Abstract
The discovery of the engram, the physical substrate of memory, is a central challenge for the sciences of memory. Following the application of optogenetics to the neurobiological study of memory, scientists and philosophers claim that the engram has been found. In this paper, I evaluate the implications of applying optogenetic tools to the localization of the engram. I argue that conceptions of engram localization need to be revised to be made consistent with optogenetic studies of the engram. I distinguish between challenges to vehicle and content localization. First, I consider the silent engram hypothesis. According to this hypothesis, optogenetic studies indicate that synaptic efficacy, the traditional engram-bearing vehicle, is important merely for retrieval. I argue that this interpretation rests upon a misunderstanding of accessibility. Second, I argue that optogenetic-based strategies and findings conflict with preservationist and constructivist views on memory storage. There is an enduring trace, but stored content may change over time and experience, resulting in doubt about what constitutes a single engram.
Similar content being viewed by others
Notes
Lashley marks a skeptical point in the search after the engram, but several important historical landmarks have occurred since his time. For a more detailed account of the history see, e.g., Josselyn et al. (2017).
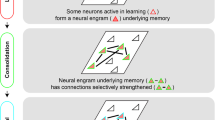
Neuroscientists refer to these criteria in various ways. Josselyn, Köhler and Frankland claim that the criteria for finding the engram include a persistence criterion—“a persistent change in the brain that results from a specific experience” and a content-bearing criterion—“the content of an engram reflects what transpired at encoding and predicts what can be recovered during subsequent retrieval” (2015, p. 521). I have called these criteria contiguity and similarity, respectively. Specificity, for example, is invoked by Inokuchi and colleagues who take it as a basic assumption that “memory is encoded in a specific cell ensemble that is activated during learning… individual memories are generally represented by different cell ensembles” (Yokose et al. 2017, p. 1). Philosophers apply roughly similar criteria, see e.g., De Brigard (2014a).
These criteria are meant to delineate the properties that researchers assume to characterize the engram. If vindicated, the characterization would provide evidence in favor of a certain descriptive account of the engram. These properties do not necessarily constitute the engram. The characterization of the phenomenon may still be revised in light of new or conflicting evidence (See, e.g., Feest, 2017; Colaço, 2020).
This advantage should not distract us from Jackie Sullivan’s important remark on the need to “recognize the value of methodological and perspectival pluralism for identifying the benefits and limitations of novel experimental techniques” (2018, p. 1100) when it comes to optogenetics. Optogenetics may have off-target effects, behavioral effects that occur due to the transient inhibition of a neural area and alternative tools may be used to circumvent such limitations.
While Abdou et al. (2018) is not the first study to show the application of optogenetics to the study of the engram (See, e.g., Robins 2018 for several examples), this study is notable for establishing the specificity of the engram and its intimate relation to synaptic efficacy which will be discussed in the next section.
Fear, in mice, is manifested in freezing behavior.
Such memories might be more accurately described as distorted rather than newly created memories. Nevertheless, the ability to induce such memories supports the idea that optogenetics is used to evaluate the similarity of a retrieved memory to the event encoded by altering the underlying vehicles, thus constituting a form of mimicking natural memory formation and retrieval.
An ensuing impression might be that the silent state of the engram is an artifact of using optogenetic tools. Although this is a possible interpretation, the fact that direct optogenetic activation of non-engram cells does not cause a similar behavioral expression tells against it.
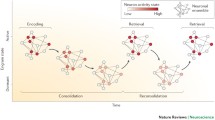
Nevertheless, reconsolidation does raise several difficulties regarding the stability of the engram’s content over time. I touch upon this issue in the next section.
Preservationists assume that a causal condition is necessary and sufficient for remembering but usually do not make a distinction between the way a memory is casually transmitted and the transmitted content.
The trace is the same entity persisting through time although its informational properties may vary.
One may object that a constructivist view is not committed to engrams being dispositional. An alternative option is to think of them as distributed neural connections where information is stored holistically. Accepting a distributed trace, however, assumes a form of Transmissibility, which constructivists deny. In the last part of this section, I consider a hypothetical experiment whose results constructivists could use to answer this challenge without accepting Transmissibility. I thank an anonymous reviewer for raising this point.
References
Abdou, K., Shehata, M., Choko, K., Nishizono, H., Matsuo, M., Muramatsu, S. I., & Inokuchi, K. (2018). Synapse-specific representation of the identity of overlapping memory engrams. Science, 360, 1227–1231.
Bechtel, W. (2008). Mental mechanisms: Philosophical perspectives on cognitive neuroscience. Routledge.
Colaço, D. (2020). Recharacterizing scientific phenomena. European Journal for Philosophy of Science, 10, 1–19.
Cooper, J. M. (Ed.). (1997). Plato: Complete works. Hackett.
De Brigard, F. (2014a). The nature of memory traces. Philosophy Compass, 9, 402–414.
De Brigard, F. (2014b). Is memory for remembering? Recollection as a form of episodic hypothetical thinking. Synthese, 191, 155–185.
De Brigard, F. (2017). Cognitive systems and the changing brain. Philosophical Explorations, 20, 224–241.
De Sousa, A. F., Cowansage, K. K., Zutshi, I., Cardozo, L. M., Yoo, E. J., Leutgeb, S., & Mayford, M. (2019). Optogenetic reactivation of memory ensembles in the retrosplenial cortex induces systems consolidation. Proceedings of the National Academy of Sciences, 116, 8576–8581.
Feest, U. (2017). Phenomena and objects of research in the cognitive and behavioral sciences. Philosophy of Science, 84, 1165–1176.
Hardt, O., & Nadel, L. (2018). Systems consolidation revisited, but not revised: The promise and limits of optogenetics in the study of memory. Neuroscience Letters, 680, 54–59.
Henke, K. (2010). A model for memory systems based on processing modes rather than consciousness. Nature Reviews Neuroscience, 11, 523.
Josselyn, S. A., Köhler, S., & Frankland, P. W. (2015). Finding the engram. Nature Reviews Neuroscience, 16, 521.
Josselyn, S. A., Köhler, S., & Frankland, P. W. (2017). Heroes of the engram. Journal of Neuroscience, 37, 4647–4657.
Josselyn, S. A., & Tonegawa, S. (2020). Memory engrams: Recalling the past and imagining the future. Science, 367, 6473.
Lashley, K. S. (1950). In search of the engram. In Society for experimental biology No. 4: Physiological mechanisms in animal behavior (pp. 454–482). Cambridge University Press.
Lux, V., Masseck, O. A., Herlitze, S., & Sauvage, M. M. (2017). Optogenetic destabilization of the memory trace in CA1: Insights into reconsolidation and retrieval processes. Cerebral Cortex, 27, 841–851.
Martin, S. J., Grimwood, P. D., & Morris, R. G. (2000). Synaptic plasticity and memory: An evaluation of the hypothesis. Annual Review of Neuroscience, 23, 649–711.
McClelland, J. L., McNaughton, B. L., & O’Reilly, R. C. (1995). Why there are complementary learning systems in the hippocampus and neocortex: Insights from the successes and failures of connectionist models of learning and memory. Psychological Review, 102, 419–457.
McKenzie, S., & Eichenbaum, H. (2011). Consolidation and reconsolidation: Two lives of memories? Neuron, 71, 224–233.
Michaelian, K., & Robins, S. (2018). Beyond the causal theory? Fifty years after Martin and Deutscher. In K. Michaelian, D. Debus, & D. Perrin (Eds.), New directions in the philosophy of memory (pp. 13–32). Routledge.
Nadel, L., Winocur, G., Ryan, L., & Moscovitch, M. (2007). Systems consolidation and hippocampus: Two views. Debates in Neuroscience, 1, 55–66.
Ramirez, S., Liu, X., Lin, P. A., Suh, J., Pignatelli, M., Redondo, R., Ryan, T., & Tonegawa, S. (2013). Creating a false memory in the hippocampus. Science, 341, 387–391.
Robins, S. K. (2016). Optogenetics and the mechanism of false memory. Synthese, 193, 1561–1583.
Robins, S. K. (2018). Memory and optogenetic intervention: Separating the engram from the ecphory. Philosophy of Science, 85, 1078–1089.
Robins, S. K. (2020). Stable engrams and neural dynamics. Philosophy of Science, 87, 1130–1139.
Roy, D. S., Muralidhar, S., Smith, L. M., & Tonegawa, S. (2017). Silent memory engrams as the basis for retrograde amnesia. Proceedings of the National Academy of Sciences, 114, 9972–9979.
Ryan, T. J., Roy, D. S., Pignatelli, M., Arons, A., & Tonegawa, S. (2015). Engram cells retain memory under retrograde amnesia. Science, 348, 1007–1013.
Semon, R. (1921). The Mneme. George Allen & Unwin.
Silva, A. J., Landreth, A., & Bickle, J. (2013). Engineering the next revolution in neuroscience: The new science of experiment planning. Oxford University Press.
Sullivan, J. A. (2018). Optogenetics, pluralism, and progress. Philosophy of Science, 85, 1090–1101.
Tonegawa, S., Liu, X., Ramirez, S., & Redondo, R. (2015). Memory engram cells have come of age. Neuron, 87, 918–931.
Tulving, E., & Pearlstone, Z. (1966). Availability versus accessibility of information in memory for words. Journal of Verbal Learning and Verbal Behavior, 5, 381–391.
Winocur, G., Moscovitch, M., & Sekeres, M. (2007). Memory consolidation or transformation: Context manipulation and hippocampal representations of memory. Nature Neuroscience, 10, 555–557.
Winters, B. D., Tucci, M. C., Jacklin, D. L., Reid, J. M., & Newsome, J. (2011). On the dynamic nature of the engram: Evidence for circuit-level reorganization of object memory traces following reactivation. Journal of Neuroscience, 31, 17719–17728.
Yokose, J., Okubo-Suzuki, R., Nomoto, M., Ohkawa, N., Nishizono, H., Suzuki, A., Matsuo, M., Tsujimura, S., Takahashi, Y., Nagase, M., Watabe, A. M., Sasahara, M., Kato, F., & Inokuchi, K. (2017). Overlapping memory trace indispensable for linking, but not recalling, individual memories. Science, 355, 398–403.
Acknowledgements
I would like to thank Colin Allen, Aya Evron, J.P. Gamboa, Topaz Halperin, Arnon Levy, Sarah Robins, Oron Shagrir, Filippo Vindrola, and three anonymous reviewers for their thoughtful comments and suggestions on this article. This work was supported by the Interuniversity Ph.D. Program in the History and Philosophy of the Life Sciences, supported by the Humanities Fund of the Israeli Council of Higher Education; The Sidney M. Edelstein Center for History and Philosophy of Technology and Medicine at the Hebrew University of Jerusalem; The Jack, Joseph and Morton Mandel School for Advanced Studies in the Humanities at the Hebrew University of Jerusalem.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This article belongs to the topical collection “Interdisciplinary Perspectives on Locating Representations in the Brain”, edited by Jessey Wright and Sarah Robins.
Rights and permissions
About this article
Cite this article
Najenson, J. What have we learned about the engram?. Synthese 199, 9581–9601 (2021). https://doi.org/10.1007/s11229-021-03216-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11229-021-03216-2