Abstract
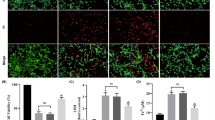
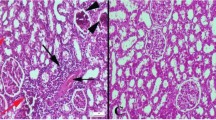
Neurotoxicity is implicated as a severe complication of chronic kidney disease (CKD). Accumulation of urea and other toxic compounds leads to oxidative stress, inflammation and destruction of the blood-brain barrier. Carbon monoxide (CO) and hydrogen sulfide (H2S) have been shown to have anti-inflammatory, anti-apoptotic, and anti-proliferative properties. The aims of the present study were evaluated the protective effects of CO-releasing molecule (CORM3) and H2S donor (NaHS) on oxidative stress and neuronal death induced by CKD in the hippocampus and prefrontal cortex by considering interaction between CO and H2S on CBS expression. CORM3 or NaHS significantly compensated deficits in the antioxidant defense mechanisms, suppressed lipid peroxidation and reduced neuronal death in hippocampus and prefrontal cortex and improvement the markers of renal injury that induced by CKD. In addition, CORM3 or NaHS significantly improved CBS expression which were reduced by CKD. However, improving effects of CORM3 on antioxidant defense mechanisms, lipid peroxidation, neuronal death, renal injury and CBS expression were prevented by amino-oxy acetic acid (AOAA) (CBS inhibitor) and reciprocally improving effects of NaHS on all above indices were prevented by zinc protoporphyrin IX (Znpp) (HO-1 inhibitor). In conclusion, this study demonstrated that formation of CO and H2S were interdependently improved CKD-induced oxidative stress and neuronal death, which is may be through increased expression of CBS.





Similar content being viewed by others
Code availability
Not applicable.
Abbreviations
- AOAA:
-
Amino-oxy acetic acid
- ANOVA:
-
One-way analysis
- BUN:
-
blood urea nitrogen
- CBS:
-
Cystathionine-β-synthase
- CKD:
-
Chronic kidney disease
- CO:
-
Carbon monoxide
- CORM3:
-
CO-releasing molecule
- CNS:
-
Central nervous system
- DG:
-
Dentate gyrus
- GSH:
-
Glutathione peroxidase
- HFD:
-
High-fat diet
- H&E:
-
Hematoxylin and Eosin
- HI:
-
hypoxic-ischemic
- HO-1:
-
Hemeoxygenase-1
- H2S:
-
Hydrogen sulfide
- HTN:
-
Hypertension
- PE:
-
Preeclampsia
- I.C.V:
-
Intra-cerebro-ventricular
- LPS:
-
Lipopolysaccharide
- LTP:
-
Long-term potentiation
- MDA:
-
Malondialdehyde
- MI:
-
Myocardial infarction
- mPFC:
-
Medial prefrontal cortex
- Nrf2:
-
Nuclear factor erythroid-2-related factor 2
- 5/6Nx:
-
5/6 nephrectomy
- ROS:
-
Reactive oxygen species
- RVLM:
-
Rostral ventrolateral medulla
- sCr:
-
Serum creatinine
- TBA:
-
thiobarbituric acid
- WB:
-
Western blot
- Znpp:
-
Zinc protoporphyrin IX
References
Adach W, Olas B (2020) A comparison of multifunctional donors of carbon monoxide: their anticoagulant, antioxidant, anti-aggregatory and cytotoxicity activities in an in vitro model. Nitric Oxide 97:20–26
Askari H, Abazari MF, Ghoraeian P et al (2018) Ameliorative effects of hydrogen sulfide (NaHS) on chronic kidney disease-induced brain dysfunction in rats: implication on role of nitric oxide (NO) signaling. Metab Brain Dis 33:1945–1954
Aziz NM, Elbassuoni EA, Kamel MY, Ahmed SM (2020) Hydrogen sulfide renal protective effects: possible link between hydrogen sulfide and endogenous carbon monoxide in a rat model of renal injury. Cell Stress Chaperones 25:211–221
Babaei H, Alibabrdel M, Asadian S, Siavashi V, Jabarpour M, Nassiri SM (2018) Increased circulation mobilization of endothelial progenitor cells in preterm infants with retinopathy of prematurity. J Cell Biochem 119:6575–6583
Bani-Hani MG, Greenstein D, Mann BE, Green CJ, Motterlini R (2006) A carbon monoxide-releasing molecule (CORM-3) attenuates lipopolysaccharide-and interferon-gamma-induced inflammation in microglia. Pharmacol Rep 58:132
Bannenberg GL, Vieira HL (2009) Therapeutic applications of the gaseous mediators carbon monoxide and hydrogen sulfide. Expert Opin Ther Pat 19:663–682
Bronas UG, Puzantian H, Hannan M (2017) Cognitive impairment in chronic kidney disease: vascular milieu and the potential therapeutic role of exercise. Biomed Res Int 2017:2726369. https://doi.org/10.1155/2017/2726369
Campbell NK, Fitzgerald HK, Dunne A (2021) Regulation of inflammation by the antioxidant haem oxygenase 1. Nat Rev Immunol 21:411–425
Donertas Ayaz B, Oliveira AC, Malphurs WL et al (2021) Central administration of hydrogen sulfide donor NaHS reduces Iba1-positive cells in the PVN and attenuates rodent angiotensin II hypertension. Front NeuroSci 15:690919
Du J, Wang P, Gou Q et al (2022) Hydrogen sulfide ameliorated preeclampsia via suppression of toll-like receptor 4-activated inflammation in the rostral ventrolateral medulla of rats. Biomed Pharmacother 150:113018
Farrugia G, Szurszewski JH (2014) Carbon monoxide, hydrogen sulfide, and nitric oxide as signaling molecules in the gastrointestinal tract. Gastroenterology 147:303–313
Gava AL, Freitas FP, Balarini CM, Vasquez EC, Meyrelles SS (2012) Effects of 5/6 nephrectomy on renal function and blood pressure in mice. Int J Physiol Pathophysiol Pharmacol 4:167
Greabu M, Totan A, Miricescu D, Radulescu R, Virlan J, Calenic B (2016) Hydrogen sulfide, oxidative stress and periodontal diseases: a concise review. Antioxidants 5:3
Habibitabar E, Moridi H, Shateri H, Karimi SA, Salehi I, Komaki A, Sarihi A (2020) Chronic NaHS treatment improves spatial and passive avoidance learning and memory and anxiety-like behavior and decreases oxidative stress in rats fed with a high-fat diet. Brain Res Bull 164:380–391
Hadadha M, Vakili A, Bandegi AR (2015) Effect of the inhibition of hydrogen sulfide synthesis on ischemic injury and oxidative stress biomarkers in a transient model of focal cerebral ischemia in rats. J Stroke Cerebrovasc Dis 24:2676–2684
Hamidizad Z, Kadkhodaee M, Karimian SM et al (2022) Therapeutic effects of CORM3 and NaHS in chronic kidney disease induced cognitive impairment via the interaction between carbon monoxide and hydrogen sulfide on Nrf2/HO-1 signaling pathway in rats. Chemico-Biol Interact 368:110217
Han Y, Qin J, Chang X, Yang Z, Du J (2006) Hydrogen sulfide and carbon monoxide are in synergy with each other in the pathogenesis of recurrent febrile seizures. Cell Mol Neurobiol 26:101–107
Huang YQ, Jin HF, Zhang H, Tang CS, Du JB (2021) Interaction among hydrogen sulfide and other gasotransmitters in mammalian physiology and pathophysiology. Adv Exp Med Biol 1315:205–236. https://doi.org/10.1007/978-981-16-0991-6_9
Ismailova A, Kuter D, Bohle DS, Butler IS (2018) An overview of the potential therapeutic applications of CO-releasing molecules. Bioinorg Chem Appl 2018:8547364. https://doi.org/10.1155/2018/8547364
Jabarpour M, Siavashi V, Asadian S, Babaei H, Jafari SM, Nassiri SM (2018) Hyperbilirubinemia-induced pro-angiogenic activity of infantile endothelial progenitor cells. Microvasc Res 118:49–56
Jin Hf, Du Jb, Li Xh, Wang Yf, Liang Yf, Tang Cs (2006) Interaction between hydrogen sulfide/cystathionine γ-lyase and carbon monoxide/heme oxygenase pathways in aortic smooth muscle cells 1. Acta Pharmacol Sin 27:1561–1566
Jing W, Jabbari B, Vaziri ND (2018) Uremia induces upregulation of cerebral tissue oxidative/inflammatory cascade, down-regulation of Nrf2 pathway and disruption of blood brain barrier. Am J translational Res 10:2137
Kajimura M, Fukuda R, Bateman RM, Yamamoto T, Suematsu M (2010) Interactions of multiple gas-transducing systems: hallmarks and uncertainties of CO, NO, and H2S gas biology. Antioxid Redox Signal 13:157–192
Kianian F, Seifi B, Kadkhodaee M, Sadeghipour HR, Ranjbaran M (2022) Nephroprotection through modifying the apoptotic tnf-α/erk1/2/bax signaling pathway and oxidative stress by long-term sodium hydrosulfide administration in ovalbumin-induced chronic asthma. Immunol Investig 51:602–618
Kitada M, Xu J, Ogura Y, Monno I, Koya D (2020) Manganese superoxide dismutase dysfunction and the pathogenesis of kidney disease. Front Physiol 11:755
Kosuge Y, Osada N, Shimomura A et al (2018) Relevance of the hippocampal endoplasmic reticulum stress response in a mouse model of chronic kidney disease. Neurosci Lett 677:26–31
Kovalčíková A, Gyurászová M, Vavrincová-Yaghi D et al (2018) Oxidative stress in the brain caused by acute kidney injury. Metab Brain Dis 33:961–967
Kshirsagar V, Thingore C, Gursahani M, Gawali N, Juvekar A (2021) Hydrogen sulfide ameliorates lipopolysaccharide-induced memory impairment in mice by reducing apoptosis, oxidative, and inflammatory effects. Neurotox Res 39:1310–1322
Kujal P, Vernerová Z (2008) 5/6 nephrectomy as an experimental model of chronic renal failure and adaptation to reduced nephron number. Cesk Fysiol 57:104–109
Li N, Wang MJ, Jin S et al (2016) The H2S donor NaHS changes the expression pattern of H2S-producing enzymes after myocardial infarction. Oxid Med Cell Longev 2016:6492469. https://doi.org/10.1155/2016/6492469
Ling K, Men F, Wang W-C, Zhou Y-Q, Zhang H-W, Ye D-W (2017) Carbon monoxide and its controlled release: therapeutic application, detection, and development of carbon monoxide releasing molecules (CORMs) miniperspective. J Med Chem 61:2611–2635
Liu WQ, Chai C, Li XY, Yuan WJ, Wang WZ, Lu Y (2011) The cardiovascular effects of central hydrogen sulfide are related to K(ATP) channels activation. Physiol Res 60:729–738. https://doi.org/10.33549/physiolres.932092
Liu S, Xin D, Wang L et al (2017) Therapeutic effects of L-Cysteine in newborn mice subjected to hypoxia-ischemia brain injury via the CBS/H2S system: role of oxidative stress and endoplasmic reticulum stress. Redox Biol 13:528–540
Ma Y, Yang X, Wang H et al (2021) CBS-derived H2S facilitates host colonization of Vibrio cholerae by promoting the iron-dependent catalase activity of KatB. PLoS Pathog 17:e1009763
Magierowski M, Magierowska K, Hubalewska-Mazgaj M et al (2016) Interaction between endogenous carbon monoxide and hydrogen sulfide in the mechanism of gastroprotection against acute aspirin-induced gastric damage. Pharmacol Res 114:235–250
Magierowski M, Magierowska K, Hubalewska-Mazgaj M et al (2018) Cross-talk between hydrogen sulfide and carbon monoxide in the mechanism of experimental gastric ulcers healing, regulation of gastric blood flow and accompanying inflammation. Biochem Pharmacol 149:131–142
Matta SMd, Janaina Matos M, Kummer AM, Barbosa IG, Teixeira AL, Silva ACS (2014) Cognitive alterations in chronic kidney disease: an update. Braz J Nephrol 36:241–245
Olson KR, Donald JA (2009) Nervous control of circulation–the role of gasotransmitters, NO, CO, and H2S. Acta Histochem 111:244–256
Paul BD, Snyder SH (2015) Modes of physiologic H2S signaling in the brain and peripheral tissues. Antioxid Redox Signal 22:411–423
Pérez-de-Puig I, Martín A, Gorina R, de la Rosa X, Martinez E, Planas AM (2013) Induction of hemeoxygenase-1 expression after inhibition of hemeoxygenase activity promotes inflammation and worsens ischemic brain damage in mice. Neuroscience 243:22–32
Prabhakar NR (2012) Carbon monoxide (CO) and hydrogen sulfide (H2S) in hypoxic sensing by the carotid body. Respir Physiol Neurobiol 184:165–169
Queiroga CS, Vercelli A, Vieira HL (2015) Carbon monoxide and the CNS: challenges and achievements. Br J Pharmacol 172:1533–1545
Robert K, Vialard F, Thiery E, Toyama K, Sinet P-M, Janel N, London J (2003) Expression of the cystathionine β synthase (CBS) gene during mouse development and immunolocalization in adult brain. J Histochem Cytochem 51:363–371
Schwarz AJ, Danckaert A, Reese T et al (2006) A stereotaxic MRI template set for the rat brain with tissue class distribution maps and co-registered anatomical atlas: application to pharmacological MRI. NeuroImage 32:538–550
Shintani T, Iwabuchi T, Soga T et al (2009) Cystathionine β-synthase as a carbon monoxide–sensitive regulator of bile excretion. Hepatology 49:141–150
Siavashi V, Nassiri SM, Rahbarghazi R, Vafaei R, Sariri R (2016) ECM-dependence of endothelial progenitor cell features. J Cell Biochem 117:1934–1946
Small DM, Coombes JS, Bennett N, Johnson DW, Gobe GC (2012) Oxidative stress, anti-oxidant therapies and chronic kidney disease. Nephrology 17:311–321
Soriano RN, Kwiatkoski M, Batalhão ME, Branco LGdS, Carnio EC (2012) Interaction between the carbon monoxide and nitric oxide pathways in the locus coeruleus during fever. Neuroscience 206:69–80
Tian L, Qiu H, Pun VC, Ho K-F, Chan CS, Ignatius T (2015) Carbon monoxide and stroke: a time series study of ambient air pollution and emergency hospitalizations. Int J Cardiol 201:4–9
Uddin MJ, Pak ES, Ha H (2018) Carbon monoxide releasing molecule-2 protects mice against acute kidney injury through inhibition of ER stress. Korean J Physiol Pharmacol 22:567–575
Voskoboeva E, Semyachkina A, Yablonskaya M, Nikolaeva E (2018) Homocystinuria due to cystathionine beta-synthase (CBS) deficiency in Russia: molecular and clinical characterization. Mol Genet Metab Rep 14:47–54
Wang J, Zhang D, Fu X et al (2018) Carbon monoxide-releasing molecule-3 protects against ischemic stroke by suppressing neuroinflammation and alleviating blood-brain barrier disruption. J Neuroinflammation 15:188. https://doi.org/10.1186/s12974-018-1226-1
Wang R (2004) Signal transduction and the gasotransmitters: NO, CO, and H2S in biology and medicine. Springer Science & Business Media
Watanabe K, Watanabe T, Nakayama M (2014) Cerebro-renal interactions: impact of uremic toxins on cognitive function. Neurotoxicology 44:184–193
Wesseling S, Fledderus JO, Verhaar MC, Joles JA (2015) Beneficial effects of diminished production of hydrogen sulfide or carbon monoxide on hypertension and renal injury induced by NO withdrawal. Br J Pharmacol 172:1607–1619
Wilson JL, Fayad Kobeissi S, Oudir S et al (2014) Design and synthesis of new hybrid molecules that activate the transcription factor Nrf2 and simultaneously release carbon monoxide. Chemistry–A Eur J 20:14698–14704
Wu X, Liu C, Wang J, Guan Y, Song L, Chen R, Gong M (2022) Catalpol exerts antidepressant-like effects by enhancing anti-oxidation and neurotrophy and inhibiting neuroinflammation via activation of HO-1. Neurochem Res 47:2975–2991
Wu J, Zhao Y-M, Deng Z-K (2018) Tangeretin ameliorates renal failure via regulating oxidative stress, NF-κB–TNF-α/iNOS signalling and improves memory and cognitive deficits in 5/6 nephrectomized rats. Inflammopharmacology 26:119–132
Yabluchanskiy A, Sawle P, Homer-Vanniasinkam S, Green CJ, Foresti R, Motterlini R (2012) CORM-3, a carbon monoxide-releasing molecule, alters the inflammatory response and reduces brain damage in a rat model of hemorrhagic stroke. Crit Care Med 40:544–552
Yaghmai R, Kashani AH, Geraghty MT et al (2002) Progressive cerebral edema associated with high methionine levels and betaine therapy in a patient with cystathionine β-synthase (CBS) deficiency. Am J Med Genet 108:57–63
Yamamoto M, Shimizu T, Zou S, Shimizu S, Higashi Y, Fujieda M, Saito M (2020) Brain hydrogen sulfide suppresses the micturition reflex via brain GABA receptors in rats. Nitric Oxide 104:44–50
Zalba G, Fortuño A, Díez J (2006) Oxidative stress and atherosclerosis in early chronic kidney disease. Nephrol Dial Transplant 21:2686–2690
Funding
This research was supported by a grant (no = 49186) from Tehran University of Medical Sciences.
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Material preparation, data collection and analysis were performed by Zeinab Hamidizad, Mehri Kadkhodaee, Farzaneh Kianian, Mina Ranjbaran, Behjat Seifi1. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval
All processes of dealing with the animals were conducted in accordance with the Animal Ethics Committee of the Faculty of Medicine, Tehran University of Medical Sciences (Approval ID: IR.TUMS.MEDICINE.REC.1399.511).
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Conflict of interest
The authors declare that they have no conflict of interest. Availability of data and material the data that support the findings of this study are available from the corresponding author upon reasonable request.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Hamidizad, Z., Kadkhodaee, M., Kianian, F. et al. The effects of CORM3 or NaHS on the oxidative stress caused by chronic kidney disease in rats: potential interaction between CO and H2S signaling pathway. Metab Brain Dis 38, 2653–2664 (2023). https://doi.org/10.1007/s11011-023-01264-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11011-023-01264-w