Abstract
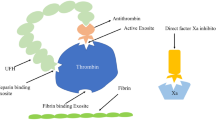
Anticoagulation during Impella® support is a challenge due to its complications and inconsistent practice across the globe. This observational, retrospective chart review included all patients with Impella® support at our advanced cardiac center at a quaternary care hospital in the Middle East gulf region. The study was conducted over six years (2016–2022), a time period during which manufacturer recommendations for purge solution, anticoagulation protocols as well as Impella® place in therapy and utilization were all evolving. We aimed to evaluate the efficacy of different anticoagulation practices and association with complications and outcomes. Forty-one patients underwent Impella® during the study period, including 25 patients with support for more than 12 h, and are the focus of our analysis. Cardiogenic shock (n = 25, 60.9%) was the primary indication for Impella®, followed by facilitating high-risk PCI (n = 15, 36.7%) and left ventricular afterload reduction in patients undergoing veno-arterial extracorporeal membrane oxygenation (n = 1, 2.4%). Our overall Impella® usage evolved over the years from a primary use to facilitate a high-risk PCI to the recent more common use of LV unloading in cardiogenic shock. No patients experienced device malfunction and the incidence of other complications including ischemic stroke and bleeding were comparable to those reported in the literature (12.2% and 24% respectively). The 30-day all-cause mortality of 41 patients was 53.6%. In line with the evolving recommendations and evidence, we observed an underutilization of non-heparin-based purge solutions and inconsistent management of anticoagulation in the setting of both Impella® and VA ECMO which necessitates more education and protocols.



Similar content being viewed by others
References
Nouri SN, Malick W, Masoumi A et al (2022) Impella® percutaneous left ventricular assist device as mechanical circulatory support for cardiogenic shock: a retrospective analysis from a tertiary academic medical center. Catheter Cardiovasc Interv 99(1):37–47
Werdan K, Gielen S, Ebelt H et al (2014) Mechanical circulatory support in cardiogenic shock. Eur Heart J 35:156–167
Seyfarth M, Sibbing D, Bauer I et al (2008) A randomized clinical trial to evaluate the safety and efficacy of a percutaneous left ventricular assist device versus intra-aortic balloon pumping for treatment of cardiogenic shock caused by myocardial infarction. J Am Coll Cardiol 52(19):1584–1588
Ouweneel DM, Eriksen E, Sjauw KD et al (2017) Percutaneous mechanical circulatory support versus intra-aortic balloon pump in cardiogenic shock after acute myocardial infarction. J Am Coll Cardiol 69(3):278–287
Schrage B, Ibrahim K, Loehn T et al (2019) Impella® support for acute myocardial infarction complicated by cardiogenic shock. Circulation 139(10):1249–1258
Weil BR, Konecny F, Suzuki G, Iyer V, Canty JM Jr (2016) Comparative hemodynamic effects of contemporary percutaneous mechanical circulatory support devices in a porcine model of acute myocardial infarction. JACC Cardiovasc Interv 9:2292–2303
Remmelink M, Sjauw KD, Henriques JP et al (2010) Effects of mechanical left ventricular unloading by Impella® on left ventricular dynamics in high-risk and primary percutaneous coronary intervention patients. Catheter Cardiovasc Interv 75:187–194
Abiomed. Reinforcement for Purge Management Recommendations. Impella® Update February 2017. Volume 2021. Abiomed; 2017. http://abiomed-private.s3.amazonaws.com/assets/files/14864898121bf5cc54722a9e59410f33c96eadb10a.pdf. Accessed July 19, 2022.
Vandenbriele C, Arachchillage DJ, Frederiks P et al (2022) Anticoagulation for percutaneous ventricular assist device-supported cardiogenic shock: JACC review topic of the week. J Am Coll Cardiol 79(19):1949–1962
Reed BN, DiDomenico RJ, Allender JE et al (2019) Survey of anticoagulation practices with the Impella® percutaneous ventricular assist device at high-volume centers. J Interv Cardiol 2019:3791307
Newsome AS, Taylor A, Garner S (2020) Anticoagulation of a percutaneous left ventricular assist Device. Using a low-dose heparin purge solution protocol: a case series. J Pharm Pract 33(4):471–476
Gilaman VDS, Popovsky M et al (2021) Bicarbonate as an alternative to heparin in Impella® purge fluid: understanding the biochemical basis. ASAIO J 67:101
Abiomed. 2017. 5% Dextrose with Heparin as Default Impella® Purge Solution. Impella® Update September 2015. Volume 2021: Abiomed. https://www.heartrecovery.com/education/education-library/faq-anticoagulation. Accessed July 19, 2022.
Abiomed. 2022 Heparin Free purge. Impella® Update May 2022. Volume 2022: Abiomed. https://www.heartrecovery.com/education/education-library/faq-anticoagulation. Accessed July 19, 2022.
Beavers C, Dunn S, DiDomenico R (2022) Bicarbonate-based purge solution during impella® support: a growing alternative. J Am Coll Cardiol 79(9 Supplement):633
Abiomed. 2020. Anticoagulation Therapy with Impella® Heparin Infusion. Impella® Update February 2020. Volume 2020: Abiomed. https://www.heartrecovery.com/resources/downloads/product-update-anti-coagulation-therapy-with-impella-heparin-infusion. Accessed July 19, 2022.
Fröhlich M, Lefering R, Probst C et al (2014) Epidemiology and risk factors of multiple-organ failure after multiple trauma. J Trauma Acute Care Surg 76:921–928
Peltan ID, Vande Vusse LK, Maier RV, Watkins TR (2015) An International normalized ratio-based definition of acute traumatic coagulopathy is associated with mortality, venous thromboembolism, and multiple organ failure after injury. Crit Care Med 43(7):1429–1438
Beavers CJ, DiDomenico RJ, Dunn SP et al (2021) Optimizing anticoagulation for patients receiving Impella® support. Pharmacotherapy 41(11):932–942
Mehran R, Rao SV, Bhatt DL et al (2011) Standardized bleeding definitions for cardiovascular clinical trials: a consensus report from the bleeding academic research consortium. Circulation 123(23):2736–2747
Abiomed. Impella® Ventricular Support Systems for Use during Cardiogenic Shock and High-Risk PCI: Impella® 2.5, Impella® 5.0, Impella® LD, and Impella® CP (Shock); Impella® 2.5 and Impella® CP (HRPCI) Instructions for Use & Clinical Reference Manual. Abiomed; 2020. https://www.heartrecovery.com/products-and-services/Impella®. Accessed July 19, 2022.
Lemaire A, Anderson MB, Lee LY et al (2014) The Impella® device for acute mechanical circulatory support in patients in cardiogenic shock. Ann Thorac Surg 97(1):133–138
Lauten A, Engstrom AE, Jung C et al (2013) Percutaneous left-ventricular support with the Impella®-2.5-assist device in acute cardiogenic shock: results of the Impella®-EUROSHOCK-Regis- try. Circ Heart Fail 6(1):23–30
Mastroianni C, Bouabdallaoui N, Leprince P et al (2017) Short-term mechanical circulatory support with the Impella® 5.0 device for cardiogenic shock at La Pitie-Salpetriere. Eur Heart J Acute Cardiovasc Care 6(1):87–92
Subramaniam AV, Barsness GW, Vallabhajosyula S et al (2019) Complications of temporary percutaneous mechanical circulatory support for cardiogenic shock: an appraisal of contemporary literature. Cardiol Ther 8:211–228
Ouweneel DM, de Brabander J, Karami M et al (2019) Real-life use of left ventricular circulatory support with Impella® in cardiogenic shock after acute myocardial infarction: 12 years AMC experience. Eur Heart J Acute Cardiovasc Care 8:338–349
Ali JM, Abu-Omar Y (2020) Complications associated with mechanical circulatory support. Ann Transl Med 8(13):835
Jennings DL, Nemerovski CW, Kalus JS (2013) Effective anticoagulation for a percutaneous ventricular assist device using a heparin-based purge solution. Ann Pharmacother 24(10):1364–1367
Haase M, Haase-Fielitz A, Plass M et al (2013) Prophylactic perioperative sodium bicarbonate to prevent acute kidney injury following open heart surgery: a multicentre double-blinded randomized controlled trial. PLoS Med 10(4):e1001426
Hemradj VV, Karami M, Sjauw KD et al (2020) Pre-PCI versus immediate post-PCI Impella initiation in acute myocardial infarction complicated by cardiogenic shock. PLoS ONE 5(7):e0235762
Helgestad OKL, Josiassen J, Hassager C et al (2020) Contemporary trends in use of mechanical circulatory support in patients with acute MI and cardiogenic shock. Open Heart 7(1):e001214
Omar HR, Mirsaeidi M, Socias S et al (2015) Plasma free hemoglobin is an independent predictor of mortality among patients on extracorporeal membrane oxygenation support. PLoS ONE 10(4):e0124034
O’Neill WW, Schreiber T, Wohns DH et al (2014) The current use of Impella® 2.5 in acute myocardial infarction complicated by cardiogenic shock: results from the USpella registry. J Interv Cardiol 27(1):1–11
Funding
The authors have no relevant financial or non-financial interests to disclose.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interest
The authors have no relevant financial or non-financial interests to disclose.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Hisham, M., Ghalib, H.H., Kakar, V. et al. Anticoagulation practices and complications associated with Impella® support at an advanced cardiac center in the Middle East gulf region. J Thromb Thrombolysis 56, 164–174 (2023). https://doi.org/10.1007/s11239-023-02807-9
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11239-023-02807-9