Abstract
Introduction
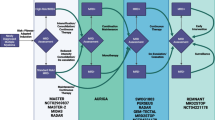
Beyond focal radiation, there is no consensus standard therapy for pediatric high-grade glioma (pHGG) and outcomes remain dismal. We describe the largest molecularly-characterized cohort of children with pHGG treated with a 3-drug maintenance regimen of temozolomide, irinotecan, and bevacizumab (TIB) following radiation.
Methods
We retrospectively reviewed 36 pediatric patients treated with TIB at Seattle Children’s Hospital from 2009 to 2018 and analyzed survival using the Kaplan–Meier method. Molecular profiling was performed by targeted DNA sequencing and toxicities, steroid use, and palliative care utilization were evaluated.
Results
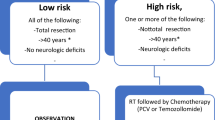
Median age at diagnosis was 10.9 years (18 months–18 years). Genetic alterations were detected in 26 genes and aligned with recognized molecular subgroups including H3 K27M-mutant (12), H3F3A G34-mutant (2), IDH-mutant (4), and hypermutator profiles (4). Fifteen patients (42%) completed 12 planned cycles of maintenance. Side effects associated with chemotherapy delays or modifications included thrombocytopenia (28%) and nausea/vomiting (19%), with temozolomide dosing most frequently modified. Median event-free survival (EFS) and overall survival (OS) was 16.2 and 20.1 months, with shorter survival seen in DIPG (9.3 and 13.3 months, respectively). Survival at 1, 2, and 5 years was 80%, 10% and 0% for DIPG and 85%, 38%, and 16% for other pHGG.
Conclusion
Our single-center experience demonstrates tolerability of this 3-drug regimen, with prolonged survival in DIPG compared to historical single-agent temozolomide. pHGG survival was comparable to analogous 3-drug regimens and superior to historical agents; however, cure was rare. Children with pHGG remain excellent candidates for the study of novel therapeutics combined with standard therapy.



Similar content being viewed by others
Data availability
All data generated or analysed during this study are included in this published article [and its supplementary information files].
References
Mackay A, Burford A, Carvalho D, Izquierdo E, Fazal-Salom J, Taylor KR, Bjerke L, Clarke M, Vinci M, Nandhabalan M, Temelso S, Popov S, Molinari V, Raman P, Waanders AJ, Han HJ, Gupta S, Marshall L, Zacharoulis S, Vaidya S, Mandeville HC, Bridges LR, Martin AJ, Al-Sarraj S, Chandler C, Ng H-K, Li X, Mu K, Trabelsi S, Brahim DHM-B, Kisljakov AN, Konovalov DM, Moore AS, Carcaboso AM, Sunol M, De Torres C, Cruz O, Mora J, Shats LI, Stavale JN, Bidinotto LT, Reis RM, Entz-Werle N, Farrell M, Cryan J, Crimmins D, Caird J, Pears J, Monje M, Debily M-A, Castel D, Grill J, Hawkins C, Nikbakht H, Jabado N, Baker SJ, Pfister SM, Jones DTW, Fouladi M, Von Bueren AO, Baudis M, Resnick A, Jones C (2017) Integrated molecular meta-analysis of 1,000 pediatric high-grade and diffuse intrinsic pontine glioma. Cancer Cell 32(4):520–537.e525. https://doi.org/10.1016/j.ccell.2017.08.017
Cooney T, Lane A, Bartels U, Bouffet E, Goldman S, Leary SES, Foreman NK, Packer RJ, Broniscer A, Minturn JE, Shih C-S, Chintagumpala M, Hassall T, Gottardo NG, Dholaria H, Hoffman L, Chaney B, Baugh J, Doughman R, Leach JL, Jones BV, Fouladi M, Warren KE, Monje M (2017) Contemporary survival endpoints: an International Diffuse Intrinsic Pontine Glioma Registry study. Neuro-Oncology 19(9):1279–1280. https://doi.org/10.1093/neuonc/nox107
Louis DN, Perry A, Reifenberger G, Von Deimling A, Figarella-Branger D, Cavenee WK, Ohgaki H, Wiestler OD, Kleihues P, Ellison DW (2016) The 2016 World Health Organization Classification of Tumors of the Central Nervous System: a summary. Acta Neuropathol 131(6):803–820. https://doi.org/10.1007/s00401-016-1545-1
Ostrom QT, Gittleman H, Truitt G, Boscia A, Kruchko C, Barnholtz-Sloan JS (2018) CBTRUS Statistical report: primary brain and other central nervous system tumors diagnosed in the United States in 2011–2015. Neuro-Oncology 20(suppl_4):iv1–iv86. https://doi.org/10.1093/neuonc/noy131
Janssens GO, Gandola L, Bolle S, Mandeville H, Ramos-Albiac M, Van Beek K, Benghiat H, Hoeben B, Morales La Madrid A, Kortmann R-D, Hargrave D, Menten J, Pecori E, Biassoni V, Von Bueren AO, Van Vuurden DG, Massimino M, Sturm D, Peters M, Kramm CM (2017) Survival benefit for patients with diffuse intrinsic pontine glioma (DIPG) undergoing re-irradiation at first progression: a matched-cohort analysis on behalf of the SIOP-E-HGG/DIPG working group. Eur J Cancer 73:38–47. https://doi.org/10.1016/j.ejca.2016.12.007
Lu VM, Welby JP, Mahajan A, Laack NN, Daniels DJ (2019) Reirradiation for diffuse intrinsic pontine glioma: a systematic review and meta-analysis. Child's Nerv Syst 35(5):739–746. https://doi.org/10.1007/s00381-019-04118-y
Rao AD, Rashid AS, Chen Q, Villar RC, Kobyzeva D, Nilsson K, Dieckmann K, Nechesnyuk A, Ermoian R, Alcorn S, MacDonald SM, Ladra MM, Ford EC, Winey BA, Figueiredo MLS, Chen MJ, Terezakis SA (2017) Reirradiation for recurrent pediatric central nervous system malignancies: a multi-institutional review. Int J Radiat Oncol Biol Phys 99(3):634–641. https://doi.org/10.1016/j.ijrobp.2017.07.026
Tsang DS, Oliveira C, Bouffet E, Hawkins C, Ramaswamy V, Yee R, Tabori U, Bartels U, Huang A, Millar BA, Crooks B, Bowes L, Zelcer S, Laperriere N (2019) Repeat irradiation for children with supratentorial high-grade glioma. Pediatr Blood Cancer. https://doi.org/10.1002/pbc.27881
Bernier-Chastagner V, Grill J, Doz F, Bracard S, Gentet JC, Marie-Cardine A, Luporsi E, Margueritte G, Lejars O, Laithier V, Mechinaud F, Millot F, Kalifa C, Chastagner P (2005) Topotecan as a radiosensitizer in the treatment of children with malignant diffuse brainstem gliomas. Cancer 104(12):2792–2797. https://doi.org/10.1002/cncr.21534
Broniscer A, Baker SJ, Stewart CF, Merchant TE, Laningham FH, Schaiquevich P, Kocak M, Morris EB, Endersby R, Ellison DW, Gajjar A (2009) Phase I and pharmacokinetic studies of erlotinib administered concurrently with radiotherapy for children, adolescents, and young adults with high-grade glioma. Clin Cancer Res 15(2):701–707. https://doi.org/10.1158/1078-0432.ccr-08-1923
Chassot A, Canale S, Varlet P, Puget S, Roujeau T, Negretti L, Dhermain F, Rialland X, Raquin MA, Grill J, Dufour C (2012) Radiotherapy with concurrent and adjuvant temozolomide in children with newly diagnosed diffuse intrinsic pontine glioma. J Neurooncol 106(2):399–407. https://doi.org/10.1007/s11060-011-0681-7
Fouladi M, Nicholson HS, Zhou T, Laningham F, Helton KJ, Holmes E, Cohen K, Speights RA, Wright J, Pollack IF (2007) A phase II study of the farnesyl transferase inhibitor, tipifarnib, in children with recurrent or progressive high-grade glioma, medulloblastoma/primitive neuroectodermal tumor, or brainstem glioma: a children's oncology group study. Cancer 110(11):2535–2541. https://doi.org/10.1002/cncr.23078
Kilburn LB, Kocak M, Schaedeli Stark F, Meneses-Lorente G, Brownstein C, Hussain S, Chintagumpala M, Thompson PA, Gururangan S, Banerjee A, Paulino AC, Kun L, Boyett JM, Blaney SM (2013) Phase I trial of capecitabine rapidly disintegrating tablets and concomitant radiation therapy in children with newly diagnosed brainstem gliomas and high-grade gliomas. Neuro-Oncology 15(6):759–766. https://doi.org/10.1093/neuonc/nos315
Korones DN, Fisher PG, Kretschmar C, Zhou T, Chen Z, Kepner J, Freeman C (2008) Treatment of children with diffuse intrinsic brain stem glioma with radiotherapy, vincristine and oral VP-16: a Children's Oncology Group phase II study. Pediatr Blood Cancer 50(2):227–230. https://doi.org/10.1002/pbc.21154
Pollack IF, Jakacki RI, Blaney SM, Hancock ML, Kieran MW, Phillips P, Kun LE, Friedman H, Packer R, Banerjee A, Geyer JR, Goldman S, Poussaint TY, Krasin MJ, Wang Y, Hayes M, Murgo A, Weiner S, Boyett JM (2007) Phase I trial of imatinib in children with newly diagnosed brainstem and recurrent malignant gliomas: a Pediatric Brain Tumor Consortium report1. Neuro-Oncology 9(2):145–160. https://doi.org/10.1215/15228517-2006-031
Turner CD, Chi S, Marcus KJ, Macdonald T, Packer RJ, Poussaint TY, Vajapeyam S, Ullrich N, Goumnerova LC, Scott RM, Briody C, Chordas C, Zimmerman MA, Kieran MW (2007) Phase II study of thalidomide and radiation in children with newly diagnosed brain stem gliomas and glioblastoma multiforme. J Neurooncol 82(1):95–101. https://doi.org/10.1007/s11060-006-9251-9
Cohen BH, Geyer JR, Miller DC, Curran JG, Zhou T, Holmes E, Ingles SA, Dunkel IJ, Hilden J, Packer RJ, Pollack IF, Gajjar A, Finlay JL (2015) Pilot study of intensive chemotherapy with peripheral hematopoietic cell support for children less than 3 years of age with malignant brain tumors, the CCG-99703 phase I/II study. A report from the Children's Oncology Group. Pediatr Neurol 53(1):31–46. https://doi.org/10.1016/j.pediatrneurol.2015.03.019
Duffner P, Krischer J, Burger P, Cohen M, Backstrom J, Horowitz M, Sanford R, Friedman H, Kun L (1996) Treatment of infants with malignant gliomas: the Pediatric Oncology Group experience. J Neuro-Oncol. https://doi.org/10.1007/bf00250203
Cohen KJ, Heideman RL, Zhou T, Holmes EJ, Lavey RS, Bouffet E, Pollack IF (2011) Temozolomide in the treatment of children with newly diagnosed diffuse intrinsic pontine gliomas: a report from the Children's Oncology Group. Neuro-Oncology 13(4):410–416. https://doi.org/10.1093/neuonc/noq205
Cohen KJ, Pollack IF, Zhou T, Buxton A, Holmes EJ, Burger PC, Brat DJ, Rosenblum MK, Hamilton RL, Lavey RS, Heideman RL (2011) Temozolomide in the treatment of high-grade gliomas in children: a report from the Children's Oncology Group. Neuro-Oncology 13(3):317–323. https://doi.org/10.1093/neuonc/noq191
Jonathan L, Finlay JMB, Yates AJ, Wisoff JH, Milstein JM, Russell Geyer J, Bertolone SJ, McGuire P, Cherlow JM, Tefft M, Turski PA, Wara WM, Edwards M, Sutton LN, Berger MS, Epstein F, Ayers G, Allen JC, Packer RJ (1995) Randomized phase iii trial in childhood high-grade astrocytoma comparing vincristine, lomustine, and prednisone with the eight-drugs-in-l-day regimen. J Clin Oncol. https://doi.org/10.1200/jco.1995.13.1.112
Cohen MH, Shen YL, Keegan P, Pazdur R (2009) FDA drug approval summary: bevacizumab (Avastin(R)) as treatment of recurrent glioblastoma multiforme. Oncologist 14(11):1131–1138. https://doi.org/10.1634/theoncologist.2009-0121
Parekh C, Jubran R, Erdreich-Epstein A, Panigrahy A, Bluml S, Finlay J, Dhall G (2011) Treatment of children with recurrent high grade gliomas with a bevacizumab containing regimen. J Neurooncol 103(3):673–680. https://doi.org/10.1007/s11060-010-0444-x
Houghton PJ, Stewart CF, Cheshire PJ, Richmond LB, Kirstein MN, Poquette CA, Tan M, Friedman HS, Brent TP (2000) Antitumor activity of temozolomide combined with irinotecan is partly independent of O6-methylguanine-DNA methyltransferase and mismatch repair phenotypes in xenograft models. Clin Cancer Res 6(10):4110–4118
Pritchard CC, Salipante SJ, Koehler K, Smith C, Scroggins S, Wood B, Wu D, Lee MK, Dintzis S, Adey A, Liu Y, Eaton KD, Martins R, Stricker K, Margolin KA, Hoffman N, Churpek JE, Tait JF, King M-C, Walsh T (2014) Validation and implementation of targeted capture and sequencing for the detection of actionable mutation, copy number variation, and gene rearrangement in clinical cancer specimens. J Mol Diagn 16(1):56–67. https://doi.org/10.1016/j.jmoldx.2013.08.004
Kuo AJ, Paulson VA, Hempelmann JA, Beightol M, Todhunter S, Colbert BG, Salipante SJ, Konnick EQ, Pritchard CC, Lockwood CM (2020) Validation and implementation of a modular targeted capture assay for the detection of clinically significant molecular oncology alterations. Pract Lab Med 19:e00153. https://doi.org/10.1016/j.plabm.2020.e00153
Hoeman CM, Cordero FJ, Hu G, Misuraca K, Romero MM, Cardona HJ, Nazarian J, Hashizume R, McLendon R, Yu P, Procissi D, Gadd S, Becher OJ (2019) ACVR1 R206H cooperates with H3.1K27M in promoting diffuse intrinsic pontine glioma pathogenesis. Nat Commun. https://doi.org/10.1038/s41467-019-08823-9
Lu G, Rao M, Zhu P, Liang B, El-Nazer RT, Fonkem E, Bhattacharjee MB, Zhu JJ (2019) Triple-drug therapy with bevacizumab, irinotecan, and temozolomide plus tumor treating fields for recurrent glioblastoma: a retrospective study. Front Neurol 10:42. https://doi.org/10.3389/fneur.2019.00042
Hummel TR, Salloum R, Drissi R, Kumar S, Sobo M, Goldman S, Pai A, Leach J, Lane A, Pruitt D, Sutton M, Chow LM, Grimme L, Doughman R, Backus L, Miles L, Stevenson C, Fouladi M, Dewire M (2016) A pilot study of bevacizumab-based therapy in patients with newly diagnosed high-grade gliomas and diffuse intrinsic pontine gliomas. J Neurooncol 127(1):53–61. https://doi.org/10.1007/s11060-015-2008-6
Zaky W, Wellner M, Brown RJ, Bluml S, Finlay JL, Dhall G (2013) Treatment of children with diffuse intrinsic pontine gliomas with chemoradiotherapy followed by a combination of temozolomide, irinotecan, and bevacizumab. Pediatr Hematol Oncol 30(7):623–632. https://doi.org/10.3109/08880018.2013.829895
Grill J, Massimino M, Bouffet E, Azizi AA, McCowage G, Cañete A, Saran F, Le Deley M-C, Varlet P, Morgan PS, Jaspan T, Jones C, Giangaspero F, Smith H, Garcia J, Elze MC, Rousseau RF, Abrey L, Hargrave D, Vassal G (2018) Phase II, open-label, randomized, multicenter trial (HERBY) of bevacizumab in pediatric patients with newly diagnosed high-grade glioma. J Clin Oncol 36(10):951–958. https://doi.org/10.1200/jco.2017.76.0611
Yang T, Temkin N, Barber J, Geyer JR, Leary S, Browd S, Ojemann JG, Ellenbogen RG (2013) Gross total resection correlates with long-term survival in pediatric patients with glioblastoma. World Neurosurg 79(3–4):537–544. https://doi.org/10.1016/j.wneu.2012.09.015
Mackay A, Burford A, Molinari V, Jones DTW, Izquierdo E, Brouwer-Visser J, Giangaspero F, Haberler C, Pietsch T, Jacques TS, Figarella-Branger D, Rodriguez D, Morgan PS, Raman P, Waanders AJ, Resnick AC, Massimino M, Garrè ML, Smith H, Capper D, Pfister SM, Würdinger T, Tam R, Garcia J, Thakur MD, Vassal G, Grill J, Jaspan T, Varlet P, Jones C (2018) Molecular, pathological, radiological, and immune profiling of non-brainstem pediatric high-grade glioma from the HERBY phase II randomized trial. Cancer Cell 33(5):829–842.e825. https://doi.org/10.1016/j.ccell.2018.04.004
Salloum R, Dewire M, Lane A, Goldman S, Hummel T, Chow L, Miles L, Sutton M, Stevenson C, Fouladi M, Leach J (2015) Patterns of progression in pediatric patients with high-grade glioma or diffuse intrinsic pontine glioma treated with bevacizumab-based therapy at diagnosis. J Neurooncol 121(3):591–598. https://doi.org/10.1007/s11060-014-1671-3
Pound CM, Clark C, Ni A, Athale U, Lewis V, Halton JM (2012) Corticosteroids, behavior, and quality of life in children treated for acute lymphoblastic leukemia; a multicentered trial. J Pediatr Hematol Oncol 34(7):517–523. https://doi.org/10.1097/MPH.0b013e318257fdac
Dietrich J, Rao K, Pastorino S, Kesari S (2011) Corticosteroids in brain cancer patients: benefits and pitfalls. Expert Rev Clin Pharmacol 4(2):233–242. https://doi.org/10.1586/ecp.11.1
Pitter KL, Tamagno I, Alikhanyan K, Hosni-Ahmed A, Pattwell SS, Donnola S, Dai C, Ozawa T, Chang M, Chan TA, Beal K, Bishop AJ, Barker CA, Jones TS, Hentschel B, Gorlia T, Schlegel U, Stupp R, Weller M, Holland EC, Hambardzumyan D (2016) Corticosteroids compromise survival in glioblastoma. Brain 139(5):1458–1471. https://doi.org/10.1093/brain/aww046
Hue CD, Cho FS, Cao S, Bass CRD, Meaney DF, Morrison B (2015) Dexamethasone potentiates in vitro blood-brain barrier recovery after primary blast injury by glucocorticoid receptor-mediated upregulation of ZO-1 tight junction protein. J Cereb Blood Flow Metab 35(7):1191–1198. https://doi.org/10.1038/jcbfm.2015.38
Acknowledgements
We thank the many children and families who struggled through these impossibly difficult diseases and entrusted us with their care. We thank our entire pediatric neuro-oncology team, especially C. Hoeppner, S. Holtzclaw, I. Lam, M. Field, S. Chaffee, A. Laurine, S. Stasi, R. Miller, E. Stowe, G. Mun, and W. Iwata for their dedication and compassion. We thank our child life specialists, especially A. Sevilla and Erin Behem. We thank the radiation oncology and palliative care teams, especially R. Hays and A. Towbridge, who work tirelessly to improve the lives of our patients. We thank J. Stevens, as well as the Seattle Children’s Hospital’s Department of Anatomic Pathology and TTS Brain Tumor Committee, for assistance in tissue collection and research coordination.
Funding
We are grateful for generous funding from the Seattle Run of Hope (S.E.S.L., J.M.O., N.A.V), the Pediatric Brain Tumor Research Fund Guild of Seattle Children’s Hospital (S.E.S.L., J.M.O, N.A.V.), the McKenna Claire Foundation (N.A.V.), Unravel Pediatric Cancer (N.A.V., J.M.O.), Team Cozzi Foundation (N.A.V.), the Julianna Sayler Foundation (N.A.V.), the Grousemont Foundation (N.A.V.), the Michael Mosier Defeat DIPG Foundation (N.A.V.), and the ChadTough Foundation (N.A.V.).
Author information
Authors and Affiliations
Contributions
Conception and design: EC, SL, and NV. Data collection and assembly: EC, BC, CL, and VP. Data analyses and interpretation: all authors with survival analyses by SL, molecular analyses by CL and VP, and pathologic analyses by BC. Manuscript writing and editing: EC and NV with critical feedback from all authors. Final approval of manuscript: All authors. Accountable for all aspects of the work: All authors.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no competing interests.
Ethical approval
This retrospective chart review study involving human participants was in accordance with the ethical standards of our institutional and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards. The Institutional Review Board of Seattle Children’s Hospital approved this study (IRB#14449).
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Crotty, E.E., Leary, S.E.S., Geyer, J.R. et al. Children with DIPG and high-grade glioma treated with temozolomide, irinotecan, and bevacizumab: the Seattle Children’s Hospital experience. J Neurooncol 148, 607–617 (2020). https://doi.org/10.1007/s11060-020-03558-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11060-020-03558-w