Abstract
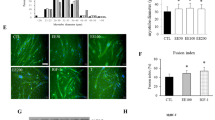
Follistatin (FS) is a high affinity activin-binding protein, neutralizing the effects of the Transforming Growth Factor-beta (TGF-β) superfamily members, as myostatin (MSTN). Since MSTN emerged as a negative regulator, FS has been considered as a stimulator of skeletal muscle growth and differentiation. Here, we studied the effect of FS administration on the Ca2+-homeostasis of differentiating C2C12 skeletal muscle cells. FS-treatment increased the fusion index, the size of terminally differentiated myotubes, and transiently elevated the expression of the calcium-dependent protein phosphatase, calcineurin, at the beginning of differentiation. Functional experiments did not detect any alterations in the Ca2+ transients following the stimulation by KCl or caffeine in myotubes. On the other hand, decreased Ca2+-uptake capability was determined by calculating the maximal pump rate (332 ± 17 vs. 279 ± 11 µM/s, in control and FS-treated myotubes, respectively; p < 0.05). In the same way, the expression and ATPase activity of the neonatal sarcoplasmic/endoplasmic reticulum Ca2+ATPase (SERCA1b) were decreased (0.59 ± 0.01 vs. 0.19 ± 0.01 mM ATP/min, in control and FS-treated myotubes, respectively; p < 0.05). However, the expression level of other proteins involved in Ca2+-homeostasis and differentiation (calsequestrin, STIM1, MyoD) were not affected. Our results suggest that the FS controlled myotube growth is paralleled with the tight regulation of cytosolic calcium concentration, and the decline of SERCA1b appears to be one of the key components in this process.
Graphical abstract







Similar content being viewed by others
Abbreviations
- BCA:
-
Bicinchoninic acid
- BSA:
-
Bovine serum albumin
- CaN:
-
Calcineurin
- CPA:
-
Cyclopiazonic acid
- CSQ:
-
Calsequestrin
- ECL:
-
Enhanced chemiluminescence
- FBS:
-
Foetal bovine serum
- FITC:
-
Fluoresceinisothiocyanate
- FS:
-
Follistatin
- HS:
-
Horse serum
- MSTN:
-
Myostatin
- PBS:
-
Phosphate-buffered saline
- RyR:
-
Ryanodine receptor
- SDS–PAGE:
-
Sodium dodecyl sulphate polyacrylamide gel electrophoresis
- SERCA:
-
Sarcoplasmic/endoplasmic reticulum Ca2+-ATPase
- SOCE:
-
Store-operated Ca2+ entry
- SR:
-
Sarcoplasmic reticulum
- STIM:
-
Stromal interaction molecule
- TGF-β:
-
Transforming Growth Factor-beta
References
Allen DL, Uyenishi JJ, Cleary AS, Mehan RS, Lindsay SF, Reed JM (2010) Calcineurin activates interleukin-6 transcription in mouse skeletal muscle in vivo and in C2C12 myotubes in vitro. Am J Physiol Regul Integr Comp Physiol 298(1):R198–R210. doi:10.1152/ajpregu.00325.2009
Alzuherri H, Chang KC (2003) Calcineurin activates NF-kappaB in skeletal muscle C2C12 cells. Cell Signal 15(5):471–478. doi:10.1016/S0898-6568(02)00120-1
Amthor H, Christ B, Rashid-Doubell F, Kemp CF, Lang E, Patel K (2002) Follistatin regulates bone morphogenetic protein-7 (BMP-7) activity to stimulate embryonic muscle growth. Dev Biol 243(1):115–127. doi:10.1006/dbio.2001.0555
Amthor H, Nicholas G, McKinnell I, Kemp CF, Sharma M, Kambadur R et al (2004) Follistatin complexes myostatin and antagonises myostatin-mediated inhibition of myogenesis. Dev Biol 270(1):19–30. doi:10.1016/j.ydbio.2004.01.046
Berchtold MW, Brinkmeier H, Müntener M (2000) Calcium ion in skeletal muscle: its crucial role for muscle function, plasticity, and disease. Physiol Rev 80(3):1215–1265
Bollo M, Paredes RM, Holstein D, Zheleznova N, Camacho P, Lechleiter JD (2010) Calcineurin interacts with PERK and dephosphorylates calnexin to relieve ER stress in mammals and frogs. PLoS One 5(8):e11925. doi:10.1371/journal.pone.0011925
Chen JL, Walton KL, Winbanks CE, Murphy KT, Thomson RE, Makanji Y et al (2014) Elevated expression of activins promotes muscle wasting and cachexia. FASEB J 28(4):1711–1723. doi:10.1096/fj.13-245894
Fodor, J., Tóth, A., Vincze, J., Oláh, T., Dienes, B., Zádor, E., et al. (2015) The effect of follistatin on the calcium homeostasis and differentiation of the C2C12 skeletal muscle cells. J. Muscle Res Cell Motil 36:131. doi:10.1007/s10974-015-9407-3
Furutani Y, Murakami M, Funaba M (2009) Differential responses to oxidative stress and calcium influx on expression of the transforming growth factor-beta family in myoblasts and myotubes. Cell Biochem Funct 27(8):578–582. doi:10.1002/cbf0.1614
Geyer N, Diszházi G, Csernoch L, Jóna I, Almássy J (2015) Bile acids activate ryanodine receptors in pancreatic acinar cells via a direct allosteric mechanism. Cell Calcium 58(2):160–170. doi:10.1016/j.ceca.2015.03.009
Grynkiewicz G, Poenie M, Tsien R (1985) A new generation of Ca2+ indicators with greatly improved fluorescence properties. J Biol Chem 260:3440–3450
Jain SS, Paglialunga S, Vigna C, Ludzki A, Herbst EA, Lally JS, Schrauwen P, Hoeks J, Tupling AR, Bonen A, Holloway GP (2014) High-fat diet-induced mitochondrial biogenesis is regulated by mitochondrial-derived reactive oxygen species activation of CaMKII. Diabetes 63(6):1907–1913. doi:10.2337/db13-0816
Kanno Y, Ota R, Someya K, Kusakabe T, Kato K, Inouye Y (2013) Selective androgen receptor modulator, YK11, regulates myogenic differentiation of C2C12 myoblasts by follistatin expression. Biol Pharm Bull 36(9):1460–1465. doi:10.1248/bpb.b13-00231
Kielbasa OM, Reynolds JG, Wu CL, Snyder CM, Cho MY, Weiler H et al (2011) Myospryn is a calcineurin-interacting protein that negatively modulates slow-fiber-type transformation and skeletal muscle regeneration. FASEB J 25(7):2276–2286. doi:10.1096/fj.10-169219
Kocamiş H, Gulmez N, Aslan S, Nazli M (2004) Follistatin alters myostatin gene expression in C2C12 muscle cells. Acta Vet Hung 52(2):135–141. doi:10.1556/AVet.52.2004.2.1
Langley B, Thomas M, Bishop A, Sharma M, Gilmour S, Kambadur R (2002) Myostatin inhibits myoblast differentiation by down-regulating MyoD expression. J Biol Chem 20 277(51):49831–49840. doi:10.1074/jbc.M204291200
Lara-Pezzi E, Felkin LE, Birks EJ, Sarathchandra P, Panse KD, George R et al (2008) Expression of follistatin-related genes is altered in heart failure. Endocrinology 149(11):5822–5827. doi:10.1210/en.2008-0151
Lee SJ. (2007) Sprinting without myostatin: a genetic determinant of athletic prowess. Trends Genet 23:475–477. doi:10.1016/j.tig.2007.08.008
Lee SJ, McPherron AC (2001) Regulation of myostatin activity and muscle growth. Proc Natl Acad Sci USA 98(16):9306–9311. doi:10.1073/pnas.151270098
Lee SJ, Lee YS, Zimmers TA, Soleimani A, Matzuk MM, Tsuchida K et al (2010) Regulation of muscle mass by follistatin and activins. Mol Endocrinol 24(10):1998–2008. doi:10.1210/me.2010-0127
Lee SJ, Huynh TV, Lee YS, Sebald SM, Wilcox-Adelman SA, Iwamori N et al (2012) Role of satellite cells versus myofibers in muscle hypertrophy induced by inhibition of the myostatin/activin signaling pathway. Proc Natl Acad Sci USA 109(35):E2353–E2360. doi:10.1073/pnas.1206410109
Lee KJ, Hyun C, Woo JS, Park CS, Kim do H, Lee EH (2014) Stromal interaction molecule 1 (STIM1) regulates sarcoplasmic/endoplasmic reticulum Ca2+-ATPase 1a (SERCA1a) in skeletal muscle. Pflugers Arch 466(5):987–1001. doi:10.1007/s00424-013-1361-6
Lin C, Zhao X, Sun D, Zhang L, Fang W, Zhu T, Wang Q, Liu B, Wei S, Chen G, Xu Z, Gao X (2016) Transcriptional activation of FS by Nrf2 protects pulmonary epithelial cells against silica nanoparticle-induced oxidative stress. Sci Rep 6:21133. doi:10.1038/srep21133
Liu D, Black BL, Derynck R (2001) TGF-beta inhibits muscle differentiation through functional repression of myogenic transcription factors by Smad3. Genes Dev 15(22):2950–2966. doi:10.1101/gad.925901
Madaro L, Marrocco V, Carnio S, Sandri M, Bouché M (2013) Intracellular signaling in ER stress-induced autophagy in skeletal muscle cells. FASEB J 27(5):1990–2000. doi:10.1096/fj.12-215475
Maruyama K, MacLennan DH (1988) Mutation of aspartic acid-351, lysine-352, and lysine-515 alters the Ca2+ transport activity of the Ca2+-ATPase expressed in COS-1 cells. Proc Natl Acad Sci USA 85(10):3314–3318
Massagué J, Chen YG (2000) Controlling TGF-beta signaling. Genes Dev 14(6):627–644. doi:10.1101/gad.14.6.627
McPherron AC, Lee SJ (1997) Double muscling in cattle due to mutations in themyostatin gene. Proc Natl Acad Sci USA 94(23):12457–12461
McPherron AC, Lawler AM, Lee SJ (1997) Regulation of skeletal muscle mass in mice by a new TGF-beta superfamily member. Nature 387(6628):83–90. doi:10.1038/387083a0
Medeiros EF, Phelps MP, Fuentes FD, Bradley TM (2009) Overexpression of follistatin in trout stimulates increased muscling. Am J Physiol Regul Integr Comp Physiol 297(1):R235–R242. doi:10.1152/ajpregu.91020.2008
Murphy RM, Larkins NT, Mollica JP, Beard NA, Lamb GD (2009) Calsequestrin content and SERCA determine normal and maximal Ca2+ storage levels in sarcoplasmic reticulum of fast- and slow-twitch fibres of rat. J Physiol 587(2):443–460. doi:10.1113/jphysiol.2008.163162
Nakanishi K, Dohmae N, Morishima N (2007) Endoplasmic reticulum stress increases myofiber formation in vitro. FASEB J 21(11):2994–3003. doi:10.1096/fj.06-6408com
Naro F, De Arcangelis V, Coletti D, Molinaro M, Zani B, Vassanelli S et al (2003) Increase in cytosolic Ca2+ induced by elevation of extracellular Ca2+ in skeletal myogenic cells. Am J Physiol Cell Physiol 284(4):C969–C976. doi:10.1152/ajpcell.00237.2002
Oláh T, Fodor J, Ruzsnavszky O, Vincze J, Berbey C, Allard B et al (2011) Overexpression of transient receptor potential canonical type 1 (TRPC1) alters both store-operated calcium entry and depolarization-evoked calcium signals in C2C12 cells. Cell Calcium 49(6):415–425. doi:10.1016/j.ceca.2011.03.012
Olson EN, Williams RS (2000) Remodeling muscles with calcineurin. Bioessays 22(6):510–9 (review. Erratum in: Bioessays 22(11):1049). doi:10.1002/(SICI)1521-1878(200006)22:6<510::AID-BIES4>3.0.CO;2-1
Pan Y, Zvaritch E, Tupling AR, Rice WJ, de Leon S, Rudnicki M et al (2003) Targeted disruption of the ATP2A1 gene encoding the sarco(endo)plasmic reticulum Ca2+ ATPase isoform 1 (SERCA1) impairs diaphragm function and is lethal in neonatal mice. J Biol Chem 278(15):13367–13375. doi:10.1074/jbc.M213228200
Putney JW Jr (1986) A model for receptor-regulated calcium entry. Cell Calcium 7(1):1–12
Putney JW (2013) Alternative forms of the store-operated calcium entry mediators, STIM1 and Orai1. Curr Top Membr 71:109–123. doi:10.1016/B978-0-12-407870-3.00005-6
Sakuma K, Yamaguchi A (2010) The functional role of calcineurin in hypertrophy, regeneration, and disorders of skeletal muscle. J Biomed Biotechnol 2010:721219. doi:10.1155/2010/721219
Sárközi S, Almássy J, Lukács B, Dobrosi N, Nagy G, Jóna I (2007) Effect of natural phenol derivatives on skeletal type sarcoplasmic reticulum Ca2+-ATPase and ryanodine receptor. J Muscle Res Cell Motil 28(2–3):167–174. doi:10.1007/s10974-007-9113-x
Stiber JA, Rosenberg PB (2011) The role of store-operated calcium influx in skeletal muscle signaling. Cell Calcium 49(5):341–349. doi:10.1016/j.ceca.2010.11.012
Tóth A, Fodor J, Vincze J, Oláh T, Juhász T, Zákány R, Csernoch L, Zádor E (2015) The effect of SERCA1b silencing on the differentiation and calcium homeostasis of C2C12 skeletal muscle cells. PLoS One 10(4):e0123583. doi:10.1371/journal.pone.0123583
Wang Y, Xu L, Duan H, Pasek DA, Eu JP, Meissner G (2006) Knocking down type 2 but not type 1 calsequestrin reduces calcium sequestration and release in C2C12 skeletal muscle myotubes. J Biol Chem 281(22):15572–15581. doi:10.1074/jbc.M600090200
Winbanks CE, Weeks KL, Thomson RE, Sepulveda PV, Beyer C, Qian H et al (2012) Follistatin mediated skeletal muscle hypertrophy is regulated by Smad3 and mTOR independently of myostatin. J Cell Biol 197(7):997–1008. doi:10.1083/jcb.201109091
Zádor E (2008) dnRas stimulates autocrine-paracrine growth of regenerating muscle via calcineurin-NFAT-IL-4 pathway. Biochem Biophys Res Commun 375(2):265–270. doi:10.1016/j.bbrc.2008.08.024
Zádor E, Kósa M (2015) The neonatal sarcoplasmic/endoplasmic reticulum calcium ATPase (SERCA1b): a neglected pump in scope. Pflugers Arch 467(7):1395–1401. doi:10.1007/s00424-014-1671-3
Zádor E, Vangheluwe P, Wuytack F (2007) The expression of the neonatal sarcoplasmic reticulum Ca2+ pump (SERCA1b) hints to a role in muscle growth and development. Cell Calcium 41(4):379–388. doi:10.1016/j.ceca.2006.08.001
Zádor E, Owsianik G, Wuytack F (2011) Silencing SERCA1b in a few fibers stimulates growth in the entire regenerating soleus muscle. Histochem Cell Biol 135(1):11–20. doi:10.1007/s00418-010-0766-y
Zhao Y, Ogawa H, Yonekura S, Mitsuhashi H, Mitsuhashi S, Nishino I, et al. (2015) Functional analysis of SERCA1b, a highly expressed SERCA1 variant in myotonic dystrophy type 1 muscle. Biochim Biophys Acta 1852(10 PtA):2042–2047. doi:10.1016/j.bbadis.2015.07.006
Zimmers TA, Davies MV, Koniaris LG, Haynes P, Esquela AF, Tomkinson KN et al (2002) Induction of cachexia in mice by systemically administered myostatin. Science 296(5572):1486–1488. doi:10.1126/science.1069525
Acknowledgements
JA and JF were the recipients of the Janos Bolyai Research Scholarship of the Hungarian Academy of Sciences and JA the Lajos Szodoray Scholarship of the University of Debrecen, Faculty of Medicine. The work was supported by the National Research, Development and Innovation Office (NKFIH K115461, and PD 112199). We are grateful to Luca Mendler (Department of Biochemistry, Faculty of Medicine, University of Szeged) for support. The publication is supported by the GINOP-2.3.2-15-2016-00040 project. The project is co-financed by the European Union and the European Regonal Development Fund.
Author information
Authors and Affiliations
Contributions
Substantial contributions to the conception or design of the work; or the acquisition, analysis, or interpretation of data for the work: JF, AG, TO, JA, EZ, LC. Drafting the work or revising it critically for important intellectual content: JF, AG, TO, JA, EZ, LC. Final approval of the version to be published: JF, AG, TO, JA, EZ, LC. Agreement to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved: JF, AG, TO, JA, EZ, LC.
Corresponding author
Ethics declarations
Conflict of interest
All authors disclose that there are neither any financial nor any personal relationships with other people or organisations that could inappropriately influence (bias) their work. There are no conflicts of interests.
Rights and permissions
About this article
Cite this article
Fodor, J., Gomba-Tóth, A., Oláh, T. et al. Follistatin treatment suppresses SERCA1b levels independently of other players of calcium homeostasis in C2C12 myotubes. J Muscle Res Cell Motil 38, 215–229 (2017). https://doi.org/10.1007/s10974-017-9474-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10974-017-9474-8