Abstract
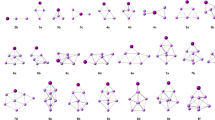
The possibility of formation of various ion clusters for lithium salts LiXF6 (X = As, P) is studied. The dynamic matrix of the clusters in a gas phase is calculated by numerical and analytical differentiation of the full energy of clusters in the MO LCAO approximation by the Hartree-Fock-Roothaan (HFR) method with the aid of program package PC GAMESS. Stable ionic clusters are ion pairs Li+[XF6]− with bi- and tridentate cation coordination relative to the octahedral anion, ion triplets [XF6]−Li+[XF6]− and Li+[XF6]−Li+ with bi- and tridentate coordination, and ion dimers { Li+[XF6]−}2 with bidentate coordination. Trimers {Li+[XF6]−}3 and tetramers {Li+[XF6]−}4 in the form of symmetrical ring structures with monodentate coordination are stable only for [AsF6]−. For stable ion species, densities of vibrational states and IR spectra are calculated.
Similar content being viewed by others
REFERENCES
Lithium Battery Technology, Venkatasetty, H.V., Ed., New York: Wiley, 1984.
Aurbach, D., J. Power Sourses, 2003, vol. 119–121, p. 497.
Deng, Z. and Irish, D.E., Can. J. Chem., 1991, vol. 69, p. 1766.
Deng, Z. and Irish, D.E., J. Chem. Soc., Faraday Trans., 1992, vol. 88, no.19, p. 2891.
Eschmann, J., Strasser, J., Xu, M., Okamoto, Y., Eyring, E.M., and Petrucci, S., J. Phys. Chem., 1990, vol. 94, p. 3908.
Bushkova, O.V., Lirova, B.I., and Zhukovskii, V.M., Zh. Fiz. Khim., 1999, vol. 73, p. 640.
Perelygin, I.S. and Klimchuk, M.A., Koord. Khim., 1990, vol. 16, p. 1042.
Laik, B., Legrand, L., Chausse, A., and Messina, R., Electrochim. Acta, 1998, vol. 44, p. 773.
Zhukovskii, V.M., Bushkova, O.V., Lirova, B.I., and Kruglyashov, A.L., Elektrokhim. Energ., 2001, vol. 1, no.1–2, p. 43.
Bushkova, O.V., Lirova, B.I., and Zhukovskii, V.M., Elektrokhimiya, 2003, vol. 39, p. 578.
Inoue, N., Xu, M., and Petrucci, S., J. Phys. Chem., 1987, vol. 91, p. 4628.
Perelygin, I.S., Shatokhin, S.A., and Kononenko, A.B., Zh. Strukt. Khim., 1992, vol. 33, p. 163.
Perelygin, I.S., in Ionnaya sol’vatatsiya (Ionic Solvation), Krestov, G.A., Ed., Moscow: Nauka, 1987, p. 100.
Begun, G.M. and Rutenberg, A.C., Inorg. Chem., 1967, vol. 6, p. 2212.
Granovsky, A.A., http://classic.chem.msu.su/gran/gamess/index.html.
Schmidt, M.W., Baldridge, K.K., Boatz, J.A., Elbert, S.T., Gordon, M.S., Jensen, J.J., Koseki, S., Matsunada, N., Nguyen, K.A., Su, S., Windus, T.L., Dupuis, M., and Montgomery, J.A., J. Comput. Chem., 1993, vol. 14, p. 1347.
Roothaan, C.C.J., Rev. Mod. Phys., 1951, vol. 23, p. 69.
Curtiss, L.A., McGrath, M.P., Blandeau, J.-P., Davis, N.E., Binning, R.C., and Radom, J.L., J. Chem. Phys., 1995, vol. 103, p. 6104.
Dunning, T.H., J. Chem. Phys., 1989, vol. 90, p. 1007.
McLean, A.D. and Chandler, G.S., J. Chem. Phys., 1980, vol. 72, p. 5639.
Author information
Authors and Affiliations
Corresponding author
Additional information
__________
Translated from Elektrokhimiya, Vol. 41, No. 5, 2005, pp. 546–555.
Original Russian Text Copyright © 2005 by Popov, Nikiforov, Bushkova, Zhukovskii.
Rights and permissions
About this article
Cite this article
Popov, S.E., Nikiforov, A.E., Bushkova, O.V. et al. Quantum-Chemical Investigation of Ionic Association of Lithium Salts LiXF6 (X = As, P). Russ J Electrochem 41, 476–484 (2005). https://doi.org/10.1007/s11175-005-0093-0
Received:
Issue Date:
DOI: https://doi.org/10.1007/s11175-005-0093-0