and Dietz 1994) and collected frass (insect droppings) samples from our focal trees. The method has been described in detail in Seress et al. (2018), here we summarize it only briefly. From February, we monitored the leaf emergence phenology individually for each focal tree and started sampling frass when >50% of the buds had emerged and the shape of the leaves was clearly recognizable. We suspended frassfall traps (cheese cloth net attached to a 0.5 × 0.5 m wooden frame) under the canopies of the focal trees (one trap per tree) and collected frass samples from the traps every 3–5 days (depending on the prevailing weather conditions) between March and June in four consecutive years (from 2014 to 2017). This sampling period corresponds to the breeding season of great tits and several other insectivorous birds for whom caterpillars provide the main component of nestling diet. The collected frass samples were dried at room temperature (additional drying was not required, for further details see Seress et al. (2018), sorted (i.e., debris and powder removed) and weighed to the nearest mg. From the dry frass mass and temperature data (recorded by weather loggers (Voltcraft DL 101 T) throughout the whole sampling period) we calculated caterpillar biomass expressed as hourly caterpillar biomass (mg/h/0.25m2) using the equation of Tinbergen and Dietz (1994). From Balatonfüred we had 397 (2014), 447 (2015), 390 (2016), 296 (2017) frass samples and from Veszprém 386 (2014), 429 (2015), 404 (2016) and 312 (2017) samples.
Because canopy volume above the frassfall traps can directly affect the amount of frass production, and canopy height is usually correlated with canopy volume (Troxel et al. 2013), we estimated the height of each focal tree’s canopy (to the nearest m) to control for the potential differences in canopy height (for further details of the field methods see Seress et al. 2018). Because vegetation density in the surroundings of our focal trees can also influence caterpillar abundance (Blair and Launer 1997), to control for this variable, we also calculated the percentage of area covered by tree canopies in the 30 m radius of each focal tree (henceforth ‘canopy cover’) from orthophotos taken in 2015 using the QGIS software (QGIS Development Team 2016).
Night light intensity measurements
We characterized the focal trees’ night light regimes by conducting light intensity measurements in 2017, between 12 January and 8 April, intentionally before foliage development, to avoid the shading effect). The public lighting system in our study area consists mostly of high-pressure sodium lamps (HPS lamps with orange light, i.e. a narrow emitting spectrum peaking around 600 nm), both on streets and in park areas, with only a small fraction of the surveyed light sources being LED lights (three lamps, 6% at Veszprém, and none at Balatonfüred). Given that there were no significant changes in the physical structure (e.g. number of buildings, roads and density of vegetation) and street-lighting system of our study sites between 2014 and 2017, we assume that our focal trees’ night light environment did not change between the study years. We recorded the level of ALAN (in lux) during one whole night for each focal tree with a purpose-built, calibrated light-logger (for its detailed description see ESM, ‘Additional details on methods: light logger description’) which was suspended in the focal tree’s canopy at 2.5–3 m above ground and ca. 1.5–2 m from the tree trunk in order to reduce the shading effect. The logger had three sensors around its perimeter, enabling us to detect light sources from every direction, and each sensor recorded light intensity every second (range of sensitivity: 188*10−6 – 88*103 lx). Light intensity measurements were conducted during the astronomical night (i.e., when the center of the Sun is at least 18 degrees below the horizon; starting and ending times were determined separately for each date from the timeanddate.com database, Time and Date AS © (www.timeanddate.com). The very high number of recordings per tree [mean ± SE = 17,755 ± 2799, range: 12999–20,821] yielded a detailed picture of the ALAN regimes for each focal tree (for visualised light intensity recording examples see: ESM, Fig. S2). To characterize ALAN intensity for each focal tree, we used the mean light intensity calculated as the mean of all recordings of the three sensors.
To assess the validity of our light intensity measurements, we performed two sets of analyses. First, to test the repeatability of the light-logger’s recordings, we selected five focal trees in our study site in Veszprém and conducted five repeated light measurements on each of them (i.e. during 25 consecutive nights) as follows. After each night (measurement) the light-logger was relocated to a different focal tree, and after one set of measurements (i.e. one measurement per each point, 5 nights), we again hung the logger to the first focal tree to start the next set of measurements. We found that the mean light intensity values were highly repeatable between these consecutive measurements (intra class correlation, using the R ‘ICC’ (Wolak 2015) and function ICCest, rICC = 0.84; CI: 0.62–1.05; N = 5, k = 5), indicating that the variability of measured light levels on the same tree was much lower than the variability between trees. Additionally, to test if there was any consistent bias in the mean values of the repeated measurements (e.g. due to a several days long cloudy period followed by a several days long moon-lit period), we built a linear mixed-effects model (LME, using the function ‘lme’ of the package ‘nlme’; Pinheiro et al. 2013) that contained mean light intensity as dependent variable, measurement set ID as a fixed five-level factor and focal tree ID as a random factor. This model indicated no significant differences between the repeated measurements (F = 2.02, df = 16, p = 0.14). Because of the high repeatability of measurements, we decided to record light intensity for only one night for each focal tree to describe the intensity of ALAN in their surroundings.
Second, after sunset we surveyed the number of artificial light sources (street lamps and household light sources) within the 25 m radius around each focal tree, and tested the correlation between the number of light sources and the mean light intensity values recorded by the light-logger (see above). The rationale behind using an area of 25 m radius for the light source survey was that a street lamp is typically capable to illuminate its immediate surrounding area with 5 lx (corresponding to the Hungarian recommendations for outdoor lighting; Arató 2003), and light intensity drops close to zero at 25 m distance from the source. Our survey indicated that the number of artificial light sources ranged between 0 and 9 per focal tree within 25 m (ESM, Fig. S2), and the number of light sources was significantly and positively correlated with light intensity recorded by our light-logger for the same focal trees (Kendall’s rank correlation, τ = 0.365, p < 0.01, n = 36 trees).
Statistical analyses
We tested the repeatability of caterpillar biomass measured on the focal tree individuals across the four study years using a generalized linear mixed-effects approach as implemented by the ‘rpt’ command of package ‘rptR’ (Nakagawa and Schielzeth 2010). In this model the dependent variable was the log transformed mean amount of caterpillar biomass (mg/h/0.25m2) of individual trees calculated for each year separately, the predictors were the year of sample collection (as a factor) and the tree species, and the random factor was the trees’ ID.
Because the urban habitat characteristics of the two sites differed markedly (see study site descriptions above), we compared the ALAN regimes between the study sites. In order to do so, we compared the mean light intensity values measured for individual trees by a Mann-Whitney U Test, due to the non-normal distribution of the data.
Finally, we investigated the relationship between mean night light intensity and caterpillar biomass in an LME model. In the initial model, caterpillar biomass ((mg/h/0.25m2) values were log-transformed using the formula loge(x + 0.0001)) was the dependent variable (using the mean value of each tree from each sampling year, i.e. the annual mean biomass of each individual tree), while predictors were mean light intensity, study site, year of sample collection (as a factor), tree species, canopy height and canopy cover. The model also included the light intensity × study site and light intensity × tree species interactions and tree ID as random factor. Our idea behind testing the light intensity × study site interaction was that light pollution could have site-specific effects on caterpillar populations because of the different habitat characteristics. We tested the light intensity × tree species interaction because different tree species may have different caterpillar fauna that could have different responses to ALAN. The initial model was reduced by backwards stepwise model selection, excluding the term (interaction or main effect) with the highest P value in each step until only significant (p < 0.05) terms remained. Additionally, by conducting pairwise post-hoc tests (using the emmeans function from the ‘emmeans’ package; Lent et al. 2018) we also compared the mean caterpillar biomass (with Tukey method) between tree species and sampling years as estimated from the results of the final LME model. Because of the spatial structure of our sampling locations both within and among sites, we built an additional model to test spatial autocorrelation using the package ‘ncf’(Bjornstad 2019). We controlled for spatial autocorrelation by updating our LME models described above with data on the spatial coordinates of the trees. All statistical analyses were performed in the R statistical environment (R Core team 2018).
Results
We found that focal tree individuals consistently differed in their caterpillar biomass production: the repeatability of caterpillar biomass of individual trees across the four years was low but statistically significant (R = 0.279, p < 0.001).
Mean night light intensity and its standard deviation in the canopy of trees was 1.52 ± 0.79 lx (range: 0.69–3.18 lx) in Veszprém, whereas tree canopies in Balatonfüred were darker, with a mean value and standard deviation of 0.76 ± 0.1 lx (range: 0.68–1.05 lx). The difference between the two sites was statistically significant (W = 295, p < 0.01, Mann-Whitney U Test).
Caterpillar biomass was not related to the night light intensity measured on the same focal trees (Table 1), and this lack of effect seems robust. First, light intensity was unrelated to caterpillar biomass when the two study sites were analysed together in a model that controlled for the effects of potential confounding variables (Table 1, Fig. 1). Second, we got similar results when the two study sites were analysed separately (ESM, Table S1). Third, the light intensity × study site and light intensity × tree species interactions were non-significant (Table 1), indicating that the relationship between light intensity and caterpillar biomass did not change between sites or between different tree species. In our final LME model, only tree species and study year had significant effects on caterpillar biomass (Table 1). Similarly, light intensity was consistently unrelated to caterpillar biomass when we analysed the tree species separately (ESM, Table S2). The pairwise post-hoc comparisons indicated that sessile oaks supported the highest caterpillar biomass, differing significantly from every other tree species, except for the silver linden (ESM, Table S3; Fig. S3). The pairwise post-hoc comparisons between years showed that caterpillar biomass was significantly lower in 2016 compared to all other years (ESM, Table S4; Fig. S4), but it did not differ significantly between the other years. We detected spatial autocorrelation in the data in both locations, but caterpillar biomass remained unrelated to light intensity in the models that controlled for this effect (ESM, Table S5).
Discussion
Light pollution is a global environmental problem to which a broad range of organisms are very sensitive. Here we used correlational data from two urban study sites to explore whether ALAN influences the population sizes of arboreal caterpillars that play important roles in many ecosystems including forested urban areas. Although ALAN is thought to have various impacts on both adult and larval lepidopterans (see Introduction), we found no strong effects of light pollution on local caterpillar abundances. We believe this result is robust for several reasons.
First, our analyses produced consistent conclusions between two urban sites and between different tree species. In both study sites, the range of light intensity values we recorded (0.68–3.18 lx) were within the expected and recommended outdoor street lighting levels (Arató 2003; Fotios and Goodman 2012). Another study investigating the effect of ALAN on avian reproductive physiology used the same lighting range to mimic urban night light conditions (Dominoni et al. 2013). Light intensity levels (mean lux values) differed significantly among our study sites, which can be explained with the size and the structure of the two cities: Veszprém is larger and the distribution of sampled trees in the city is more scattered, thus trees are more exposed to street lights. In Balatonfüred all sampled trees were in a continuous, central park region where street lighting has lower intensity and the vegetation is denser. Despite this difference between the two study sites in lighting levels, the effect of ALAN was consistently non-significant in both cities.
Second, we found that the caterpillar biomass of individual trees was significantly repeatable across the four study years. Considering the dependence of caterpillar biomass on the climatic conditions (Reynolds et al. 2007) and also that environmental conditions often strongly fluctuate between years, this detectable consistency within individual trees is remarkable. The repeatability cannot be explained by differences between tree species, since we controlled for that effect in the analysis. Our results also do not support that it is simply related to canopy height and canopy cover, since these factors did not influence the measured caterpillar biomass in our study sites. It may be explained, however, by other tree traits like age, or nutritional and immune state (Kaitaniemi and Ruohomäki 2001a; Howe and Jander 2008) that we did not assess in our study. This year-to-year consistency in prey biomass of individual trees may be important for birds and other predators of arboreal caterpillars because it can help them to predict territory quality, especially in urban environments where caterpillar abundance is generally low.
For the lack of association between light pollution intensity and caterpillar biomass in our study system there are several possible explanations. One potential reason can be that other environmental and ecological factors that we were not able to control for could have masked ALAN’s effects, by having stronger impacts on caterpillar biomass than the recorded variation in ALAN. One such environmental factor is the altered urban vegetation, including the presence of non-native species that replace caterpillars’ original host plants (Burghardt et al. 2010), as well as variation in vegetation structure, since denser or more open parts of the canopies provide different habitat qualities for caterpillars (Roland 1993; Dulaurent et al. 2011). Further possible detrimental effects may result from intensive urban vegetation management practices, like frequent mowing of grass and removal of leaf litter (typical in our study sites), as these activities reduce the availability of suitable locations for caterpillars to pupate. Furthermore, local microclimate, which depends on multiple factors, can also have great influence on caterpillar abundance (Casey et al. 1988; Moore et al. 1988; Savilaakso et al. 2009) and may be highly variable within cities. The health status of trees (Miller et al. 2006; Dale and Frank 2014) and as a consequence their nutrition content or resistance ability against herbivores (Kaitaniemi and Ruohomäki 2001b) is also among the important caterpillar biomass determining factors, just like the local predation pressure (Kozlov et al. 2017). All of these diverse factors could affect microhabitat quality to a different extent and their interactions may further increase the small-scale spatial heterogeneity.
Another possible explanation for the absence of ALAN’s effect on caterpillar biomass is that light pollution could have antagonistic effects on the same lepidopteran populations, e.g. during their different life stages. For example, while ALAN has many potentially detrimental effects on adults (moths, see Introduction), it could have some beneficial effects during the larval stages. For example, ALAN could positively affect caterpillar development directly, by accelerating larval growth rates (Gotthard 2000) or indirectly, via enhancing host plant quality as a food source (Wang 2008), either by increasing plant growth rate (Cathey and Campbell 1975), or seasonally advancing the timing of budburst (Ffrench-Constant et al. 2016). A recent study conducted in an oak forest found that green and white LED light has a major positive effect on local caterpillar biomass compared to plots with red light or no artificial illumination (Welbers et al. 2017). Interestingly, the effect of ALAN on peak caterpillar biomass was prominent only at the study site with highest caterpillar abundance and not in other study sites with evergreen vegetation and much lower caterpillar abundance. Whether these results could be generalized in urban systems is an open question, however in many cities (including our study sites), the most common public light sources are HPS lamps. HPS lamps produce an orange light which might mitigate the impacts of ALAN given that caterpillars appear more responsive to blue and green light (Welbers et al. 2017). Furthermore, several studies have reported significantly lower caterpillar biomass in urban compared to forested areas (reviewed by Seress et al. 2018), in contrast to the increase in biomass reported by Welbers et al. (2017) under green and white light. These differences might all contribute to that we did not find any apparent effect of artificial light pollution on caterpillar biomass.
In our analyses, tree species was a significant predictor of caterpillar biomass. Other studies have also showed that e.g. oak species in general support higher amounts and diversity of phytophagous arthropods compared to other tree species (Csóka 1998), similarly to that we found here. Thus, it is also possible that our sampled tree species differ in their local moth faunas, and the variability in different moth species’ responses to ALAN result in that we found no overall relationship when all trees species were analysed together. Our results do not support this theory, however. We did not find a significant interaction effect between light intensity and tree species on caterpillar biomass (Table 1), and the effect of light intensity was also non-significant for any of the sampled tree species, including oaks, when these were analysed separately (ESM, Table S2). Our knowledge on the Hungarian moth fauna further supports this conclusion, because usually only a few common species contribute to the major part of the annual moth biomass (Valtonen et al. 2017).
It is important to note that in our case even the lowest recorded lux values (0.68 lx at site Balatonfüred) were still at least 6 times brighter than natural night light conditions. Thus, a further explanation for our results could be that even very low levels of public lighting (e.g. similar to the lower values of the range in our survey) are strong enough to cause serious detrimental effects in arboreal caterpillar populations, resulting in uniformly low caterpillar biomass on urban trees. Indeed, several studies have noted significant ecological impacts at artificial light intensities below 1 lx (e.g. Azam et al. 2018; Sullivan et al. 2019). Finally, it is also possible that the abundance of caterpillars in urban areas is less responsive to ALAN intensity than we previously thought. Local caterpillar abundances are strongly depending on the behaviour of adults, and several studies imply that not all moth species are sensitive to light or light pollution (e.g. see van Langevelde et al. 2011; Van Geffen et al. 2014; Altermatt and Ebert 2016). This idea is also supported by the review of Fox (2013) that investigated the potential causes of declines in moth populations in Great Britain, and failed to find any direct evidence for ALAN being a major cause. Furthermore, perhaps urban moth populations are less susceptible to light pollution either due to the differences in species composition between urban and non-urban lepidopteran communities (New 2015; Lizee et al. 2016), or due to local adaptations to the urban habitats (i.e. if the individuals most sensitive to ALAN are selected against). In line with these assumptions, a recent study found that individuals from urban moth populations living under an increased exposure to ALAN for a long time are significantly less attracted by light sources compared to individuals from pristine populations (Altermatt and Ebert 2016).
In summary, our results show no significant effect of ALAN on urban caterpillar biomass, suggesting that other ecological factors are more important drivers of variation in caterpillar abundance in cities – thus, their effects should be investigated in more detail. However, we feel important to note that our conclusions are based on correlational results, and this approach may have limited power for detecting the effect of ALAN either due to various confounding environmental factors or due to possible antagonistic effects of light pollution (see above). To have a clearer knowledge on how and to which extent light pollution affects urban caterpillar populations we would definitely need more experimental studies, for example in which ALAN intensity is experimentally manipulated around urban trees.
References
Acharya L, Fenton MB (1999) Bat attacks and moth defensive behaviour around street lights. Can J Zool 77:27–33. https://doi.org/10.1139/z98-202
Altermatt F, Ebert D (2016) Reduced flight-to-light behaviour of moth populations exposed to long-term urban light pollution. Biol Lett 12:20160111. https://doi.org/10.1098/rsbl.2016.0111
Arató A (2003) VILÁGÍTÁSTECHNIKA, 2.1. HOLUX Kft., Budapest
Azam C, Le Viol I, Bas Y et al (2018) Evidence for distance and illuminance thresholds in the effects of artificial lighting on bat activity. Landsc Urban Plan 175:123–135. https://doi.org/10.1016/j.landurbplan.2018.02.011
Bauer R (1993) Untersuchung zur Anlockung von nachtaktiven Insekten durch Beleuchtungseinrichtungen. Verlag nicht ermittelbar
Bjornstad ON (2019) Spatial Covariance Functions. https://cran.r-project.org/web/packages/ncf/ncf.pdf. Accessed 22 Mar 2019
Blair RB, Launer AE (1997) Butterfly diversity and human land use: species assemblages along an urban gradient. Biol Conserv 80:113–125. https://doi.org/10.1016/S0006-3207(96)00056-0
Brown SA (2014) Circadian clock-mediated control of stem cell division and differentiation: beyond night and day. Development 141:3105–3111. https://doi.org/10.1242/dev.104851
Burghardt KT, Tallamy DW, Philips C, Shropshire KJ (2010) Non-native plants reduce abundance, richness, and host specialization in lepidopteran communities. Ecosphere 1:1–22. https://doi.org/10.1890/ES10-00032.1
Casey TM, Joos B, Fitzgerald TD, Yurlina ME, Young PA (1988) Synchronized group foraging, thermoregulation, and growth of eastern tent caterpillars in relation to microclimate. Physiol Zool 61:372–377. https://doi.org/10.1086/physzool.61.4.30161255
Cathey HM, Campbell LE (1975) Effectiveness of five vision-lighting sources on photo-regulation of 22 species of ornamental plants. J Am Soc Hortic Sci 100:65–71
Csóka G (1998) Insect herbivore guild of the oaks native to Hungary. Erdészeti Kut 88:311–318
Dale AG, Frank SD (2014) The effects of urban warming on herbivore abundance and street tree condition. PLoS One 9:201407. https://doi.org/10.1371/journal.pone.0102996
Davies TW, Smyth T (2018) Why artificial light at night should be a focus for global change research in the 21st century. Glob Chang Biol 24:872–882
Delisle J, West RJ, Bowers WW (1998) The relative performance of pheromone and light traps in monitoring the seasonal activity of both sexes of the eastern hemlock looper, Lambdina fiscellaria fiscellaria. Entomol Exp Appl 89:87–98. https://doi.org/10.1023/A:1003450432755
Dominoni D, Quetting M, Partecke J (2013) Artificial light at night advances avian reproductive physiology. Proc R Soc B Biol Sci 280:20130213. https://doi.org/10.1098/rspb.2012.3017
Dominoni DM, Carmona-Wagner EO, Hofmann M, Kranstauber B, Partecke J (2014) Individual-based measurements of light intensity provide new insights into the effects of artificial light at night on daily rhythms of urban-dwelling songbirds. J Anim Ecol 83:681–692. https://doi.org/10.1111/1365-2656.12150
Dulaurent AM, Porté AJ, van Halder I, Vétillard F, Menassieu P, Jactel H (2011) A case of habitat complementation in forest pests: pine processionary moth pupae survive better in open areas. For Ecol Manag 261:1069–1076. https://doi.org/10.1016/j.foreco.2010.12.029
Eisenbeis G, Hänel A (2009) Light pollution and the impact of artificial night lighting on insects. In: ecology of cities and towns: a comparative approach. Pp 243–263
Ferrante M, Lo Cacciato A, Lövei GL (2014) Quantifying predation pressure along an urbanisation gradient in Denmark using artificial caterpillars. Eur J Entomol 111:649–654. https://doi.org/10.14411/eje.2014.082
Ffrench-Constant RH, Somers-Yeates R, Bennie J et al (2016) Light pollution is associated with earlier tree budburst across the United Kingdom. Proc R Soc B Biol Sci 283:20160813. https://doi.org/10.1098/rspb.2016.0813
Fotios S, Goodman T (2012) Proposed UK guidance for lighting in residential roads. In: Lighting Research and Technology. pp 69–83
Fox R (2013) The decline of moths in Great Britain: a review of possible causes. Insect Conserv Divers 6:5–19. https://doi.org/10.1111/j.1752-4598.2012.00186.x
Gaston KJ, Bennie J, Davies TW, Hopkins J (2013) The ecological impacts of nighttime light pollution: a mechanistic appraisal. Biol Rev 88:912–927. https://doi.org/10.1111/brv.12036
Gotthard K (2000) Increased risk of predation as a cost of high growth rate: an experimental test in a butterfly. J Anim Ecol 69:896–902. https://doi.org/10.1046/j.1365-2656.2000.00432.x
Hölker F, Wolter C, Perkin EK, Tockner K (2010) Light pollution as a biodiversity threat. Trends Ecol Evol 25:681–682
Howe GA, Jander G (2008) Plant immunity to insect herbivores. Annu Rev Plant Biol 59:41–66. https://doi.org/10.1146/annurev.arplant.59.032607.092825
Kaitaniemi P, Ruohomäki K (2001a) Sources of variability in plant resistance against insects: free caterpillars show strongest effects. Oikos. 95:461–470. https://doi.org/10.1034/j.1600-0706.2001.950311.x
Kaitaniemi P, Ruohomäki K (2001b) Sources of variability in plant resistance against insects: free caterpillars show strongest effects. Oikos 95:461–470. https://doi.org/10.1034/j.1600-0706.2001.950311.x
Knop E, Zoller L, Ryser R, Gerpe C, Hörler M, Fontaine C (2017) Artificial light at night as a new threat to pollination. Nature 548:206–209. https://doi.org/10.1038/nature23288
Kozlov MV, Lanta V, Zverev V, Rainio K, Kunavin MA, Zvereva EL (2017) Decreased losses of woody plant foliage to insects in large urban areas are explained by bird predation. Glob Chang Biol 23:4354–4364. https://doi.org/10.1111/gcb.13692
Lent R, Singmann H, Love J, et al (2018) Emmeans: estimated marginal means, aka least-squares means. R Packag. version 1
Lizee MH, Tatoni T, Deschamps-Cottin M (2016) Nested patterns in urban butterfly species assemblages: respective roles of plot management, park layout and landscape features. Urban Ecosyst 19:205–224. https://doi.org/10.1007/s11252-015-0501-5
Macgregor CJ, Pocock MJO, Fox R, Evans DM (2015) Pollination by nocturnal Lepidoptera, and the effects of light pollution: a review. Ecol Entomol 40:187–198
Miller TEX, Tyre AJ, Louda SM (2006) Plant reproductive allocation predicts herbivore dynamics across spatial and temporal scales. Am Nat 168:608–616. https://doi.org/10.1086/509610
Moore LV, Myers JH, Eng R (1988) Western tent caterpillars prefer the sunny side of the tree, but why? Oikos 51:321. https://doi.org/10.2307/3565313
Naef-Daenzer B, Keller LF (1999) The foraging performance of great and blue tits (Parus major and P. caeruleus) in relation to caterpillar development, and its consequences for nestling growth and fledging weight. J Anim Ecol 68:708–718. https://doi.org/10.1046/j.1365-2656.1999.00318.x
Nakagawa S, Schielzeth H (2010) Repeatability for Gaussian and non-Gaussian data: a practical guide for biologists. Biol Rev 85:935–956
Nemec SJ (1969) Use of artificial lighting to reduce Heliothis Spp. populations in cotton Fields1. J Econ Entomol 62:1138–1140. https://doi.org/10.1093/jee/62.5.1138
New TR (2015) Insect conservation and urban environments
Pinheiro J, Bates D, DebRoy S, et al (2013) nlme: Linear and Nonlinear Mixed Effects Models. R package version 3.1–120, http://CRAN.R-project.org/package=nlme. J. Apic. Res. 1–339
QGIS Development Team (2016) QGIS geographic information system. Open Source Geospatial Found. Proj
R Core team (2018) R Core Team. R A Lang. Environ. Stat. Comput. R Found. Stat. Comput. , Vienna, Austria. ISBN 3–900051–07-0, URL http//www.R-project.org/.R Core Team (2016). R A Lang. Environ. Stat. Comput. R Found 55:275–286
Reynolds LV, Ayres MP, Siccama TG, Holmes RT (2007) Climatic effects on caterpillar fluctuations in northern hardwood forests. Can J For Res 37:481–491. https://doi.org/10.1139/X06-211
Rich C, Longcore T (2006) Ecological consequences of artificial night lighting. Island Press
Roland J (1993) Large-scale forest fragmentation increases the duration of tent caterpillar outbreak. Oecologia 93:25–30. https://doi.org/10.1021/ma071303h
Rydell J (1992) Exploitation of insects around streetlamps by bats in Sweden. Funct Ecol 6:744. https://doi.org/10.2307/2389972
Rydell J (2006) Bats and their insect prey at streetlights. In: Ecological Consequences of Artificial Night Lighting
Savilaakso S, Koivisto J, Veteli TO, Roininen H (2009) Microclimate and tree community linked to differences in lepidopteran larval communities between forest fragments and continuous forest. Divers Distrib 15:356–365. https://doi.org/10.1111/j.1472-4642.2008.00542.x
Schacht W, Witt T (1986) Warum nachtaktive Insekten künstliche Lichtquellen anfliegen (Insecta). Entomofauna 7:121–128
Seress G, Hammer T, Bókony V, Vincze E, Preiszner B, Pipoly I, Sinkovics C, Evans KL, Liker A (2018) Impact of urbanization on abundance and phenology of caterpillars and consequences for breeding in an insectivorous bird. Ecol Appl 28:1143–1156. https://doi.org/10.1002/eap.1730
Simon TJ, Bradstreet DC (1991) Comparative tolerability profile of omeprazole in clinical trials. Dig Dis Sci 36:1384–1389. https://doi.org/10.1007/BF01296803
Sower LL, Shorey HH, Gaston LK (1970) Sex pheromones of noctuid moths. XXI. Light: dark cycle regulation and light inhibition of sex pheromone release by females of Trichoplusia ni. Ann Entomol Soc Am 63:1090–1092. https://doi.org/10.1093/aesa/63.4.1090
Sullivan SMP, Hossler K, Meyer LA (2019) Artificial lighting at night alters aquatic-riparian invertebrate food webs. Ecol Appl 29. https://doi.org/10.1002/eap.1821
Tarlow EM, Hau M, Anderson DJ, Wikelski M (2003) Diel changes in plasma melatonin and corticosterone concentrations in tropical Nazca boobies (Sula granti) in relation to moon phase and age. Gen Comp Endocrinol 133:297–304. https://doi.org/10.1016/S0016-6480(03)00192-8
Tinbergen JM, Dietz MW (1994) Parental energy expenditure during brood rearing in the great tit (Parus major) in relation to body mass, temperature, food availability and clutch size. Funct Ecol 8:563. https://doi.org/10.2307/2389916
Troxel B, Piana M, Ashton MS, Murphy-Dunning C (2013) Relationships between bole and crown size for young urban trees in the northeastern USA. Urban For Urban Green 12:144–153. https://doi.org/10.1016/j.ufug.2013.02.006
Valtonen A, Hirka A, Szőcs L, Ayres MP, Roininen H, Csóka G (2017) Long-term species loss and homogenization of moth communities in Central Europe. J Anim Ecol 86:730–738. https://doi.org/10.1111/1365-2656.12687
Van Geffen KG, Groot AT, Van Grunsven RHA et al (2015) Artificial night lighting disrupts sex pheromone in a noctuid moth. Ecol Entomol 40:401–408. https://doi.org/10.1111/een.12202
Van Geffen KG, Van Grunsven RHA, Van Ruijven J et al (2014) Artificial light at night causes diapause inhibition and sex-specific life history changes in a moth. Ecol Evol 4:2082–2089. https://doi.org/10.1002/ece3.1090
Van Grunsven RHA, Lham D, Van Geffen KG, Veenendaa EM (2014) Range of attraction of a 6-W moth light trap. Entomol Exp Appl 152:87–90. https://doi.org/10.1111/eea.12196
van Langevelde F, Ettema JA, Donners M, WallisDeVries MF, Groenendijk D (2011) Effect of spectral composition of artificial light on the attraction of moths. Biol Conserv 144:2274–2281. https://doi.org/10.1016/j.biocon.2011.06.004
Wang YF (2008) Non-blocking extended OVSF codes on multi-rate CDMA systems. Comput Commun 31:35–48. https://doi.org/10.1016/j.comcom.2007.10.015
Welbers AAMH, van Dis NE, Kolvoort AM, Ouyang J, Visser ME, Spoelstra K, Dominoni DM (2017) Artificial light at night reduces daily energy expenditure in breeding great tits (Parus major). Front Ecol Evol 5:20170530. https://doi.org/10.3389/fevo.2017.00055
Woelfle MA, Ouyang Y, Phanvijhitsiri K, Johnson CH (2004) The adaptive value of circadian clocks: an experimental assessment in cyanobacteria. Curr Biol 14:1481–1486. https://doi.org/10.1016/j.cub.2004.08.023
Wolak M (2015) Facilitating estimation of the Intraclass correlation coefficient. ICC Packag 1–9
Acknowledgments
The study was financed by a grant from the National Research, Development and Innovation Office (NKFIH) of Hungary (K112838) and also supported by the 1783-3/2018/FEKUSTRAT grant of the Hungarian Ministry of Human Capacities. PÁ was supported by the Collegium Talentum 2019 Prgramme. GS was supported by an NKFIH postdoctoral grant (PD 120998) during the preparation of the manuscript. We are also thankful to Csenge Sinkovics, Dávid Németh, Eszter Sebestyén, Réka Somogyi and Tamás Hammer for their help with the caterpillar frass collection and processing. Finally, we thank the Associate Editor (Travis Longcore) and the two anonymous reviewers for their constructive and useful suggestions on the earlier version of the manuscript.
Funding
Open access funding provided by University of Pannonia (PE).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
All procedures were in accordance with Hungarian laws, licensed by the Middle Transdanubian Inspectorate for Environmental Protection, Natural Protection and Water Management (permission numbers: 31559/2011 and 24,861/2014).
Conflict of interest
We have no conflict of interest.
Human and animal studies
Our research did not involve any experiments on human participants or on animals.
Additional information
Áron Péter and Gábor Seress are joint first authors of this study
Electronic supplementary material
ESM 1
(DOCX 5471 kb)
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Péter, Á., Seress, G., Sándor, K. et al. The effect of artificial light at night on the biomass of caterpillars feeding in urban tree canopies. Urban Ecosyst 23, 1311–1319 (2020). https://doi.org/10.1007/s11252-020-00999-z
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11252-020-00999-z